Extracellular Polymeric Substances (EPS): The Architectural Scaffold of Biofilms and Target for Advanced Therapeutics
Extracellular Polymeric Substances (EPS) form the fundamental, self-produced matrix of microbial biofilms, determining their structural integrity, functional properties, and formidable resistance to antimicrobials and host defenses.
Extracellular Polymeric Substances (EPS): The Architectural Scaffold of Biofilms and Target for Advanced Therapeutics
Abstract
Extracellular Polymeric Substances (EPS) form the fundamental, self-produced matrix of microbial biofilms, determining their structural integrity, functional properties, and formidable resistance to antimicrobials and host defenses. This article synthesizes current research for a scientific audience, exploring the complex composition and architecture of EPS, advanced methodologies for its study and disruption, innovative strategies for biofilm control, and the validation of EPS-targeting therapies. We examine how a deep understanding of EPS mechanics, constituent interactions, and inhibition mechanisms is paving the way for novel anti-biofilm agents and treatment paradigms to combat persistent infections and antimicrobial resistance.
Deconstructing the EPS Matrix: Composition, Architecture, and Core Functions
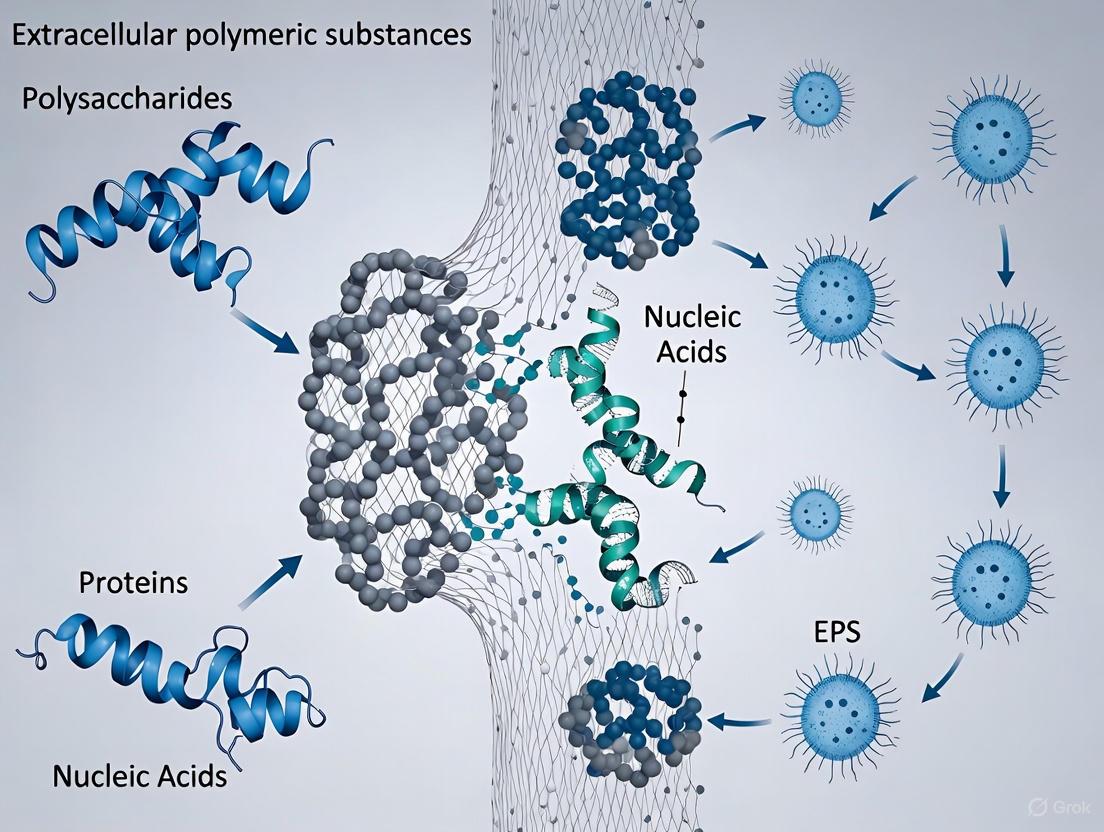
Extracellular Polymeric Substances (EPS) represent the fundamental construction material of microbial biofilms, forming a complex, hydrated matrix that provides structural and functional integrity to these microbial societies. This in-depth technical guide delineates the core components of EPS—a dynamic mixture of polysaccharides, proteins, nucleic acids, and lipids—and elucidates their synergistic role in establishing the biofilm architecture. We provide a comprehensive overview of advanced analytical techniques and detailed experimental protocols for the extraction, quantification, and characterization of EPS constituents. Designed for researchers, scientists, and drug development professionals, this review synthesizes current methodologies and data to facilitate a deeper understanding of structure-function relationships within the EPS matrix, thereby informing the development of targeted anti-biofilm strategies.
A biofilm is a community of microbial cells embedded in a matrix of extracellular polymeric substances (EPS) and is considered the predominant mode of microbial life. The EPS matrix is a biological barrier produced by a variety of microorganisms primarily for defense and as a consequence of their physiological processes [1]. This matrix is not merely a passive scaffold but a dynamic, functional, and critical component that determines the physicochemical properties of a biofilm [2]. It provides compositional support and protection for microbial communities from harsh environments, including antimicrobial agents and host immune responses [2]. The EPS establishes the structural integrity of biofilms and is a fundamental component that dictates their resilience [2]. Comprehending the intricate composition and organization of EPS is therefore paramount for any research aimed at controlling biofilm-related infections or harnessing their beneficial applications.
Core Components of the EPS Matrix
The EPS matrix is a complex amalgamation of biological polymers, primarily secreted by the microorganisms themselves. These components interact through weak physicochemical forces to form a cohesive, gel-like network that encompasses the microbial cells [3]. The composition is highly dynamic and can vary significantly based on the microbial species, environmental conditions, and substrate availability [4]. The table below summarizes the primary constituents and their key functions within the biofilm matrix.
Table 1: Major Constituents of Extracellular Polymeric Substances (EPS) and Their Functions
| EPS Component | Primary Functions | Key Characteristics |
|---|---|---|
| Polysaccharides | Structural scaffolding, adhesion, cohesion, water retention, nutrient entrapment [3] [5]. | Often the most abundant component; can be neutral or anionic (e.g., containing uronic acids); highly diverse in monomer composition [2]. |
| Proteins | Structural support, enzymatic activity (exoenzymes), adhesion, cellular recognition [3] [5]. | Include structural proteins and extracellular enzymes (exoenzymes) for nutrient acquisition; can be a dominant component in some biofilms [2]. |
| Nucleic Acids | Structural integrity, genetic information transfer, horizontal gene transfer, cohesion [3] [5]. | Referred to as extracellular DNA (eDNA); released via cell lysis or active secretion; contributes to antimicrobial resistance spread [5]. |
| Lipids | Hydrophobicity, structural organization, signaling [3] [4]. | Less studied but a quantifiable constituent; can influence interaction with hydrophobic surfaces [4]. |
| Other Components | Structural stability, ion exchange [2]. | Includes humic substances and various minerals like calcite (CaCO3) resulting from biomineralization [2]. |
Quantitative Composition of EPS
The proportional makeup of EPS is not fixed. A study analyzing 10 soil bacterial and 10 soil fungal species found that the concentration of specific EPS constituents was strongly influenced by the microbial type and environmental conditions, such as carbon source (glycerol vs. starch) and the presence of a surface for attachment (quartz matrix) [4]. The following table presents quantitative data from this study, illustrating the variability in EPS composition.
Table 2: Quantitative Analysis of EPS Constituents from Soil Bacteria and Fungi Grown Under Different Conditions [4]
| Culture Condition | Total Carbohydrates (µg/ml) | Total Proteins (µg/ml) | DNA (µg/ml) | Amino Sugars (µg/ml) | Key Findings |
|---|---|---|---|---|---|
| Glycerol Media | Variable | Variable | Variable | Variable | Serves as a labile carbon source. |
| Starch Media | Higher than in glycerol | Variable | Variable | Variable | Led to a higher EPS-carbohydrate/protein ratio. |
| With Quartz Matrix | Increased | Variable | Variable | Variable | Enhanced EPS production, particularly carbohydrates. |
Advanced Methodologies for EPS Analysis
A multifaceted approach is required to fully characterize the structural, biochemical, and functional properties of EPS. The following experimental workflows and techniques are central to modern EPS research.
Experimental Workflow for EPS Analysis
The typical pipeline for EPS analysis involves sample preparation, EPS extraction, and a suite of analytical techniques to quantify and characterize the individual components.
Detailed Experimental Protocols
EPS Extraction Using Cation Exchange Resin (CER)
This is a widely used chemical method for extracting EPS from biofilms [4].
- Principle: The CER disrupts the EPS matrix by exchanging divalent cations (e.g., Ca²⁺, Mg²⁺) that cross-link polymer chains, particularly polysaccharides, thereby releasing the EPS into solution.
- Procedure:
- Collect Aliquots: Obtain 10 ml aliquots from the microbial culture (e.g., after a 4-day incubation) [4].
- Add CER: Add the recommended amount of cation exchange resin (e.g., Amberlite HPR1100). The optimal resin dosage must be determined for the specific microbial culture, often based on volatile suspended solids [4].
- Mix: Stir the mixture gently for a specified period (e.g., 2 hours) at low speed to avoid cell lysis.
- Separate: Centrifuge the suspension (e.g., at 10,000 × g for 20 minutes at 4°C) to remove the CER and residual cells.
- Recover EPS: The resulting supernatant contains the extracted EPS. This supernatant can be filtered (e.g., 0.22 µm pore size) to ensure the removal of any remaining cells or debris.
- Storage: Store the purified EPS extract at -20°C until further analysis [4].
Biofilm Quantification and Viability Assays
- Colony-Forming Unit (CFU) Assay: This fundamental method quantifies viable bacterial cells within biofilms.
- Protocol: After growing biofilms on surfaces, dislodge the cells by sonication or vigorous vortexing. Serially dilute the suspension and spread onto appropriate agar plates. After incubation, count the colonies to calculate CFU per unit area (e.g., CFU/cm²) [6].
- Crystal Violet (CV) Assay: This assay quantifies the total biofilm biomass (cells and matrix).
- Protocol: Fix biofilms grown in microtiter plates with methanol or ethanol. Stain with a crystal violet solution (e.g., 0.1% w/v). Wash off unbound dye, solubilize the bound dye with acetic acid or ethanol, and measure the absorbance at 570-600 nm [6].
- MTT Assay: This colorimetric assay assesses the metabolic activity of cells within the biofilm.
- Protocol: Incubate biofilms with the MTT reagent (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide). Metabolically active cells reduce MTT to purple formazan crystals. Solubilize the crystals with DMSO or isopropanol and measure the absorbance at 570 nm [6].
Analytical Techniques for EPS Characterization
Fourier Transform Infrared (FTIR) Spectroscopy
FTIR spectroscopy is a powerful technique for identifying the chemical functional groups and overall composition of EPS.
- Principle: Organic molecules absorb infrared light at specific wavelengths, causing bond vibrations (stretching and bending). The resulting absorption spectrum provides a molecular fingerprint of the sample [3].
- Protocol for ATR/FT-IR: Place a dried EPS sample or a hydrated biofilm directly onto the crystal of an Attenuated Total Reflectance (ATR) accessory. Acquire the spectrum in the mid-IR range (e.g., 4000-400 cm⁻¹). The absorption bands can be assigned to specific EPS components [3]:
- Proteins: Amide I (~1650 cm⁻¹, C=O stretch), Amide II (~1550 cm⁻¹, N-H bend)
- Polysaccharides/Nucleic Acids: Region 900-1250 cm⁻¹ (C-O, C-O-C, P=O stretches)
- Lipids: Region 2800-3000 cm⁻¹ (C-H stretches)
- Data Analysis: Monitor changes in band intensity ratios (e.g., Amide II/Polysaccharide) to understand shifts in EPS composition during biofilm development [3].
Microscopy for Architectural Analysis
- Confocal Laser Scanning Microscopy (CLSM): Allows for non-invasive, three-dimensional visualization of hydrated biofilms. When used with fluorescent stains (e.g., concanavalin A for polysaccharides, SYTO dyes for nucleic acids), it reveals the spatial organization of EPS components and cells [6].
- Scanning Electron Microscopy (SEM): Provides high-resolution, topographical images of the biofilm surface. Requires sample dehydration and coating with a conductive material, which can introduce artifacts but reveals intricate structural details [6].
The Scientist's Toolkit: Key Research Reagent Solutions
The following table details essential reagents, materials, and instruments used in EPS and biofilm research, as cited in the referenced literature.
Table 3: Essential Research Reagents and Materials for EPS and Biofilm Studies
| Reagent / Material | Function / Application | Example from Literature |
|---|---|---|
| Cation Exchange Resin (CER) | Chemical extraction of EPS from biofilms by disrupting ionic bonds. | Amberlite HPR1100 for EPS extraction from bacterial and fungal cultures [4]. |
| Crystal Violet | Staining and quantification of total biofilm biomass. | Used in CV assay to measure biofilm formation of P. aeruginosa and E. coli on food contact surfaces [6]. |
| MTT Reagent | Assessment of metabolic activity and viability of cells within biofilms. | MTT assay used to evaluate biofilm health and resilience [6]. |
| Hydrolytic Enzymes | Targeted degradation of specific EPS components to study their functional role. | Serratiopeptidase (protease) and Alpha-amylase used to disrupt biofilms and potentiate antibiotic action [3]. |
| Quartz Matrix | Provides a solid surface to induce and study biofilm formation under controlled conditions. | Sterile quartz (SiO₂) used to study EPS production by soil bacteria and fungi [4]. |
| Fourier Transform Infrared (FTIR) Spectrometer | Chemical characterization and identification of functional groups in EPS. | ATR/FT-IR used to analyze the chemical content of biofilms, identifying proteins, polysaccharides, and nucleic acids [3]. |
| Confocal Laser Scanning Microscope (CLSM) | 3D visualization of biofilm architecture and spatial distribution of EPS components. | Used to analyze biofilm thickness and cell distribution of P. aeruginosa and E. coli [6]. |
Functional Relationships of EPS Components in Biofilm Physiology
The individual components of the EPS do not function in isolation but interact synergistically to confer critical properties to the biofilm. Understanding these relationships is key to developing control strategies.
The EPS matrix is a sophisticated biological system whose complexity is only beginning to be fully understood. The combination of robust extraction protocols, quantitative assays, and advanced analytical techniques, as detailed in this guide, provides a powerful framework for deconstructing this complexity. For researchers in microbiology and drug development, a thorough grasp of EPS composition and function is indispensable for innovating new ways to combat biofilm-associated infections or to manipulate biofilms for industrial and environmental benefit.
The extracellular polymeric substance (EPS) is a self-produced, hydrated matrix that encapsulates microbial cells in biofilms, serving as the primary architectural scaffold for these complex communities [7]. This matrix is far from an inert substance; it is a dynamic and functional component that determines the physicochemical properties of the biofilm and provides critical functions including structural integrity, protection from environmental stresses, and resistance to antimicrobial agents [8] [2]. The EPS matrix facilitates cell-cell communication, retains extracellular enzymes, and acts as a nutrient source [8]. For pathogenic bacteria, the EPS is a major virulence factor, contributing to chronic infections by shielding bacteria from host immune defenses and antibiotic treatments [9] [10]. The transition from free-floating planktonic cells to a sessile biofilm lifestyle is a fundamental survival strategy for bacteria, and this transition is intrinsically linked to the production of EPS components [10]. While the EPS is a composite mixture of polysaccharides, proteins, nucleic acids, and lipids, this review focuses on three key structural components that are nearly universal in bacterial biofilms: exopolysaccharides, functional amyloids, and extracellular DNA (eDNA). Understanding the structure, function, and regulation of these core components is pivotal for developing novel anti-biofilm strategies to combat the global health threat of antibiotic-resistant, biofilm-associated infections.
Exopolysaccharides: The Structural Backbone
Definition, Chemical Nature, and Prevalence
Exopolysaccharides are high-molecular-weight sugar-based polymers secreted by microorganisms into their environment [2]. They are a major fraction of the biofilm EPS in both Gram-positive and Gram-negative bacteria [9]. These polymers can be homopolysaccharides (composed of a single type of monosaccharide) or, more commonly, heteropolysaccharides consisting of a mixture of neutral and charged sugar residues [9]. Many known exopolysaccharides, such as alginate, are polyanionic due to the presence of uronic acids or ketal-linked pyruvates, although polycationic exopolysaccharides like Pel also exist [9]. The composition and quantity of exopolysaccharides can vary significantly depending on the microbial species, the age of the biofilm, and environmental conditions such as nutrient availability, temperature, and pH [7]. Exopolysaccharides can constitute between 50% to 90% of a biofilm's total organic matter, establishing them as a fundamental component determining the biofilm's physicochemical properties [2].
Key Functions in Biofilm Development and Architecture
Exopolysaccharides are involved in multiple critical stages of biofilm formation and maintenance. Their functions extend beyond mere structural support and include:
- Surface Attachment and Colonization: They mediate the initial reversible and irreversible attachment of planktonic cells to both biotic and abiotic surfaces, enabling colonization [9] [7].
- Intercellular Adhesion and Aggregation: Exopolysaccharides act as a glue, promoting cell-to-cell adhesion and the formation of microcolonies, which are the building blocks of the mature biofilm architecture [9] [11].
- Mechanical Stability and Hydration: They form a hydrated polymer network that provides mechanical stability to the biofilm and prevents bacterial desiccation [9]. The gel-forming capability of exopolysaccharides, as seen with Bacillus subtilis EpsA-O, allows the biofilm to span intercellular space and form complex 3D structures that can resist physical forces like shear stress [11].
- Protective Barrier: The exopolysaccharide matrix acts as a barrier that inhibits the penetration of antibiotics, antiseptics, and host defense molecules, thereby conferring significant resistance to the enclosed bacterial community [9] [7].
- Nutrient Source: Exopolysaccharides can serve as a carbon and energy source for the biofilm community [9].
Table 1: Major Biofilm Exopolysaccharides, Their Producers, and Structural Features
| Exopolysaccharide | Producing Organism(s) | Chemical Composition / Key Features |
|---|---|---|
| PNAG/PIA | Staphylococcus aureus, S. epidermidis, E. coli, Acinetobacter baumannii | β(1,6)-linked N-acetylglucosamine; partially deacetylated (5-40%); cationic character [9] |
| Alginate | Pseudomonas aeruginosa, Azotobacter vinelandii | Polyanionic; polymer of D-mannuronic acid and L-guluronic acid [9] [2] |
| Psl | Pseudomonas aeruginosa (strain PAO1) | Pentasaccharide repeating unit; neutral; mannose, rhamnose, glucose [9] [10] |
| Pel | Pseudomonas aeruginosa (strain PA14) | Cationic; partially deacetylated; N-acetylgalactosamine, N-acetylglucosamine [9] [10] |
| Cellulose | Acetobacter xylinum, E. coli | Unbranched β-1,4-linked glucan; provides strength and rigidity [2] [10] |
| EpsA-O | Bacillus subtilis | Branched trisaccharide backbone with pyruvate side chains; forms gels; molecular mass ~2.5 MDa [11] |
Regulatory Mechanisms: The Role of c-di-GMP
The biosynthesis of biofilm exopolysaccharides is tightly regulated, with the second messenger cyclic di-guanosine monophosphate (c-di-GMP) playing a central role. Intracellular c-di-GMP levels are controlled by the opposing activities of diguanylate cyclases (DGCs), which synthesize it, and phosphodiesterases (PDEs), which degrade it [10]. High intracellular concentrations of c-di-GMP promote the transition from a planktonic to a sessile biofilm lifestyle by post-translationally activating the biosynthetic machineries for exopolysaccharides like cellulose, alginate, Pel, and PNAG [10]. This activation triggers the secretion of EPS components and leads to the maturation of complex biofilm structures. Conversely, a decrease in c-di-GMP leads to biofilm dispersal through the production of surfactant molecules and the reactivation of cell motility [10].
Diagram 1: Regulation of biofilm formation and dispersal by the second messenger c-di-GMP. High intracellular c-di-GMP, synthesized by DGCs, promotes exopolysaccharide production and biofilm formation. PDEs break down c-di-GMP, leading to low intracellular levels and biofilm dispersal [10].
Functional Amyloids: The Proteinaceous Framework
Distinction from Pathological Amyloids
Functional bacterial amyloids (FuBAs) are protein structures that self-assemble into fibrils with a characteristic cross-β-sheet structure, serving a defined biological purpose for the organism, in contrast to the disease-associated amyloids like those in Alzheimer's disease [12]. While they share the amyloid fold with pathological amyloids, FuBAs are distinct in that their formation is a highly controlled process, optimized for efficient and rapid extracellular self-assembly without forming toxic oligomers inside the cell [12]. These fibrils are a major threat to human health as they strengthen the biofilm's structural integrity, promote antibiotic resistance (AMR), and protect against host immune system attacks [12].
Major Functional Amyloid Systems in Biofilms
The most extensively characterized functional amyloids in bacterial biofilms are curli in E. coli and Salmonella, and Fap in Pseudomonas.
Curli in E. coli: Curli fibrils are essential for surface attachment and biofilm formation. Their biogenesis is a highly regulated process involving the csgBAC and csgDEFG operons [12]. The major subunit, CsgA, is secreted as an unstructured monomer through the outer membrane pore protein CsgG. On the cell surface, the nucleator protein CsgB facilitates the polymerization of CsgA into mature amyloid fibrils. The chaperone-like proteins CsgC and CsgE prevent premature fibrillation inside the cell, ensuring CsgA and CsgB are secreted as monomers [12]. CsgA contains five imperfect repeats rich in glutamine and asparagine, which are crucial for its amyloidogenicity and its ability to interact with host proteins like fibronectin and plasminogen [12].
Fap in Pseudomonas: The Functional amyloid in Pseudomonas (Fap) system is encoded by the fapA-F operon. FapC is the major amyloid-forming protein, while FapB acts as a nucleator. Similar to curli, FapC is secreted as an unfolded monomer and fibril formation occurs at the cell surface [12]. FapC contains three imperfect repeat motifs of approximately 37 amino acids that stack into the β-sheet structure of the amyloids. These repeats, which also contain conserved glutamine and asparagine residues, are critical for the stability and efficient aggregation of Fap fibrils [12]. The Fap system is found in many Pseudomonas strains, including the clinically relevant P. aeruginosa, where it is considered a virulence-enhancing factor in chronic infections like those in cystic fibrosis patients [12].
Table 2: Characteristics of Major Bacterial Functional Amyloids
| Feature | Curli (E. coli) | Fap (Pseudomonas) |
|---|---|---|
| Major Subunit | CsgA | FapC |
| Nucleator | CsgB | FapB |
| Operon | csgBAC, csgDEFG | fapA-F |
| Membrane Transporter | CsgG (outer membrane pore) | FapF (trimeric β-barrel) |
| Periplasmic Chaperone | CsgC, CsgE | Information Not Specified |
| Repeat Motifs | Five imperfect ~20 amino acid repeats | Three imperfect ~37 amino acid repeats |
| Key Residues | Glutamine, Asparagine | Glutamine, Asparagine |
Extracellular DNA (eDNA): The Versatile Matrix Component
Origin and Structural Characteristics
Extracellular DNA (eDNA) is a ubiquitous nucleic acid biopolymer critical for the integrity of the biofilm matrix [13]. While initially considered a mere remnant of lysed cells, eDNA is now recognized as an essential structural component [13] [8]. eDNA often originates from the genomic DNA (gDNA) of bacterial cells, released through explosive cell lysis, which in some species like Staphylococcus aureus is under genetic control [13] [8]. However, eDNA and chromosomal gDNA are not necessarily structurally or compositionally identical [13]. In P. aeruginosa, eDNA is organized in distinct grid-like structures and appears to be derived from whole genomic DNA [8]. In other species, eDNA can be fragmented, which may enhance its interaction with matrix proteins and promote more abundant biofilm formation [13]. A groundbreaking discovery is that eDNA in the biofilm matrix can adopt the rare Z-form conformation, which is stabilized by bacterial DNABII proteins and confers exceptional structural integrity and resistance to degradation by DNases [14].
Critical Functions in the Biofilm Matrix
eDNA performs multiple vital functions that are crucial for different stages of the biofilm lifecycle:
- Structural Integrity and Stability: eDNA stabilizes the biofilm matrix by interacting with other EPS components, such as exopolysaccharides and proteins, through electrostatic and other physiochemical interactions [13]. It functions as an intercellular connector, providing structural rigidity and contributing to the mechanical stability of the biofilm [13] [8].
- Cell Adhesion and Aggregation: During the initial stages of biofilm formation, eDNA facilitates the irreversible attachment of cells to surfaces and promotes cell-to-cell adhesion, helping to form the initial aggregate structure [13].
- Genetic Information and Horizontal Gene Transfer: As a repository of genetic information, eDNA facilitates horizontal gene transfer between cells in close proximity within the biofilm, promoting the spread of antibiotic resistance genes [13] [8].
- Protection and Immune Evasion: The eDNA meshwork, particularly in its Z-form, acts as a physical barrier that hinders the penetration of antimicrobial agents and protects the bacterial community from host defense responses [13] [14]. It has also been shown that bacterial DNABII proteins can stabilize both bacterial and host-derived eDNA in the Z-form, which may play a role in immune evasion [14].
Experimental Approaches for Component Analysis
Key Methodologies and Workflows
Research into the structural components of biofilms relies on a multidisciplinary toolkit that combines biochemical, genetic, biophysical, and microscopy-based techniques. Below are detailed protocols for key experimental approaches cited in this field.
Protocol 1: Isolation and Chemical Analysis of Exopolysaccharides (e.g., EpsA-O from B. subtilis) [11]
- Biofilm Cultivation and EPS Extraction: Grow B. subtilis in optimized liquid medium (e.g., MSgg) to promote biofilm formation. Harvest the biofilm biomass (e.g., pellicle from air-liquid interface) and separate cells from the EPS via centrifugation. Precipitate the crude EPS from the supernatant using cold ethanol.
- Purification: Re-dissolve the crude EPS precipitate and subject it to High-Performance Liquid Chromatography with Size-Exclusion Chromatography (HPLC-SEC). Collect the high-molecular-weight fraction corresponding to the exopolysaccharide (e.g., EpsA-O eluting at ~18 minutes). Confirm purity by assessing protein and nucleic acid contamination (e.g., absorbance at 260/280 nm, specific assays).
- Compositional Analysis:
- Sugar Composition: Perform acid hydrolysis of the purified EPS to break it down into monosaccharides. Derivatize the released sugars to alditol acetates or trimethylsilylethers and analyze via Gas Chromatography-Mass Spectrometry (GC-MS) to identify and quantify neutral sugars (e.g., Galactose, N-acetylglucosamine, N-acetylgalactosamine).
- Glycosidic Linkage Analysis: Use methylation analysis followed by GC-MS to determine the positions of glycosidic linkages between monosaccharides in the polymer.
- Structural Elucidation: Dissolve the purified EPS in D₂O. Acquire ¹H, ¹³C, and two-dimensional (e.g., COSY, TOCSY, HSQC, HMBC) Nuclear Magnetic Resonance (NMR) spectra. Analyze the chemical shifts, coupling constants, and through-bond correlations to determine the precise structure of the repeating unit, including anomeric configuration and linkage positions.
- Physicochemical Characterization:
- Molecular Mass Determination: Use the HPLC-SEC system with a multi-angle light scattering (MALS) detector or universal calibration with standards to estimate the average molecular mass.
- Rheological Analysis: Perform viscosity measurements of EPS solutions at different concentrations to determine the critical overlap concentration (c*) and intrinsic viscosity ([η]). Conduct oscillatory shear experiments to measure storage (G') and loss (G") moduli, identifying the gel point and characterizing viscoelastic properties.
Protocol 2: Demonstrating the Structural Role of eDNA via DNase Treatment [13]
- Biofilm Growth: Grow biofilms in vitro using appropriate media and surfaces (e.g., microtiter plates, flow cells) for a defined period to allow mature biofilm development.
- Treatment with DNase I: Prepare a solution of DNase I enzyme in a suitable buffer containing Mg²⁺ and Ca²⁺ (cofactors for enzyme activity). Gently apply the DNase I solution to the mature biofilms. Include a control group treated with buffer only or with heat-inactivated enzyme.
- Incubation and Disruption: Incubate the biofilms with DNase I for a predetermined time (e.g., 1-2 hours) at 37°C. The enzyme will hydrolyze the phosphodiester bonds in eDNA.
- Assessment of Biofilm Disruption:
- Quantitative Assessment: Use crystal violet staining to quantify the total remaining biofilm biomass after DNase treatment and washing. A significant reduction in staining compared to the control indicates biofilm disruption.
- Qualitative/Microscopic Assessment: For biofilms grown in flow cells or on coverslips, use staining with DNA-binding fluorescent dyes (e.g., SYTO dyes, TOTO-1) and analyze via Confocal Laser Scanning Microscopy (CLSM) before and after treatment. Observe the dissolution of the eDNA grid-like structures and overall changes in biofilm architecture.
- Evaluation of Enhanced Antibiotic Efficacy: Co-treat biofilms with a combination of DNase I and a relevant antibiotic. Compare the reduction in viable cell counts (via colony forming unit assays) to treatments with DNase I or antibiotic alone. Enhanced killing with the combination treatment demonstrates that eDNA disruption improves antibiotic penetration.
Diagram 2: Experimental workflow for assessing the structural role of eDNA in biofilms using DNase I treatment. Hydrolysis of eDNA leads to biofilm disruption, which can be quantified and visualized, and subsequently correlated with increased antibiotic susceptibility [13].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Biofilm Structural Analysis
| Reagent / Material | Function / Application in Research |
|---|---|
| DNase I | An endonuclease that cleaves DNA. Used to disrupt biofilms by degrading the eDNA scaffold, thereby demonstrating eDNA's structural role and increasing antibiotic penetration [13]. |
| Fluorescently Labeled Lectins | Lectins are sugar-binding proteins. When fluorescently tagged, they are used for in situ labeling and visualization of specific glycoconjugates and exopolysaccharides within the biofilm matrix without the need for purification [8]. |
| Anti-PNAG Monoclonal Antibody (e.g., mAb F598) | A specific antibody used for immunochemical detection, localization, and quantification of the poly-N-acetylglucosamine (PNAG) exopolysaccharide on a wide range of bacterial and fungal pathogens [9]. |
| Dispersin B | A glycoside hydrolase enzyme that specifically degrades PNAG. Used to disrupt PNAG-dependent biofilms and to study the functional role of this exopolysaccharide [9]. |
| DNABII Protein Antibodies | Antibodies targeting bacterial DNABII proteins (e.g., integration host factor IHF). Used to disrupt the biofilm matrix by displacing these proteins, which destablizes the protective Z-form eDNA structure [14]. |
| SYTO Dyes / TOTO-1 | Cell-permeable (SYTO) and cell-impermeant (TOTO-1) fluorescent nucleic acid stains. Used to label and visualize the spatial distribution and organization of eDNA within the biofilm matrix using techniques like Confocal Laser Scanning Microscopy (CLSM) [13] [8]. |
The biofilm matrix is a sophisticated biological construct where exopolysaccharides, functional amyloids, and extracellular DNA act in concert to create a resilient and protective environment for microbial communities. Exopolysaccharides provide the foundational scaffold and mediate initial surface interactions, functional amyloids contribute robust proteinaceous fibrils that enhance structural integrity, and eDNA acts as a versatile polyelectrolyte that cross-links the matrix and provides mechanical stability, often in a nuclease-resistant Z-form. The production of these components is not haphazard but is precisely regulated by signaling molecules like c-di-GMP. The synergistic interactions between these three core components are what ultimately confer upon biofilms their formidable resistance to mechanical and chemical stresses. Disrupting the synthesis, assembly, or interactions of exopolysaccharides, functional amyloids, and eDNA represents a promising frontier for developing novel anti-biofilm therapies. Such strategies, including enzymatic degradation of matrix components (e.g., Dispersin B, DNase) or interference with regulatory pathways (e.g., c-di-GMP signaling), hold the potential to sensitize persistent biofilm-associated infections to conventional antibiotics and immune clearance, thereby addressing a critical need in modern healthcare.
Extracellular Polymeric Substances (EPS) form the foundational architecture of microbial biofilms, serving as a dynamic scaffold that determines the community's physical integrity, resilience, and function [2]. This self-produced matrix, constituting 50% to 90% of a biofilm's total organic matter, is a complex composite of polysaccharides, proteins, extracellular DNA (eDNA), lipids, and other macromolecules [2] [15]. The biofilm lifecycle is a meticulously orchestrated process, from initial attachment to eventual dispersion, and each stage is critically mediated by the changing composition and role of the EPS [16]. For researchers and drug development professionals, understanding the functional dynamics of the EPS throughout this lifecycle is paramount to developing effective strategies to combat biofilm-associated infections, which account for 65-80% of all human microbial infections [16]. This guide provides an in-depth technical analysis of the EPS's role, supported by current experimental data and methodologies.
Stage 1: Attachment and The Foundation of the EPS Matrix
The biofilm lifecycle initiates with the attachment of planktonic cells to a surface. This transition is not passive but is actively facilitated by early EPS components that overcome repulsive forces and enable irreversible adhesion.
- Mechanisms of Attachment: The initial, reversible attachment is influenced by substratum properties, hydrodynamic conditions, and cell surface characteristics like hydrophobicity, fimbriae, and flagella [15]. The subsequent transition to irreversible attachment is cemented by the production of early EPS components. eDNA, for instance, acts as a critical initial adhesin in many species, while type IV pili and other surface proteins facilitate attachment and twitching motility [17].
- Environmental Cues: Surface sensing triggers profound changes in gene expression, leading to the upregulation of EPS production machinery. Studies show that the presence of a mineral surface, such as quartz, can significantly enhance EPS production, underscoring the importance of environmental context [18].
The following diagram illustrates the signaling and structural pathways that drive the initial stages of biofilm formation.
Stage 2: Maturation and The EPS as a Structured Ecosystem
Following attachment, the biofilm enters a maturation phase where it develops a complex, three-dimensional architecture. The EPS is not a homogeneous slurry but a highly organized and functional ecosystem.
- Architectural Complexity: The EPS matrix forms a scaffold for the development of microcolonies that evolve into towering structures interspersed with fluid channels [16]. These channels facilitate the distribution of nutrients and oxygen, creating diverse microniches within the biofilm [16].
- Component Specialization: During maturation, the specific composition of the EPS is strongly influenced by the microbial species present, though environmental factors can shift the relative abundance of constituents [18]. Each component plays a specialized role:
- Exopolysaccharides (e.g., Psl, Pel, alginate, dPNAG): Provide structural integrity, mediate cell-to-cell adhesion, and contribute to antigenic variation [17] [16].
- Proteins (e.g., lectins, amyloids, DNABII): Act as adhesins, cross-link polysaccharide fibers, and stabilize the matrix. The DNABII family proteins, in particular, bind to eDNA and serve as a critical structural scaffold [17].
- Extracellular DNA (eDNA): Functions as a structural glue, promotes horizontal gene transfer, and can chelate cations or antimicrobials [17] [19].
Recent quantitative research has shed light on how environmental factors influence EPS composition. The table below summarizes key findings from a 2025 study analyzing EPS constituents from ten bacterial and fungal species under varying conditions [18].
Table 1: Influence of Substrate and Surface on EPS Constituent Concentration (μg/mL) [18]
| EPS Constituent | Glycerol Medium | Starch Medium | Glycerol + Quartz | Starch + Quartz | Primary Function in Mature Biofilm |
|---|---|---|---|---|---|
| Carbohydrates | 12.5 | 25.8 | 18.8 | 38.9 | Structural scaffolding, nutrient source [18] |
| Proteins | 15.3 | 18.1 | 16.2 | 19.5 | Enzymatic activity, structural adhesion [18] |
| DNA | 2.1 | 2.5 | 2.3 | 2.7 | Structural integrity, gene transfer [17] |
| Glucosamine | 4.2 | 5.1 | 4.8 | 5.6 | Microbial residue marker, structural component [18] |
| Mannosamine | 1.1 | 1.3 | 1.2 | 1.4 | Specific EPS marker, function under investigation [18] |
| Galactosamine | 0.9 | 1.1 | 1.0 | 1.2 | Specific EPS marker, function under investigation [18] |
| Muramic Acid | 0.5 | 0.6 | 0.6 | 0.7 | Indicator of bacterial necromass [18] |
| Carbohydrate/Protein Ratio | 0.82 | 1.42 | 1.16 | 1.99 | Indicator of matrix structural change [18] |
The data demonstrates that a more labile carbon source (starch) and the presence of a surface (quartz) significantly boost EPS production, particularly carbohydrates, thereby altering the matrix's physical properties [18].
Stage 3: Dispersion and The Enzymatic Breakdown of EPS
Dispersion, the final stage of the lifecycle, is an active process where portions of the biofilm are released to colonize new niches. This is often a regulated response to nutrient depletion or other environmental stresses and is primarily mediated by the enzymatic degradation of the EPS matrix [16].
- Active vs. Passive Dispersion: Active dispersion is a tightly regulated process often triggered by a decrease in intracellular cyclic di-GMP (c-di-GMP) levels, leading to the production of specific EPS-degrading enzymes [20]. Passive dispersion, in contrast, can result from physical shear forces or the action of exogenous agents [20].
- Key Enzymatic Targets: Research into biofilm eradication strategies has focused on exploiting these natural dispersion mechanisms by applying enzymes exogenously. The three primary enzyme classes under investigation are summarized below.
Table 2: Key Enzymatic Targets for Inducing Biofilm Dispersion
| Enzyme Class | Target in EPS | Mechanism of Action | Specific Examples & Targeted Polymers |
|---|---|---|---|
| Glycoside Hydrolases | Exopolysaccharides | Hydrolyze glycosidic bonds in polysaccharide chains, dismantling the primary structural network [16]. | Dispersin B (targets dPNAG/PIA) [16]; Alginate lyase (targets alginate) [16] |
| Proteases | Extracellular Proteins | Degrade proteinaceous components of the matrix, including adhesins and structural proteins [16]. | Serine proteases (e.g., Esp) [16]; Metalloproteases [16] |
| Deoxyribonucleases (DNases) | Extracellular DNA (eDNA) | Cleave eDNA strands, disrupting a key structural component, especially in early-stage biofilms [17] [16]. | Recombinant human DNase I (Dornase alfa) [17] |
The strategic use of these enzymes can convert resistant, sessile communities into vulnerable planktonic cells, thereby increasing their susceptibility to conventional antibiotics and host immune responses [16].
Experimental Toolkit for EPS and Biofilm Analysis
A multidisciplinary approach is essential for characterizing the complex nature of biofilms and their EPS. The following section details key experimental protocols and reagents used in the field.
Detailed Protocol: EPS Extraction and Constituent Analysis
This protocol is adapted from a 2025 study that investigated EPS composition across multiple microbial species [18].
Microbial Growth and Biofilm Cultivation:
- Inoculum: Ten soil bacterial and ten fungal species are cultured in 500 ml shake flasks.
- Culture Conditions: Cells are grown in either glycerol or starch media, with or without a sterile quartz matrix (0.4-0.8 mm, 140 g per flask). The quartz physically forces microbial growth within its matrix.
- Incubation: Flasks are incubated at 30°C with shaking at 100 rpm for 4 days.
EPS Extraction via Cation Exchange Resin (CER):
- Collection: 10 ml aliquots are collected from the cultures using a graduated cylinder.
- CER Addition: A predetermined amount of cation exchange resin (e.g., Amberlite HPR1100) is added to the aliquot, as established for Pseudomonas putida [18] [16].
- Extraction: The sample is mixed with the CER for a specified period to disrupt ionic interactions and release the EPS from the cells.
- Separation and Storage: The EPS-containing supernatant is separated from the cells and CER by centrifugation or filtration and stored at -20°C until analysis.
Analysis of EPS Constituents:
- Total Carbohydrates: Determined using an acid hydrolysis method. An EPS aliquot is hydrolyzed with 0.75 M H₂SO₄ at 100°C, diluted with PBS, and quantified with a bicinchoninic acid (BCA) microplate assay, measuring absorbance at 562 nm [18] [21].
- Total Proteins: Estimated using the Lowry assay microplate method. EPS extracts are incubated with a copper sulphate solution containing the Folin-Ciocalteu reagent, and absorbance is measured at 750 nm [18] [15].
- Amino Sugars (Muramic Acid, Glucosamine, etc.): Analyzed using chromatographic techniques (e.g., GC-MS or HPLC) after appropriate derivatization to quantify these key markers of microbial residues and specific EPS components [18].
- Extracellular DNA: Purified from EPS extracts and quantified using fluorescent dyes (e.g., PicoGreen) or other standard DNA quantification methods [18].
The Scientist's Toolkit: Essential Research Reagents
The table below lists key reagents and their functions for standard biofilm analysis, as cited in the search results.
Table 3: Essential Reagents for Biofilm Research
| Reagent / Material | Function in Biofilm Research | Example Use Case |
|---|---|---|
| Cation Exchange Resin (CER) | Extracts EPS by binding cations and disrupting ionic bonds in the matrix [18]. | EPS extraction protocol for compositional analysis [18]. |
| Crystal Violet (CV) | Stains total biofilm biomass (cells and EPS); used for high-throughput quantification [6] [22]. | Microtiter plate (96-well) biofilm assays [22]. |
| Maneval's Stain | A low-cost, dual-staining method that differentially stains cells (magenta-red) and the polysaccharide matrix (blue) for light microscopy [21]. | Visualization and differentiation of biofilm components on a glass slide [21]. |
| Calcofluor White | A fluorescent stain that binds to polysaccharides like cellulose and β-glucans in the EPS [21]. | Fluorescence microscopy visualization of the EPS matrix [21]. |
| Dispersin B | A glycoside hydrolase enzyme that specifically degrades the dPNAG exopolysaccharide [16]. | Used as a treatment to induce dispersion in dPNAG-dependent biofilms [16]. |
| Deoxyribonuclease I (DNase I) | An enzyme that degrades extracellular DNA (eDNA), a critical structural component in many biofilms [17] [16]. | Testing biofilm stability and as an adjuvant therapy to disrupt biofilms [17]. |
Visualization and Quantification Techniques
A combination of techniques is required to fully understand biofilm architecture and composition.
- Advanced Microscopy:
- Confocal Laser Scanning Microscopy (CLSM): Allows for non-invasive optical sectioning to create 3D reconstructions of live biofilms, often using fluorescent stains [6].
- Scanning Electron Microscopy (SEM): Provides high-resolution, topographical images of the biofilm surface. Requires extensive sample preparation including fixation, dehydration, and critical-point drying [6] [21].
- Spectroscopic Analysis:
- Fourier Transform Infrared (FTIR) Spectroscopy: Identifies functional groups and chemical bonds within the EPS, such as carboxyl and hydroxyl groups, providing information on the chemical nature of the matrix [6].
- Quantitative Assays:
The logical workflow for a comprehensive biofilm study, integrating these techniques, is outlined below.
The biofilm lifecycle is a dynamic continuum powered by the ever-changing role of the EPS. From a facilitator of attachment to a complex 3D ecosystem, and finally, a target for enzymatic dispersion, the EPS matrix is the key determinant of biofilm survival and resilience. The quantitative data and advanced methodologies presented here provide a roadmap for researchers to dissect the composition, architecture, and vulnerabilities of biofilms. As our understanding of EPS function deepens, so too will our ability to design targeted anti-biofilm strategies, such as novel combination therapies using dispersion enzymes and antibiotics, to address the significant challenge posed by chronic biofilm-associated infections.
In the realm of biofilm research, the extracellular polymeric substance (EPS) is not merely a passive component but the primary architect of the biofilm's structural integrity and function. This complex, self-produced hydrogel forms a protective matrix that encases microbial cells, enabling them to adhere to surfaces, cohere into communities, and withstand formidable environmental challenges. The mechanical properties of biofilms—their cohesiveness, resilience, and adhesion—are direct consequences of the EPS's physical and chemical nature. Composed of a dynamic mixture of polysaccharides, proteins, lipids, and extracellular DNA (eDNA), the EPS matrix operates as a multifunctional biopolymer that determines the physical robustness of microbial life in aggregated states. Understanding the specific mechanisms through which EPS confers these mechanical properties is crucial for advancing fundamental microbial ecology and developing strategies to combat biofilm-associated infections or harness their beneficial applications.
Fundamental Concepts: Composition Dictates Function
The mechanical functionality of EPS is an emergent property of its specific composition and the interactions between its constituent parts. The following components are particularly critical:
- Polysaccharides often form the structural backbone of the matrix, providing bulk and mediating cohesion through extensive chain entanglement and hydrogen bonding.
- Proteins contribute to adhesion through specific surface-binding domains and can enhance cohesion via cross-linking.
- Extracellular DNA (eDNA) has been increasingly recognized as a crucial structural element, forming a scaffold that confers viscoelasticity and stress-hardening capabilities—the ability to stiffen in response to applied stress [23].
- Lipids and other surfactants can modify surface tension and interfacial properties, influencing initial attachment.
The synergistic interactions between these components create a composite material whose mechanical properties are greater than the sum of its parts. For instance, eDNA can interact with polysaccharides and proteins to form a reinforced network [23].
Mechanisms of Action: How EPS Mediates Key Mechanical Properties
Cohesiveness: The Internal "Glue"
Cohesiveness refers to the strength of internal attachment within the biofilm, allowing it to resist disintegration and maintain its three-dimensional structure. This property is primarily enabled by:
- Chain Entanglement and Cross-linking: Long, polymeric EPS chains, particularly polysaccharides and eDNA, become physically entangled, creating a physical barrier to separation. Furthermore, chemical cross-links (e.g., ionic bonds mediated by divalent cations like Ca²⁺) between anionic functional groups on the polymers dramatically increase the structural integrity and cohesion of the matrix [1] [24].
- The Critical Role of eDNA: Recent studies on biofilm streamers reveal that eDNA forms the structural backbone that resists tensile forces. This network exhibits stress-hardening behavior, where its differential elastic modulus increases linearly with the external stress applied to it. This means the more the biofilm is stretched, the stiffer and more cohesive it becomes, a property directly inherited from the physical behavior of DNA molecules themselves [23].
Adhesion: The Surface "Anchor"
Adhesion is the ability of the biofilm to attach firmly to a biotic or abiotic surface. This is the foundational step of biofilm formation and is governed by:
- Initial Reversible Attachment: Pioneer cells use weak, long-range physico-chemical forces (e.g., van der Waals forces, electrostatic interactions) to initially adhere to a conditioned surface [1] [25].
- Irreversible Attachment and Anchoring: The subsequent production of EPS transitions the attachment to a permanent state. Adhesive EPS proteins and polysaccharides form specific and non-specific bonds with the substrate surface. The interplay between friction (resisting lateral expansion) and adhesion (resisting vertical detachment) is critical for subsequent biofilm development. Computational models show that strong adhesion can suppress morphological instabilities like wrinkling by resisting delamination from the substrate [25].
Resilience: The Capacity to Recover from Stress
Resilience is the biofilm's ability to adapt to and recover from mechanical and chemical challenges, such as fluid shear stress or antibiotic treatment.
- Viscoelasticity: The EPS matrix is not purely elastic but viscoelastic, meaning it exhibits both solid-like and liquid-like behaviors. This allows it to dissipate energy through viscous flow when stressed, rather than fracturing like a brittle solid [1] [23].
- Adaptive Stress Response: The stress-hardening behavior governed by eDNA and modulated by extracellular RNA (eRNA) is a key rapid-response mechanism for resilience. This purely physical mechanism allows biofilm streamers to instantaneously adjust their stiffness and viscosity to varying hydrodynamic stresses in their environment, ensuring structural integrity under fluctuating conditions [23].
- Nutrient-Linked Morphological Adaptation: Biofilms can modulate their morphology in response to environmental cues. Under low nutrient conditions, growth is halted in nutrient-deprived regions, altering internal stress distributions and changing wrinkling patterns from the center to the edge. This redistribution is a morphological adaptation that enhances access to nutrients, contributing to long-term survival [25].
Table 1: Key EPS Components and Their Mechanical Roles
| EPS Component | Primary Role in Cohesiveness | Primary Role in Adhesion | Primary Role in Resilience |
|---|---|---|---|
| Polysaccharides | Forms a hydrated gel; chain entanglement provides bulk and resists compression. | Mediates non-specific attachment to surfaces; retains water to prevent desiccation. | Creates a diffusion barrier that slows penetrance of antimicrobials. |
| Proteins | Cross-linking of proteins and other polymers enhances structural integrity. | Often contains specific adhesins that bind to surface receptors, enabling firm attachment. | Can act as degradative enzymes to modify the matrix in response to stress. |
| eDNA/eRNA | Forms a structural backbone; charge-based interactions with other polymers; key for stress-hardening [23]. | May facilitate initial attachment through electrostatic interactions. | Confers viscoelasticity and stress-hardening; eRNA stabilizes eDNA structures [23]. |
| Lipids | Can modify the hydrophobicity of the matrix, influencing cell-cell interaction. | Impacts initial attachment by altering cell surface hydrophobicity. | May contribute to barrier formation against hydrophobic antimicrobials. |
Quantitative Analysis of EPS Mechanical Properties
The mechanical properties of EPS and biofilms are quantified using rheological and mechanical tests. The following table summarizes key findings from recent research:
Table 2: Quantitative Data on EPS and Biofilm Mechanical Properties
| Study System / Organism | Property Measured | Experimental Method | Key Quantitative Finding | Citation |
|---|---|---|---|---|
| P. aeruginosa biofilm streamers | Differential Young's Modulus (Ediff) | Microfluidic extensional rheology | Ediff increases linearly with pre-stress (σ0), demonstrating clear stress-hardening. The relationship is consistent across wild-type, Pel-deficient, and Pel-overproducer strains [23]. | [23] |
| P. aeruginosa biofilm streamers | Effective Viscosity (η) | Microfluidic extensional rheology | η increases linearly with pre-stress (σ0), indicating the matrix becomes more resistant to flow under stress [23]. | [23] |
| Lattice-Network Biofilm Model | Critical Buckling Stress (σc) | Computational modeling (Langevin dynamics) | σc is highest at the biofilm center under uniform nutrient supply. Stronger adhesion raises σc and delays wrinkle onset [25]. | [25] |
| Rhizosphere Microbial Fuel Cells | Biofilm Thickness & Performance | Scanning Electron Microscopy (SEM), power density output | Optimal EPS level (64 mg·g⁻¹) maximized power density (129 ± 3 mW·m⁻²). Excessive EPS (>64 mg·g⁻¹) increased thickness to ~0.48 mm, reducing activity and performance [26]. | [26] |
| Multi-species soil biofilms | EPS Constituent Ratios | Chemical extraction & spectrophotometry | EPS carbohydrate/protein ratio was higher with a less labile carbon source (starch) and increased in the presence of a quartz matrix, indicating environmental modulation of composition [4]. | [4] |
Experimental Protocols for Analyzing EPS Mechanics
Protocol 1: EPS Extraction using Cation Exchange Resin (CER)
This method is widely used for extracting the EPS matrix without causing significant cell lysis [4].
- Culture Biofilms: Grow microbial cultures or biofilms under desired conditions. For soil bacteria/fungi, cultures can be grown in shake flasks with or without a quartz matrix for 4 days [4].
- Harvest and Aliquot: Collect aliquots of the cell culture (e.g., 10 ml) using a graduated cylinder.
- CER Addition: Add a predetermined amount of cation exchange resin (e.g., Amberlite HPR1100) to the aliquot. The optimal amount must be determined empirically for different systems (e.g., based on protocols for Pseudomonas putida) [4].
- Extraction: Stir the mixture slowly for a specified period (e.g., 1-2 hours) at low temperature (e.g., 4°C) to allow the CER to displace cations binding the EPS, releasing it into the solution.
- Separation: Centrifuge the mixture to pellet the CER and cells. The resulting supernatant contains the extracted EPS.
- Storage: Store the EPS extract at -20°C until further analysis [4].
Protocol 2: In-situ Viscoelasticity Measurement of Biofilm Streamers
This protocol characterizes the stress-hardening behavior of biofilm streamers in a microfluidic setup [23].
- Microfluidic Setup: Use a microfluidic channel with pillar-shaped obstacles to nucleate and grow reproducible biofilm streamers from a bacterial suspension (e.g., P. aeruginosa) under continuous flow.
- Fluorescence Staining and Imaging: Once streamers reach a steady state, stain them with a nucleic acid dye like propidium iodide (which binds eDNA) and image using epifluorescence microscopy to reconstruct their 3D geometry.
- Computational Fluid Dynamics (CFD): Use the 3D geometry to perform CFD simulations. This calculates the axial stress (σ) at any point along the streamer based on the flow velocity and streamer morphology.
- Extensional Rheological Measurements: Apply a controlled flow perturbation to impose a known increment of extensional stress (Δσ) on top of the pre-stress (σ₀).
- Data Analysis: Measure the resulting strain increment (Δε). Calculate the differential Young's modulus as Ediff = Δσ / Δε and the effective viscosity. Plot these parameters against the pre-stress σ₀ to quantify the stress-hardening response [23].
The following diagram illustrates the experimental workflow for analyzing biofilm streamer mechanics:
The Scientist's Toolkit: Key Research Reagents and Solutions
Table 3: Essential Reagents and Materials for EPS and Biofilm Mechanics Research
| Reagent / Material | Function / Application | Example Use Case |
|---|---|---|
| Cation Exchange Resin (CER) | Extracts EPS by displacing divalent cations that cross-link the polymer matrix, releasing it into solution. | Used in the standard CER extraction protocol for isolating EPS from bacterial and fungal cultures for subsequent analysis [4]. |
| Propidium Iodide (PI) | A fluorescent dye that binds to nucleic acids, primarily DNA. Used to visualize the eDNA component of the EPS matrix. | Staining biofilm streamers for 3D geometric reconstruction via fluorescence microscopy, which is a prerequisite for CFD stress analysis [23]. |
| Sodium Hypochlorite (NaClO) | A common disinfectant and oxidative stressor. Used to determine the minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) against biofilms. | Evaluating how biofilm formation increases disinfectant tolerance; e.g., Salmonella Infantis biofilms required up to 8-fold higher NaClO for eradication [27]. |
| Crystal Violet (CV) | A dye that stains cells and the EPS matrix. Used in the CV assay to quantify total biofilm biomass attached to a surface. | Quantifying biofilm formation by pathogens like E. coli and P. aeruginosa on different food-contact surfaces over time [6]. |
| Hyaluronic Acid-Binding Peptide (HABP) | A peptide that binds to exopolysaccharides. Can be conjugated to carriers (e.g., PEG-lipid) to create EPS-binding probes or anti-biofilm agents. | Conjugated to liposomes to create "EPS-binding liposomes" that sterically block biofilm formation by S. aureus by anchoring to the matrix [28]. |
| DNase I | An enzyme that degrades DNA. Used to interrogate the structural role of eDNA in the EPS matrix. | Treatment of P. aeruginosa streamers causes their disintegration, proving the critical structural role of eDNA [23]. |
Computational Modeling of Biofilm Mechanics
Computational models are indispensable for deciphering the complex interplay of physical forces in biofilm morphology. A lattice-network model can simulate how biofilms grow and wrinkle by accounting for friction, adhesion, and nutrient availability [25].
- Model Setup: The biofilm is modeled as a 2D triangular spring-lattice network in 3D space, where each node represents a microscopic domain. Nodes interact via harmonic potentials for stretching and bending.
- Growth Dynamics: Biomass production follows Monod kinetics, dependent on local nutrient concentration solved via a reaction-diffusion equation. Growth increases the equilibrium length of the springs.
- Force Interactions: Adhesion to the substrate is modeled using a combination of Lennard-Jones and Yukawa potentials. Friction is implemented through nodal diffusivity.
- Key Insight: The model predicts that under high friction and uniform nutrients, compressive stresses are highest at the center, causing wrinkling to initiate there. With low nutrients or low friction, growth stops at the center, shifting wrinkle initiation to the nutrient-rich edge [25].
The following diagram visualizes this computational model and its key predictions:
The mechanical prowess of biofilms, governed by their EPS matrix, is a testament to the sophistication of microbial life. The properties of cohesiveness, adhesion, and resilience are not accidental but are precisely engineered through the matrix's composition and architecture. The identification of eDNA as a primary driver of stress-hardening behavior and the intricate balance between adhesion and friction revealed by computational models represent significant strides in this field. This understanding is pivotal for the future of biofilm research, providing a mechanistic foundation for developing novel strategies to disrupt detrimental biofilms in medicine and industry, or to enhance beneficial ones in environmental and energy applications. Future research will likely focus on precisely manipulating these mechanical properties through molecular and environmental interventions, offering exciting prospects for controlling the biofilm lifecycle.
Extracellular Polymeric Substances (EPS) are complex, hydrated polymers biosynthesized by a wide range of microorganisms. They form a protective matrix around microbial cells, constituting 50–90% of a biofilm's total organic matter [29]. This matrix is primarily composed of polysaccharides, proteins, nucleic acids (eDNA), and lipids, which together create a robust, three-dimensional architecture [1] [5]. Far from being a mere physical scaffold, the EPS matrix is functionally critical for microbial survival, providing an ideal environment for chemical reactions, nutrient entrapment, and most importantly, protection against a multitude of environmental stresses [5]. This technical guide delves into the mechanisms by which EPS serves as a protective barrier, framing this function within the broader context of biofilm structure research. For researchers and drug development professionals, understanding these mechanisms is paramount to developing strategies to combat biofilm-related infections and to harness the protective properties of EPS for industrial and environmental applications.
EPS Composition and Architectural Foundation
The protective capacity of EPS is intrinsically linked to its heterogeneous chemical composition and the gel-like structure it forms. This matrix is not a random assemblage of polymers but a functionally organized ecosystem that provides mechanical stability and a first line of defense.
Key Constituents: The EPS matrix is a composite material whose properties emerge from its core components:
- Polysaccharides: These are the most studied components, providing the structural backbone of the biofilm. They vary immensely in composition and structure, which directly influences the physical properties of the biofilm, such as porosity, density, and adhesion [5].
- Proteins: Structural proteins and enzymes within the EPS contribute to matrix stability and functionality. They can facilitate adhesion and perform metabolic functions, while also contributing to the structural integrity of the biofilm [4] [5].
- Extracellular DNA (eDNA): eDNA is a crucial structural component in many biofilms, contributing to cohesion, adhesion to surfaces, and acting as a source for horizontal gene transfer [5]. Its polyanionic nature also facilitates cation binding.
- Amino Sugars: Recent research has identified quantifiable amounts of amino sugars, including mannosamine (ManN) and galactosamine (GalN), within EPS. These constituents are now recognized as important markers of microbial EPS and may play a role in its protective functions [4].
Structural Dynamics: The formation of this protective matrix is a regulated process. It begins with the initial reversible attachment of planktonic cells to a surface, mediated by weak interactions like van der Waals forces and electrostatic interactions [1]. This attachment becomes irreversible through the secretion of the sticky EPS matrix, which develops into a mature biofilm with defined architectural features [1]. This structure is not static; it creates heterogeneous microenvironments with gradients of nutrients, oxygen, and pH, which influence microbial behavior and resistance profiles [1].
Table 1: Primary Constituents of Microbial Extracellular Polymeric Substances (EPS)
| Component | Primary Functions | Significance in Biofilm Protection |
|---|---|---|
| Polysaccharides | Structural scaffolding, gel formation, water retention | Creates a physical barrier; traps water and nutrients; source of carbon and energy [5]. |
| Proteins | Enzymatic activity, structural adhesion, matrix stability | Catalyzes reactions; strengthens matrix; facilitates surface attachment [5]. |
| Extracellular DNA (eDNA) | Matrix stability, genetic exchange, cation sequestration | Promotes cohesion and adhesion; facilitates antibiotic resistance gene transfer [5]. |
| Amino Sugars (e.g., GalN, ManN) | Structural integrity, microbial marker | Proposed role in matrix stability; serves as a specific indicator for microbial EPS in complex environments [4]. |
Mechanisms of Protection Against Environmental Stressors
The EPS matrix employs a multi-faceted strategy to shield microbial communities from diverse environmental threats, ranging from chemical toxins to physical desiccation.
Physical and Chemical Shielding
The dense, gel-like nature of EPS acts as a formidable diffusion barrier. This property significantly impedes the penetration of antimicrobial agents, including antibiotics and disinfectants, into the deeper layers of the biofilm [5]. The matrix's anionic nature, largely due to the presence of uronic acids and eDNA, allows it to sequester cationic heavy metals and other toxic compounds, effectively chelating and neutralizing them before they reach the microbial cells [5]. This sequestration is a critical mechanism for survival in polluted environments.
Enhanced Desiccation Resistance
A key ecological function of EPS, particularly in soil and other non-aquatic environments, is its ability to retain water. The highly hydrated polymers act as a reservoir, maintaining a moist microenvironment around the cells even when the external conditions are dry [5]. This capability protects microbes from osmotic stress and desiccation, ensuring their survival during periods of drought. This function is so vital that environmental stresses like salinity and drought are known triggers for increased EPS production in many microorganisms [5].
Physiological and Phenotypic Adaptations
Beyond physical blocking, the EPS matrix fosters conditions that lead to physiological resistance. The heterogeneous environment within the biofilm leads to metabolic and physiological diversity among the resident cells. Gradients of nutrients and oxygen can cause subpopulations of cells to enter a slow-growing or dormant state, making them less susceptible to antimicrobials that target active cellular processes [1]. Furthermore, the proximity of cells within the matrix, facilitated by eDNA, enhances the potential for horizontal gene transfer, rapidly disseminating resistance genes across the microbial community [5].
The following diagram illustrates the coordinated multi-level defense strategy employed by the EPS matrix against environmental stressors.
Quantitative Analysis of EPS Constituents Under Varying Conditions
Understanding how environmental factors influence EPS production and composition is critical for predicting microbial behavior. A 2025 study systematically analyzed EPS from ten soil bacterial and ten soil fungal species under different growth conditions [4]. The results demonstrate that EPS composition is strongly modified by microbial type, while environmental conditions drive the quantity of EPS produced.
Table 2: Environmental Impact on EPS Production and Composition [4]
| Experimental Factor | Impact on EPS-Carbohydrate/Protein Ratio | Key Findings on EPS Constituents |
|---|---|---|
| Carbon Source Quality | Higher ratio in cultures grown in starch media compared to glycerol. | EPS-carbohydrate concentration is highly responsive to substrate quality changes. |
| Surface Availability | Ratio increased in the presence of a quartz matrix. | Confirms EPS production is crucial for attachment and biofilm formation in structured environments like soils. |
| Microbial Type (Intrinsic Factor) | Compositional changes in other constituents (e.g., amino sugars, DNA) are linked to intrinsic microbial characteristics. | Amino sugars (Muranic Acid, Mannosamine, Galactosamine, Glucosamine) were quantified, opening pathways to study their specific roles. |
Experimental Protocols for EPS Research
For researchers aiming to investigate the protective roles of EPS, robust and reproducible methodologies are essential. The following protocol, adapted from a 2025 study, provides a detailed workflow for EPS extraction and constituent analysis from microbial cultures [4].
EPS Extraction using Cation Exchange Resin (CER)
Principle: The cation exchange resin displates divalent cations (e.g., Ca²⁺, Mg²⁺) that cross-link EPS polymers, leading to the release of the matrix components into solution [4].
Procedure:
- Microbial Cultivation: Cultivate microbial strains in appropriate liquid media (e.g., glycerol or starch-based). To study the effect of a surface, include a treatment with a sterile quartz matrix (0.4–0.8 mm) in the shake flasks. Incubate with shaking (100 rpm) at 30°C for a standard growth period (e.g., 4 days) [4].
- Sample Collection: After incubation, collect a 10 mL aliquot from the cell culture using a graduated cylinder.
- CER Addition: Add a pre-determined amount of cation exchange resin (e.g., Amberlite HPR1100) to the aliquot. The optimal resin dosage (g CER/g Volatile Suspended Solids) should be determined empirically for the specific microbial culture, as per methods established for Pseudomonas putida [4] [4].
- Extraction: Stir the mixture gently for a specified period (e.g., 2 hours) at low speed to avoid cell lysis.
- Separation: Centrifuge the sample (e.g., 10,000 × g, 20 minutes) to separate the CER and cells from the EPS-containing supernatant.
- Storage: Collect the supernatant, which contains the extracted EPS, and store at -20°C until analysis.
Analysis of Key EPS Constituents
The extracted EPS can be characterized using the following analytical techniques:
Total Carbohydrate Content:
Total Protein Content:
DNA Content:
- Method: Purify DNA by adding a phenol:chloroform:isoamyl alcohol solution (24:25:1 v/v/v) to the EPS extract. Mix by inversion, centrifuge, and recover the aqueous phase containing the nucleic acids. Quantify the DNA spectrofluorometrically or via other standard nucleic acid quantification methods [4].
Amino Sugar Analysis:
- Method: Analyze for key amino sugars (Muramic Acid, Mannosamine, Galactosamine, Glucosamine) using advanced chromatographic techniques (e.g., Gas Chromatography-Mass Spectrometry) following acid hydrolysis and derivatization of the EPS samples [4].
The experimental workflow from culture to data analysis is summarized below.
The Scientist's Toolkit: Key Research Reagents and Materials
This table details essential materials and reagents used in the featured EPS extraction and analysis protocols, providing a quick reference for experimental design.
Table 3: Essential Reagents and Materials for EPS Research
| Reagent/Material | Specification/Example | Primary Function in Protocol |
|---|---|---|
| Cation Exchange Resin (CER) | Amberlite HPR1100 [4] | Displaces divalent cations to disrupt the EPS matrix and release polymers into solution. |
| Quartz Matrix | SiO₂, 0.4–0.8 mm, SOM-free [4] | Provides a standardized inert surface to study the effect of solid interfaces on EPS production. |
| Hydrolysis Acid | 0.75 M Sulfuric Acid (H₂SO₄) [4] | Hydrolyzes polysaccharide chains into monomeric sugars for colorimetric quantification. |
| Colorimetric Assay Reagents | BCA Reagent; Lowry Reagent (CuSO₄ & Folin-Ciocalteu) [4] | Enable the spectrophotometric quantification of total carbohydrate and protein content, respectively. |
| DNA Extraction Solvent | Phenol:Chloroform:Isoamyl Alcohol (24:25:1) [4] | Purifies and isolates extracellular DNA (eDNA) from the complex EPS mixture. |
| Buffer Solution | Phosphate Saline Buffer (PBS) [4] | Used for storing EPS extracts and diluting hydrolysates for analysis to maintain pH and ionic strength. |
The spatial organization of biofilms into complex, three-dimensional structures represents a fundamental aspect of microbial life, facilitating the formation of heterogeneous microenvironments and nutrient gradients that are critical for biofilm resilience and function. This architectural complexity is fundamentally governed by the extracellular polymeric substances (EPS), a self-produced matrix that serves as both the structural scaffold and functional mediator of the biofilm ecosystem [6] [1]. The EPS matrix, composed of polysaccharides, proteins, lipids, and extracellular DNA (eDNA), creates a protective niche for embedded microorganisms and directly enables the establishment of chemical and physical gradients that drive microbial heterogeneity [1] [18].
Within this EPS-delimited architecture, microorganisms cease to exist as homogeneous populations and instead form structured communities with distinct spatial distributions. This organization is not random; it results from coordinated microbial behaviors and responses to environmental conditions, culminating in a system where gradients of nutrients, oxygen, metabolic byproducts, and signaling molecules create a mosaic of microenvironments [1]. These gradients impose divergent selective pressures on microbial subpopulations, leading to phenotypic and metabolic heterogeneity that underpins many biofilm-specific properties, including enhanced tolerance to antimicrobial agents and environmental stresses [1] [31]. This whitepaper examines the mechanisms driving biofilm spatial organization, the technical approaches for its quantification, and the implications for therapeutic intervention.
Analytical Frameworks for Quantifying Spatial Organization
Understanding the spatial organization of biofilms requires a multidisciplinary approach that integrates quantitative metrics with advanced visualization techniques. The following analytical frameworks enable researchers to deconstruct the relationship between EPS composition and the formation of microenvironments and nutrient gradients.
Table 1: Core Analytical Techniques for Assessing Biofilm Spatial Organization
| Technique Category | Specific Method | Primary Measurable Parameters | Application in Spatial Analysis |
|---|---|---|---|
| Biomass Quantification | Crystal Violet (CV) Assay [6] | Total biofilm biomass (cells + EPS) | Measures overall biofilm formation capacity on different surfaces. |
| Colony Forming Unit (CFU) Assay [6] | Viable bacterial cell count | Quantifies cultivable cells within biofilm, indicating metabolic activity distribution. | |
| * Metabolic Activity profiling* | MTT Assay [6] | Cellular metabolic activity | Probes metabolic heterogeneity and gradient formation within biofilm microenvironments. |
| Chemical Composition Analysis | Fourier Transform Infrared (FTIR) Spectroscopy [6] | Functional groups, chemical bonds in EPS | Identifies chemical constituents of EPS matrix contributing to gradient formation. |
| Nuclear Magnetic Resonance (NMR) [6] | Monomeric composition of EPS | Elucidates molecular structure and composition of EPS components. | |
| Structural Visualization | Confocal Laser Scanning Microscopy (CLSM) [6] [32] | 3D architecture, thickness, cell distribution, water channels | Visualizes and quantifies 3D biofilm topography, including voids and channels. |
| Scanning Electron Microscopy (SEM) [6] [32] | High-resolution surface morphology | Reveals ultrastructural details of EPS and cell-EPS interactions at high resolution. | |
| Computational Image Analysis | BiofilmQ Software [33] | Hundreds of global and internal 3D parameters, fluorescence correlations | Enables high-throughput, spatially resolved quantification of internal biofilm properties and microenvironments. |
The data derived from these complementary techniques reveal that biofilm architecture is highly dynamic and influenced by multiple factors. For instance, studies on Pseudomonas aeruginosa and Escherichia coli have demonstrated that biofilm density, measured in CFU/cm², increases significantly over time and varies substantially across different surface materials like stainless steel, silicone rubber, aluminum, and polyethylene terephthalate [6]. Furthermore, analytical techniques like FTIR and NMR spectroscopy have identified that the chemical properties of EPS—governed by functional groups such as carboxyl and hydroxyl groups—are pivotal in bacterial aggregation and ultimately influence the final biofilm architecture and its associated gradients [6].
Experimental Workflows for Architectural and Gradient Analysis
A robust experimental pipeline for analyzing biofilm spatial organization combines cultivation, visualization, and computational quantification. The workflows below detail standardized protocols for assessing these critical parameters.
Workflow 1: 3D Architecture and Biovolume Quantification
This protocol outlines the process for growing biofilms and quantifying their 3D structure and biovolume, essential first steps in spatial analysis [32].
Protocol Details:
- Biofilm Growth: Sterile glass coverslips are placed vertically in falcon tubes containing a nutrient medium (e.g., Eaton's broth or Tryptic Soy Broth). The medium is then inoculated with a 1:100 dilution of a planktonic culture and incubated statically at 37°C in a 5% CO₂ atmosphere for periods ranging from 3 days (early growth) to 7 days (late growth) [32].
- Sample Fixation and Staining: After incubation, coverslips are washed twice with phosphate-buffered saline (PBS) to remove non-adherent cells. Biofilms are fixed with a 4% formaldehyde solution in PBS for 10 minutes at room temperature. Fixed biofilms are then stained with a fluorescent nucleic acid dye, such as propidium iodide ( diluted 1:9 in PBS, for 15 minutes in the dark [32].
- Imaging and Quantification: Processed coverslips are mounted on slides and imaged using a CLSM equipped with appropriate lasers and detectors (e.g., 514-nm laser with a 539–629 nm detection window for propidium iodide). A high-numerical-aperture oil immersion objective (e.g., 63x) is used to generate high-resolution Z-stacks. These 3D image datasets are then processed using software like Amira or BiofilmQ: noise is reduced with a median filter, microcolonies are defined by thresholding, and the biovolume is quantified from at least five non-overlapping adhesion points on the surface [32]. This workflow can reveal significant increases in median biofilm volume between early and late growth stages, highlighting architectural development [32].
Workflow 2: Computational Image Cytometry for Microenvironment Mapping
For a deeper analysis of biofilm internal heterogeneity, BiofilmQ software can be used to perform image cytometry on 3D datasets, mapping gradients and microenvironments [33].
Protocol Details:
- Image Acquisition and Segmentation: Biofilms are imaged using 3D fluorescence microscopy. The resulting image stack is loaded into BiofilmQ, and the first critical step is the accurate segmentation of the biofilm biovolume from the background. This can be achieved using built-in automatic algorithms (e.g., Otsu, Ridler-Calvard), semi-manual thresholding, or by importing a pre-segmented image generated by other tools like ilastik or U-Net [33].
- Cube-based Cytometry and Analysis: Once the biovolume is defined, BiofilmQ dissects it into a user-defined grid of cubical objects. For each cube, the software computes dozens of properties, including fluorescence intensity, local density, and texture. Crucially, it also calculates the spatial context of each cube, such as its distance to the biofilm's outer surface or the substratum. This allows for the correlation of cellular phenotype with physical position, enabling direct quantification of gradients [33].
- Data Visualization and Gating: Similar to flow cytometry, researchers can apply gates to the cube population data to isolate and analyze specific subpopulations based on their cytometric properties. BiofilmQ provides integrated tools to visualize these analyses through various graphs, such as histograms, scatter plots, and spatial maps, directly showing the distribution of parameters like metabolic activity or specific gene expression across the biofilm's 3D structure [33].
The Scientist's Toolkit: Essential Research Reagent Solutions
The experimental investigation of biofilm spatial organization relies on a curated set of reagents, materials, and software tools. The following table catalogues key solutions for researchers in this field.
Table 2: Essential Research Reagents and Tools for Spatial Organization Studies
| Reagent/Tool | Specific Example | Function in Spatial Analysis |
|---|---|---|
| Culture Surfaces | Stainless Steel (SS), Silicone Rubber (SR), Glass Coverslips [6] [32] | Provides abiotic substrate for biofilm growth; surface properties influence attachment and final architecture. |
| Fluorescent Stains | Propidium Iodide [32] | Nucleic acid stain for visualizing and quantifying total biomass in 3D using CLSM. |
| EPS Extraction Tool | Cation Exchange Resin (CER) [18] | Extracts EPS matrix from microbial cultures for subsequent compositional analysis (e.g., carbohydrates, proteins). |
| Metabolic Activity Probe | MTT Reagent [6] | Measures metabolic activity of cells within biofilms via reduction to formazan, indicating viability and heterogeneity. |
| Biomass Stain | Crystal Violet (CV) [6] | Quantifies total biofilm biomass (cells and EPS) adhered to surfaces. |
| Composition Analysis Kits | BCA Assay, Lowry Assay [18] | Colorimetric quantification of total carbohydrates and proteins, respectively, in extracted EPS. |
| Computational Tool | BiofilmQ Software [33] | Image cytometry platform for automated, high-throughput quantification of 3D biofilm internal structure and properties. |
| 3D Visualization Software | Amira Software [32] | Processes 3D image datasets from CLSM for visualization, filtering, thresholding, and biovolume quantification. |
The spatial organization of biofilms into a heterogeneous landscape of microenvironments, facilitated by the EPS matrix, is a primary determinant of their recalcitrance and ecological success. The technical frameworks outlined herein—from foundational CFU counts and CLSM to the advanced computational cytometry enabled by BiofilmQ—provide researchers with a powerful arsenal to dissect this complexity. Quantifying parameters such as biovolume, matrix composition, and the spatial distribution of metabolic activity is no longer an endpoint but a starting point for developing more effective interventions.
The future of biofilm management lies in leveraging this spatial understanding to design targeted strategies that disrupt critical architectural features. This could involve enzymes that degrade key EPS components like Pel and Psl, which maintain aggregate coherence [31], or small molecules that interfere with the sensing mechanisms guiding spatial differentiation. For drug development professionals, the recognition that bacterial aggregates in chronic infections are often non-attached [31] necessitates a shift in therapeutic targets away from surface-adhesion inhibition and toward mechanisms that disperse existing clusters or sensitize them to antibiotics. By moving beyond a purely antimicrobial approach to one that architecturally deconstructs the biofilm fortress, we can more effectively address the persistent challenges biofilms pose in both clinical and industrial settings.
Analytical and Disruption Techniques: From EPS Characterization to Targeted Degradation
Extraction and Quantification Methods for EPS Constituents
Extracellular Polymeric Substances (EPS) form the foundational matrix of biofilms, the dominant microbial lifestyle in natural, industrial, and clinical environments [34]. This complex hydrogel, accounting for over 90% of the biofilm's volume, provides structural integrity, mediates adhesion, and protects microbial communities from environmental stresses, antibiotics, and disinfectants [34] [35]. A comprehensive understanding of biofilm structure-function relationships hinges on the precise extraction and accurate quantification of EPS constituents. The EPS matrix is a complex mixture of biomolecules, primarily polysaccharides, proteins, and nucleic acids, along with other macromolecules like lipids and amino sugars, whose composition varies significantly based on microbial species, environmental conditions, and biofilm age [34] [4]. This technical guide details state-of-the-art methodologies for EPS extraction and constituent analysis, providing a critical resource for researchers aiming to elucidate the role of EPS in biofilm phenotype and function within the broader context of biofilm research.
EPS Composition and Functional Significance
The biofilm matrix is a complex, dynamic environment whose composition dictates its physical and functional properties. Proteins and exopolysaccharides represent the key components, comprising 40% to 95% of EPS [34]. Exopolysaccharides are high-molecular-weight polymers (500-2000 kDa) that act as adhesive polymers, maintaining structural integrity [34]. Proteins include both enzymatic proteins, functioning as an external digestive system, and structural proteins, which contribute to the matrix's anionic properties and cohesive stability [34]. Furthermore, environmental factors strongly influence EPS production and composition. A 2025 study demonstrated that EPS composition is strongly modified by microbial type, while environmental conditions primarily drive differences in EPS production levels. The EPS-carbohydrate/protein ratio was higher for cultures grown in starch media compared to glycerol and increased in the presence of a quartz surface [4].
Table 1: Major Constituents of Extracellular Polymeric Substances (EPS)
| EPS Constituent | Primary Functions | Key Characteristics |
|---|---|---|
| Polysaccharides | Structural integrity, cell-surface attachment, adhesion, cohesion, nutrient source [34] [4]. | High-MW polymers (500-2000 kDa); can be homopolymeric or heteropolymeric [34]. |
| Proteins | Enzymatic activity, structural support, electrostatic interactions, electron donor/acceptor in redox reactions [34]. | Includes enzymatic and structural types; anionic properties from diacid amino acids [34]. |
| Extracellular DNA (eDNA) | Horizontal gene transfer, structural stability, cell aggregation [4] [34]. | Promotes genetic exchange and contributes to structural integrity of the matrix [4]. |
| Amino Sugars | Microbial residue markers, potential structural role in EPS [4]. | Include muramic acid (MurN), glucosamine (GlcN), galactosamine (GalN), mannosamine (ManN) [4]. |
| Lipids | Hydrophobicity, structural heterogeneity, potential signaling [6]. | Identified in EPS architecture of food-borne pathogens; quantified gravimetrically [6]. |
EPS Extraction Techniques
The first critical step in EPS analysis is the extraction of the matrix from the microbial cells with minimal contamination from intracellular components. Extraction techniques can be broadly categorized into physical and chemical methods, each with varying degrees of efficiency and destructiveness [36].
Chemical Extraction Methods
Chemical methods are widely used and often provide high EPS yields.
- Cation Exchange Resin (CER) Method: This method, used in a recent 2025 study, involves adding a resin like Amberlite to cell cultures. The resin displaces divalent cations that cross-link EPS polymers, thereby liberating the EPS into the solution for subsequent analysis [4]. This method is considered relatively gentle and minimizes cell lysis [36].
- Ethanol Precipitation with Trichloroacetic Acid (TCA) De-proteinization: A common protocol for isolating exopolysaccharides from bacterial cultures involves centrifuging cultures to remove cells, adding TCA to the supernatant to precipitate proteins, and then using cold ethanol to precipitate the polysaccharides from the protein-free supernatant. The resulting pellet is dialyzed and lyophilized to yield crude EPS powder [37].
- Ethylenediaminetetraacetic Acid (EDTA) Extraction: EDTA acts as a chelating agent, binding to divalent cations and destabilizing the biofilm matrix. One study found that EDTA extraction yielded the highest EPS with minimal cell lysis for Rhodopseudomonas acidophila [36]. This method is also effective for inducing biofilm dispersal [35].
Physical Extraction Methods
Physical methods often involve mechanical disruption of the biofilm structure.
- Centrifugation: A fundamental step in nearly all extraction protocols used to separate bacterial cells from the EPS-containing supernatant after chemical treatment or to harvest EPS precipitates [4] [37].
- Sonication: The application of ultrasound energy can help dislodge EPS and break apart biofilm aggregates. However, careful optimization is required, as excessive energy can cause cell lysis and contaminate the EPS extract with intracellular materials [36].
The choice of extraction method significantly impacts the yield, composition, and subsequent analysis of EPS. Studies have shown that chemical methods generally yield higher EPS quantities compared to physical ones, though they may introduce contaminants or alter native EPS structures [36]. Therefore, the selection of an extraction protocol must align with the specific research objectives and the microbial system under investigation.
Quantification of EPS Constituents
Following extraction, a suite of analytical techniques is employed to quantify the individual components of EPS. The table below summarizes the common methods for quantifying major EPS constituents.
Table 2: Analytical Methods for Quantifying EPS Constituents
| Target Constituent | Quantification Method | Principle | Key Applications & Notes |
|---|---|---|---|
| Total Carbohydrates | Phenol-Sulfuric Acid Assay [37] | Carbohydrates dehydrated to furfurals which react with phenol to form colored compound. Absorbance at 490nm [37]. | General polysaccharide quantitation; uses glucose as standard [37]. |
| Spectrophotometric Acid-Phenol Assay [38] | An inexpensive, practical microscale method for polysaccharides in aqueous solution. | Designed for soil EPS; high precipitation recovery [38]. | |
| Bicinchoninic Acid (BCA) Assay [4] | After acid hydrolysis, carbohydrates are measured via BCA microplate assay (Absorbance 562 nm) [4]. | Used for EPS-hydrolysates; neutralization step may be omitted after dilution [4]. | |
| Total Proteins | Lowry Assay [4] | Reaction with Folin-Ciocalteu reagent after incubation with copper sulphate. Absorbance at 750 nm [4]. | Standard method for EPS-protein estimation [4]. |
| Bradford Assay [37] | Binding of Coomassie Brilliant Blue dye to protein. Absorbance at 595 nm [37]. | Used for EPS protein content; based on standard curve [37]. | |
| Extracellular DNA (eDNA) | Purification & Quantification [4] | Phenol:chloroform:isoamyl alcohol extraction followed by quantification. | Purifies DNA from EPS extracts for analysis [4]. |
| Amino Sugars | Chromatographic Analysis [4] | Analysis of hydrolysates for Muramic Acid, Glucosamine, Galactosamine, Mannosamine. | Important markers for microbial residues; GalN and ManN are EPS-exclusive [4]. |
| Lipids | Gravimetric Analysis [37] | Extraction with chloroform-methanol, solvent evaporation, and weighing. | Lipid content measured by weight [37]. |
Advanced and Emerging Quantification Techniques
Beyond the standard colorimetric assays, advanced techniques provide deeper insights into EPS composition and structure.
- Fourier Transform Infrared (FTIR) Spectroscopy: This technique identifies functional groups and chemical bonds within purified EPS. For example, FTIR has been used to confirm the presence of amino groups, amides, carboxylic acids, hydroxyl groups, and phosphates in EPS from Pseudomonas aeruginosa [37]. It provides information on the chemical content and proportions of different EPS [35].
- Rapid Spectroscopic Methods: New methods are being developed to overcome the lengthy traditional analysis. A 2025 study presented a method using UV-visible spectroscopy coupled with partial least squares (PLS) regression to quantify the exopolysaccharide pullulan directly in fermentation broth. This non-destructive method requires minimal sample preparation and allows for potential real-time bioprocess monitoring [39].
- Nuclear Magnetic Resonance (NMR): NMR spectroscopy is used to identify the monomeric composition and detailed chemical structure of organic molecules in EPS [6].
The following workflow diagram illustrates the sequential process from biofilm collection to the quantification of individual EPS constituents, integrating the methods described above.
Figure 1: Workflow for EPS Extraction and Constituent Analysis
Advanced Techniques for In-situ EPS Analysis
While extraction-based methods are fundamental, they often disrupt the native biofilm architecture. A suite of advanced, often non-destructive, techniques allows for the in-situ analysis of EPS, preserving its spatial distribution and relationship with microbial cells.
- Confocal Laser Scanning Microscopy (CLSM): This technique enables the three-dimensional visualization of biofilms by using fluorescent dyes to tag specific EPS components like polysaccharides, proteins, and nucleic acids. It allows researchers to analyze biofilm thickness, cell distribution, and the spatial arrangement of cells within the EPS matrix without physical destruction of the sample [34] [6].
- Atomic Force Microscopy (AFM): AFM provides nanoscale resolution of biofilm structures, including individual cells, appendages like flagella, and the fine structure of the EPS matrix [40]. Recent advancements, such as automated large-area AFM combined with machine learning, overcome traditional limitations by enabling high-resolution imaging over millimeter-scale areas. This provides unprecedented views of spatial heterogeneity and cellular morphology during early biofilm formation [40].
- Scanning Electron Microscopy (SEM): SEM provides high-resolution, topographical images of the biofilm surface, revealing intricate details of its structure and the interactions between cells and the contact surface. It is instrumental in visualizing morphological characteristics at different developmental stages [6].
A multidisciplinary approach that combines multiple techniques is often the most powerful strategy. For instance, the combined use of Raman Microscopy and CLSM provides comprehensive insights into EPS composition and spatial distribution without altering the native state, overcoming the limitations of staining alone [36].
The Scientist's Toolkit: Key Research Reagents and Materials
This table lists essential reagents, materials, and instruments used in the featured experiments for EPS extraction and quantification, serving as a practical reference for laboratory planning.
Table 3: Essential Research Reagents and Materials for EPS Analysis
| Item Name | Function in EPS Research | Specific Application Example |
|---|---|---|
| Cation Exchange Resin (CER) | Displaces cross-linking cations to liberate EPS from the biofilm matrix [4]. | EPS extraction from soil bacterial and fungal cultures (e.g., Amberlite HPR1100) [4]. |
| Trichloroacetic Acid (TCA) | Precipitates proteins from the EPS solution to deproteinize the sample [37]. | Used in TCA/Ethanol precipitation method for purifying bacterial exopolysaccharides [37]. |
| Ethylenediaminetetraacetic Acid (EDTA) | Chelates divalent cations to destabilize the biofilm matrix and induce dispersal [35]. | Effective chemical extraction agent; shown to yield high EPS with minimal cell lysis [36]. |
| Phenol & Sulfuric Acid | Key reagents in the colorimetric reaction for total carbohydrate quantification [37]. | Phenol-sulfuric acid method for determining total carbohydrate content in crude EPS [37]. |
| Folin-Ciocalteu Reagent | Reacts with proteins in the alkaline environment of the Lowry assay [4]. | Quantification of total protein content in EPS extracts [4]. |
| Quartz Matrix | Provides a solid surface to stimulate EPS production under physically relevant conditions [4]. | Used in cultivation flasks to study the effect of surface presence on EPS production [4]. |
| Confocal Laser Scanning Microscope (CLSM) | For non-destructive, 3D visualization of biofilm architecture and EPS distribution [34]. | In-situ analysis of biofilm structure, thickness, and component localization [6]. |
| Atomic Force Microscope (AFM) | For high-resolution topographical and nanomechanical imaging of biofilms and EPS [40]. | Visualizing cellular morphology, flagella, and EPS fibers at the nanoscale [40]. |
The precise extraction and accurate quantification of EPS constituents are fundamental to advancing our understanding of biofilm biology. As this guide outlines, a robust methodological framework exists, encompassing traditional chemical assays and advanced spectroscopic and microscopic techniques. The choice of method is critical, as it directly influences the interpretation of EPS composition and, consequently, its function. Future directions in the field point toward the integration of multidisciplinary methods, the application of machine learning and big data for analysis [41] [40], and the development of non-destructive, real-time monitoring techniques [39]. By carefully selecting and applying these methods, researchers can continue to unravel the complexities of the EPS matrix, ultimately leading to innovative strategies for managing biofilms in health, industry, and the environment.
The structural integrity and functional resilience of microbial biofilms are primarily governed by the extracellular polymeric substance (EPS), a complex, gel-like matrix that constitutes 50% to 90% of the biofilm's total organic matter [2] [42]. This matrix is a self-produced, highly hydrated network of biopolymers, including polysaccharides, proteins, nucleic acids, lipids, and humic substances, which forms a protective sanctuary for microbial communities [43] [44]. Understanding the structure-function relationships within the EPS is paramount for controlling biofilms in contexts ranging from medical implants and drug development to industrial water systems and environmental bioremediation.
Advanced analytical techniques are required to decipher the spatial organization, chemical composition, and nanomechanical properties of the EPS. This technical guide focuses on three cornerstone methodologies: Fourier Transform Infrared Spectroscopy (FTIR) for chemical characterization, Confocal Laser Scanning Microscopy (CLSM) for three-dimensional structural visualization, and Atomic Force Microscopy (AFM) for probing mechanical properties. Used in concert, these tools provide a multi-scale, multi-parameter framework for elucidating the role of EPS in biofilm architecture and stability, thereby informing targeted strategies for biofilm manipulation in pharmaceutical and clinical applications.
Each analytical technique interrogates the biofilm matrix at different length scales and provides complementary information, as summarized in Table 1.
Table 1: Comparative overview of key analytical techniques for biofilm EPS analysis.
| Technique | Primary Function | Spatial Resolution | Key Measurable Parameters | Sample Conditions |
|---|---|---|---|---|
| FTIR | Chemical identification & quantification | ~1-20 µm [45] | Functional groups, molecular bonds, secondary protein structure [45] | Can be measured in hydrated or dry state |
| CLSM | 3D structural visualization | ~200-300 nm laterally [46] | Biomass volume, thickness, porosity, co-localization of components [46] | Typically requires fluorescent staining; can image live biofilms in liquid |
| AFM | Topographical imaging & nanomechanical probing | <1 nm (true atomic resolution possible) [40] | Cohesive strength, elasticity (Young's modulus), adhesion forces [47] [40] | Can operate in ambient air or liquid, preserving native state |
Fourier Transform Infrared Spectroscopy (FTIR) for EPS Chemical Characterization
Principle and Application
FTIR spectroscopy analyzes the vibrational characteristics of chemical bonds within a sample. When infrared radiation is applied, specific bonds absorb energy at characteristic frequencies, producing a spectrum that serves as a molecular fingerprint [45]. This non-destructive technique is exceptionally valuable for identifying the major biochemical constituents of the EPS matrix without the need for extensive sample preparation.
FTIR has been successfully deployed to characterize EPS from various bacterial sources. For instance, in a study of Pseudomonas furukawaii PPS-19, FTIR revealed that its EPS was composed of primary alkane, amines, halide, hydroxyl groups, uronic acid, and saccharides [44]. Similarly, analysis of Streptococcus spp. biofilms identified specific spectral signatures for proteins, lipids, and polysaccharides, providing insights into the matrix composition of cariogenic bacteria [45].
Key Spectral Signatures in EPS Analysis
The interpretation of FTIR spectra relies on identifying absorption bands in specific wavenumber regions associated with different functional groups. Table 2 outlines the principal vibrational modes relevant to common EPS components.
Table 2: Key FTIR spectral assignments for major EPS components [45].
| Wavenumber (cm⁻¹) | Assignment and Functional Group | Associated EPS Component |
|---|---|---|
| 3200–3350 | ν(N-H), ν(O-H), Amide A | Proteins, Polysaccharides, Water |
| 2950–2960 | νas(CH₃) | Lipids |
| 2920–2940 | νas(CH₂) | Lipids |
| 2850–2860 | νs(CH₂) | Lipids |
| 1730–1740 | ν(C=O) | Phospholipids |
| 1700–1600 (Amide I) | 80% ν(C=O), 20% ν(C-N) | Proteins (secondary structure) |
| 1600–1500 (Amide II) | 60% τ(N-H), 30% ν(C-N) | Proteins |
| 1242–1230 | νas(PO₂⁻) | Nucleic Acids (eDNA), Phospholipids |
| ~1086 | νs(PO₂⁻) | Nucleic Acids (eDNA), Phospholipids |
| 1144–1137 | Oligosaccharides | Polysaccharides |
| 1080–1070 | ν(C-C), β-glucan bonds | Polysaccharides |
| 1046–999 | Skeletal vibration of d-glucose | Polysaccharides |
| 929 | (1→3)-α-d-glucan | Specific exopolysaccharides (e.g., glucans) |
| 860–852 | (1→3),(1→6)-α-d-glucan | Specific exopolysaccharides |
Experimental Protocol: FTIR Analysis of Purified EPS
1. EPS Extraction (Ethanol Precipitation) [44]:
- Grow biofilm on a suitable substrate (e.g., glass beads) for 48-72 hours under desired conditions.
- Harvest biofilm by vortexing or scraping into a suspension.
- Centrifuge the suspension (e.g., 7,000 rpm) to pellet cells and insoluble matrix.
- Precipitate EPS from the supernatant by adding cold ethanol (typically 2-3 volumes) and incubating at 4°C for several hours or overnight.
- Collect EPS by centrifugation, wash with an ethanol-acetone mixture, and air-dry the pellet.
2. FTIR Measurement:
- Prepare a KBr pellet by mixing ~1 mg of the dried EPS with 100-200 mg of potassium bromide (KBr) and pressing under vacuum, or use an ATR-FTIR (Attenuated Total Reflectance) accessory which requires minimal sample preparation.
- Acquire spectra over a range of 4000-400 cm⁻¹.
- Process spectra by performing baseline correction and normalization (often to the Amide I band) for comparative analysis.
Confocal Laser Scanning Microscopy (CLSM) for 3D Biofilm Architecture
Principle and Application
CLSM generates high-resolution, three-dimensional images of biofilm structure by using a pinhole to eliminate out-of-focus light. Its key advantage for biofilm research is the ability to non-invasively observe live biofilms in their hydrated state over time, providing dynamic spatial information about cellular distribution and the localization of specific EPS components [46].
CLSM has been instrumental in defining the roles of specific EPS components. For example, studies of Pseudomonas aeruginosa have used fluorescently conjugated lectins to visualize the spatial distribution of exopolysaccharides like Psl and Pel, revealing their distinct and sometimes co-localized patterns within the biofilm architecture [46]. Furthermore, CLSM has been used to correlate EPS manipulation with structural changes; treatment of Staphylococcus epidermidis biofilms with EPS-degrading enzymes like proteases and dispersin B resulted in significant reduction in biofilm thickness and biovolume, as quantified by CLSM analysis [43].
Experimental Protocol: CLSM for EPS Visualization
1. Biofilm Cultivation:
- Grow biofilms in optimal reactors. Flow-cell reactors are highly recommended as they provide constant nutrient supply and shear stress, generating biofilms that closely mimic natural environments [43] [46].
2. Staining EPS Components:
- General Nucleic Acids & Cells: Use SYTO 9 (green fluorescence) to stain all cells.
- Polysaccharides: Apply fluorescently conjugated lectins (e.g., ConA, WGA). For instance, Concanavalin A (Con A) Alexa Fluor 633 binds to α-mannopyranosyl and α-glucopyranosyl residues [44].
- Extracellular DNA (eDNA): Use propidium iodide which penetrates cells with compromised membranes, or specific fluorescent dyes that preferentially stain eDNA.
- Proteins: Staining can be achieved with FITC (fluorescein isothiocyanate) under controlled conditions [44].
- Incubate stains in the dark for a specified period, then gently rinse to remove unbound dye.
3. Image Acquisition and Analysis:
- Acquire Z-stack images through the entire biofilm depth with an appropriate step size (e.g., 1 µm).
- Use image analysis software such as COMSTAT [46] or ImageJ to quantify parameters like:
- Total Biomass (µm³/µm²)
- Average Thickness (µm)
- Substrate Coverage (%)
- Surface Area to Biovolume Ratio
- Co-localization coefficients of different fluorescent signals.
Figure 1: CLSM Experimental Workflow. The process from biofilm growth to quantitative 3D analysis.
Atomic Force Microscopy (AFM) for Nanomechanical Profiling
Principle and Application
AFM operates by scanning a sharp probe attached to a flexible cantilever across a sample surface. Interactions between the probe tip and the sample cause cantilever deflections, which are measured to generate topographical images with nanometer resolution. Crucially, AFM can also measure nanomechanical properties by performing force spectroscopy, where force-distance curves are collected to quantify parameters such as elastic modulus (stiffness), adhesion, and cohesion [47] [40].
AFM is uniquely capable of directly measuring biofilm cohesive strength. A foundational study used an AFM cantilever to abrade a defined area of a biofilm, calculating the cohesive energy from the volume of displaced material and the frictional energy dissipated [47]. This method demonstrated that cohesive strength increases with biofilm depth and can be enhanced by the presence of divalent cations like calcium, which promote ion bridging within the EPS matrix [43] [47]. Recent advancements, such as large-area automated AFM combined with machine learning, now enable the correlation of nanoscale properties with millimeter-scale biofilm organization, revealing intricate features like flagellar interactions and honeycomb patterning in early biofilms [40].
Experimental Protocol: AFM Cohesive Strength Measurement
1. Biofilm Preparation:
- Grow a thin, uniform biofilm on a solid substrate (e.g., a membrane).
- For measurements in controlled humidity, equilibrate the sample in a chamber with saturated NaCl solution (~90% relative humidity) for 1 hour to maintain consistent hydration [47].
2. AFM Scanning and Abrasion:
- Mount the sample in the AFM and select a sharp, calibrated Si₃N₄ tip.
- Step 1: Image a 5x5 µm area at a low applied load (~0 nN) to obtain a non-perturbative topographical baseline.
- Step 2: Zoom into a 2.5x2.5 µm sub-region and perform abrasive scanning with repeated raster scans at an elevated load (e.g., 40 nN).
- Step 3: Return to low load and re-image the 5x5 µm area to visualize the abraded region.
3. Data Analysis:
- Calculate displaced volume: Subtract the post-abrasion height image from the pre-abrasion image.
- Determine frictional energy: Analyze the lateral (frictional) signal during abrasive scanning and convert to energy using cantilever calibration.
- Compute cohesive energy (Γ): Γ = (Total Frictional Energy Dissipated) / (Total Biofilm Volume Displaced). Units are typically nanojoules per cubic micrometer (nJ/µm³) [47].
Integrated Workflow and Research Reagents
The true power of these techniques is realized when they are used in an integrated fashion. For instance, a study might use CLSM to identify structural defects in a mutant biofilm, employ FTIR to confirm that these defects are linked to a specific reduction in polysaccharide content, and finally use AFM to measure the consequent loss in mechanical robustness [43] [48]. This multi-pronged approach provides a comprehensive picture of EPS structure-function relationships.
Table 3: Essential Research Reagent Solutions for EPS and Biofilm Analysis.
| Reagent / Kit | Function / Target | Specific Example & Use Case |
|---|---|---|
| Fluorescent Lectins | Label specific polysaccharides in EPS for CLSM. | Concanavalin A (ConA) Alexa Fluor 633: Binds α-mannopyranosyl/glucopyranosyl residues in polysaccharides like Psl [44] [46]. |
| Nucleic Acid Stains | Differentiate live cells and eDNA. | SYTO 9: General nucleic acid stain for all cells. Propidium Iodide: Penetrates damaged membranes, can stain eDNA [46]. |
| EPS-Degrading Enzymes | Selectively disrupt EPS components to study function. | Proteinase K/Trypsin: Degrade protein components. Dispersin B: Degrades poly-N-acetylglucosamine (PNAG). DNase I: Degrades extracellular DNA (eDNA) [43]. |
| CDC Biofilm Reactor | Standardized system for growing reproducible, shear-consistent biofilms. | Used to grow mature S. epidermidis biofilms for EPS modification studies, superior to quiescent well-plate cultures [43]. |
Figure 2: Integrated Analytical Approach. Combining FTIR, CLSM, and AFM data from modified EPS builds a comprehensive structure-function model.
FTIR, CLSM, and AFM provide an indispensable toolkit for deconstructing the complex nature of biofilm EPS. FTIR delivers a quantitative chemical inventory, CLSM maps the three-dimensional spatial relationships of these components, and AFM quantifies the resulting mechanical integrity that defines a biofilm's resistance to removal. The integration of these techniques allows researchers to move beyond descriptive studies and establish causative links between the molecular composition of the EPS and the macroscopic properties of the biofilm. For drug development professionals, this multi-faceted understanding is critical for designing effective anti-biofilm strategies, whether by enzymatically disrupting the matrix, interfering with its assembly, or sensitizing it to conventional antimicrobials. As these technologies continue to advance, particularly with automation and machine learning integration [40], their combined application will undoubtedly unlock novel therapeutic avenues for combating biofilm-associated infections.
Bacterial biofilms are structured communities of microbial cells encased in a self-produced matrix known as extracellular polymeric substances (EPS). This EPS matrix constitutes the primary architectural component of biofilms, often metaphorically described as the "house of biofilm cells" because it determines the immediate conditions of life for the embedded microorganisms [8]. The EPS provides mechanical stability, retains water, and acts as a protective barrier against environmental threats, including antimicrobial treatments and host immune responses [8]. The composition of the EPS is complex and dynamic, primarily consisting of polysaccharides, proteins, extracellular DNA (eDNA), lipids, and other biomolecules [49]. This structural complexity contributes to the remarkable resistance of biofilms to conventional antibiotics, which can be up to 1,000 times more effective against planktonic cells than their biofilm counterparts [49].
Targeting the EPS matrix has emerged as a strategic approach in combating biofilm-associated infections and biofouling problems. Enzymatic disruption represents a promising biocatalytic strategy that specifically degrades key EPS components, compromising biofilm integrity and facilitating the eradication of embedded cells [50]. This whitepaper provides an in-depth technical examination of four major enzyme classes—proteases, DNase, Dispersin B, and glycosidases—that have demonstrated significant efficacy in disrupting biofilm architecture by targeting distinct EPS constituents.
Composition and Function of the Biofilm EPS Matrix
The extracellular polymeric substance (EPS) matrix is far more than simple "slime"; it is a highly functional, organized microenvironment that maintains biofilm structure and function. The matrix is highly hydrated, consisting of up to 97% water, with the remaining portion composed of the biopolymeric components [49]. The composition varies significantly between bacterial species and environmental conditions, but generally includes polysaccharides, proteins, nucleic acids, lipids, and various other cellular detritus [49] [8].
Table 1: Major Components of Biofilm EPS Matrix and Their Functions
| EPS Component | Primary Function | Representative Producers |
|---|---|---|
| Exopolysaccharides (PNAG, alginate, cellulose, levan, Pel, Psl) | Structural integrity, adhesion, water retention, resistance barrier | Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli |
| Extracellular Proteins (adhesins, amyloids, enzymes) | Structural support, attachment, enzymatic activity, virulence | S. aureus, E. coli, Bacillus subtilis |
| Extracellular DNA (eDNA) | Structural connectivity, cation chelation, genetic information exchange | P. aeruginosa, S. aureus, various environmental isolates |
| Lipids and Surfactants | Hydrophobicity modulation, interface interactions | P. aeruginosa, Mycobacterium species |
The functional diversity of the EPS matrix extends beyond mere structural support. Different EPS components perform specialized roles that can be categorized as constructive, sorptive, active, surface-active, informative, redox-active, and nutritive [8]. For instance, exopolysaccharides like poly-N-acetylglucosamine (PNAG) and alginate provide structural scaffolding, while extracellular DNA (eDNA) has been recently appreciated as a crucial structural component that forms filamentous networks and facilitates cell-to-cell connectivity [8]. The matrix also serves as an external digestive system by retaining extracellular enzymes close to the cells that produced them, allowing for efficient nutrient acquisition [8].
Enzymatic Disruption Strategies: Mechanisms and Targets
Core Disruption Mechanisms
Enzymatic disruption of biofilms operates through the targeted degradation of specific EPS components, leading to the collapse of the structural matrix and release of embedded bacterial cells into the more vulnerable planktonic state. The strategic approach involves selecting enzymes that correspond to the predominant constituents of the target biofilm's EPS [49]. The diagram below illustrates the targeted degradation of major EPS components by specific enzyme classes.
Glycoside Hydrolases (Dispersin B and Other Polysaccharide-Degrading Enzymes)
Glycoside hydrolases (GHs) represent a diverse class of enzymes that catalyze the hydrolysis of glycosidic bonds in bacterial exopolysaccharides. These enzymes target the carbohydrate backbone of the EPS matrix, which is fundamental to biofilm structural integrity [51].
Dispersin B is a particularly well-characterized glycoside hydrolase produced by Aggregatibacter actinomycetemcomitans [51]. This enzyme specifically targets poly-N-acetylglucosamine (PNAG), also known as polysaccharide intercellular adhesin, which is a crucial polysaccharide found in the biofilm matrix of numerous Gram-positive and Gram-negative pathogens [51]. Dispersin B has demonstrated remarkable efficacy in inhibiting biofilm formation, detaching established biofilms, disaggregating bacterial clusters, and sensitizing biofilms to antimicrobial agents and host immune defenses [52].
Other significant glycoside hydrolases with demonstrated anti-biofilm activity include:
- α-Amylase: Targets glycogen-like polysaccharides in biofilms formed by pathogens like Streptococcus mutans [49].
- Alginate lyase: Specifically degrades alginate in P. aeruginosa biofilms, particularly relevant for cystic fibrosis lung infections [49].
- Cellulase: Disrupts cellulose components in EPS produced by P. aeruginosa, Salmonella enterica, and Enterococcus faecalis [49].
- Levan hydrolase: Degrades levan, a fructan polysaccharide produced by various bacterial species including S. aureus and P. aeruginosa [49].
Proteases
Proteases target the proteinaceous components of the EPS matrix, including structural proteins, adhesins, and functional enzymes trapped within the biofilm architecture [49]. These enzymes hydrolyze peptide bonds, disrupting the protein framework that contributes to biofilm stability and attachment.
Notable proteases with anti-biofilm activity include:
- Subtilisin: A serine protease effective against E. coli biofilms by degrading protein components in the EPS [49].
- Proteinase K: Demonstrates efficacy against S. aureus and P. aeruginosa biofilms through digestion of extracellular proteins [49].
- Lysostaphin: Specifically targets the cell wall of S. aureus species, showing potent activity against staphylococcal biofilms [49].
The effectiveness of proteases depends on the protein composition of the target biofilm, which varies significantly between bacterial species. For instance, proteases have shown particular efficiency in removing Bacillus biofilms, while being less effective against Pseudomonas fluorescens biofilms that possess predominantly polysaccharide matrices [53].
Deoxyribonucleases (DNases)
Deoxyribonucleases (DNases) represent a crucial class of biofilm-dispersing enzymes that target extracellular DNA (eDNA), a component now recognized as fundamental to biofilm structure in many bacterial species [8]. eDNA was once considered merely a remnant of lysed cells but is now known to play an active structural role in biofilm architecture.
DNase I has demonstrated effectiveness against biofilms formed by P. aeruginosa and S. aureus by degrading eDNA, leading to inhibition of biofilm formation and disruption of mature structures [49]. In P. aeruginosa biofilms, eDNA is organized in distinct grid-like patterns and functions as an intercellular connector that stabilizes the biofilm matrix [8]. Similarly, in S. aureus biofilms, genomic DNA released through cidA-controlled cell lysis serves as an important structural component during biofilm development [8].
The controlled application of DNases can effectively disrupt these eDNA networks, compromising biofilm integrity and facilitating the penetration of antimicrobial agents. However, the efficacy varies depending on the relative importance of eDNA to the structural integrity of the specific biofilm being targeted.
Quantitative Efficacy Data of Anti-Biofilm Enzymes
The anti-biofilm efficacy of enzymes has been quantitatively demonstrated across numerous in vitro and in vivo studies. The table below summarizes key efficacy data for major anti-biofilm enzymes against clinically relevant pathogens.
Table 2: Quantitative Efficacy of Anti-Biofilm Enzymes Against Bacterial Pathogens
| Enzyme | Target Biofilm | Mechanism of Action | Efficacy Measures | References |
|---|---|---|---|---|
| Dispersin B | S. aureus, S. epidermidis, E. coli, Y. pestis | Hydrolyzes poly-N-acetylglucosamine (PNAG) | Inhibits biofilm formation, detaches established biofilms, increases susceptibility to antibiotics | [49] [51] |
| DNase I | P. aeruginosa, S. aureus | Degrades extracellular DNA (eDNA) | Disrupts biofilm architecture, inhibits initial attachment | [49] |
| Proteinase K | S. aureus, P. aeruginosa | Digests extracellular proteins in biofilms | Reduces biofilm biomass, decreases adhesion strength | [49] |
| Subtilisin | E. coli | Serine protease disrupting protein components | Disrupts proteinaceous matrix components | [49] |
| α-Amylase | Streptococcus mutans | Degrades glycogen-like polysaccharides | Reduces biofilm stability and accumulation | [49] |
| Alginate lyase | P. aeruginosa | Breaks down alginate in biofilm structure | Enhances antibiotic penetration, reduces biofilm viscosity | [49] |
| Cellulase | P. aeruginosa, S. enterica, E. faecalis | Targets cellulose in EPS | Concentration-dependent reduction in biomass and CFU | [49] |
The efficacy of these enzymes is influenced by multiple factors including enzyme concentration, pH, temperature, and the specific composition of the target biofilm. For instance, cellulase demonstrated pH-dependent activity against P. aeruginosa biofilms, with greater efficacy observed at pH 5 compared to pH 7 [49]. Similarly, the effectiveness of enzyme cocktails depends on complementary activities that target multiple EPS components simultaneously.
Experimental Protocols for Enzymatic Biofilm Disruption
Standardized Biofilm Cultivation Protocol
To ensure reproducible results in enzymatic disruption studies, biofilms must be cultivated under standardized conditions:
Surface Preparation: Use relevant substrates such as glass coverslips, medical-grade stainless steel coupons, or plastic surfaces appropriate for high-throughput screening. Sterilize surfaces by autoclaving or UV irradiation before use.
Inoculum Preparation: Grow planktonic cultures of target organisms (e.g., P. aeruginosa, S. aureus) to mid-logarithmic phase (OD600 ≈ 0.5) in appropriate media (e.g., Tryptic Soy Broth, Luria-Bertani Broth).
Biofilm Establishment: Dilute bacterial suspension to approximately 10^6 CFU/mL in fresh media supplemented with 1% glucose to enhance biofilm formation. Add 2-3 mL to wells containing substrates and incubate under static or flow conditions for 24-72 hours at relevant temperature (typically 37°C for human pathogens).
Media Refreshment: Replace media every 24 hours for nutrient replenishment and removal of planktonic cells, promoting robust biofilm development.
Biofilm Validation: Verify biofilm formation by crystal violet staining, scanning electron microscopy, or confocal laser scanning microscopy before enzymatic treatment.
Enzymatic Disruption Assay Protocol
The following protocol details the standardized assessment of enzymatic biofilm disruption:
Enzyme Preparation: Prepare stock solutions of test enzymes (proteases, DNase, Dispersin B, glycosidases) in appropriate buffers at 10× the desired final concentration. Include controls with heat-inactivated enzymes and buffer-only treatments.
Biofilm Treatment: Carefully remove growth media from established biofilms and gently wash with phosphate-buffered saline (PBS) to remove loosely attached cells. Add enzyme solutions at predetermined concentrations (typically 0.1-100 μg/mL depending on enzyme) and incubate for 1-24 hours at optimal temperature for enzyme activity.
Disruption Assessment: Quantify biofilm disruption using multiple complementary methods:
- Biomass Reduction: Crystal violet staining with elution and absorbance measurement at 590 nm.
- Viability Assessment: Resuscitate dispersed cells in fresh media, serially dilute, and plate for colony-forming unit (CFU) enumeration.
- Structural Analysis: Confocal laser scanning microscopy with LIVE/DEAD staining (SYTO9/propidium iodide) to visualize architectural changes and cellular viability.
- Matrix Component Analysis: Specific staining for polysaccharides (calcofluor white), proteins (SYPRO Ruby), or eDNA (TOTO-1) to assess degradation of specific EPS components.
Synergy Testing: For combination therapies, apply enzymes sequentially or simultaneously with conventional antibiotics (e.g., tobramycin, ciprofloxacin) to assess enhanced efficacy.
The experimental workflow for assessing enzymatic disruption of biofilms is systematically presented in the following diagram:
Optimization Considerations for Enzymatic Treatments
Several critical factors require optimization for successful enzymatic biofilm disruption:
Enzyme Cocktail Formulation: Combined enzymes targeting multiple EPS components (e.g., DNase + Dispersin B + protease) typically show superior efficacy compared to single enzymes [49].
pH and Temperature Profiles: Each enzyme exhibits optimal activity within specific pH and temperature ranges that must be aligned with application conditions.
Enzyme Stability: Many commercial enzymes are inactivated by standard sanitation steps used in industrial settings, necessitating careful selection of thermostable variants [53].
Delivery Systems: For in vivo applications, controlled-release systems or nanoparticle-based delivery may enhance enzyme stability and prolong activity at the biofilm site.
The Researcher's Toolkit: Essential Reagents and Materials
Successful investigation of enzymatic biofilm disruption requires access to specific reagents, enzymes, and analytical tools. The following table catalogues essential components of the biofilm researcher's toolkit.
Table 3: Essential Research Reagents for Enzymatic Biofilm Studies
| Reagent Category | Specific Examples | Function/Application | Key Considerations |
|---|---|---|---|
| Enzymes | Dispersin B, DNase I, Proteinase K, Subtilisin, Alginate lyase, α-Amylase | EPS matrix degradation, biofilm disruption | Purity, specific activity, optimal pH/temperature range |
| Bacterial Strains | P. aeruginosa (PAO1, PA14), S. aureus (ATCC 25923, biofilm-forming clinical isolates), E. coli (K-12, UTI89) | Biofilm formation models | Known EPS composition, genetic tractability, clinical relevance |
| Growth Media | Tryptic Soy Broth (TSB), Luria-Bertani (LB) Broth, M63 minimal media, Brain Heart Infusion (BHI) | Biofilm cultivation and maintenance | Composition affects EPS production and biofilm architecture |
| Staining Reagents | Crystal violet, SYTO9/propidium iodide, calcofluor white, SYPRO Ruby, TOTO-1 | Biofilm visualization and quantification | Specificity for biomass, viability, or matrix components |
| Surface Materials | Medical-grade stainless steel coupons, glass coverslips, polyvinyl chloride (PVC) plates, catheter fragments | Biofilm substrate | Surface properties significantly influence attachment |
| Analytical Tools | Microtiter plates, flow cells, confocal microscopy, scanning electron microscopy, qPCR equipment | Biofilm assessment and characterization | Resolution, throughput, and quantitative capabilities |
The selection of appropriate reagents should align with the specific research objectives, whether focused on basic mechanisms of biofilm biology, preclinical therapeutic development, or industrial biofouling control. For drug development applications, particular attention should be paid to enzyme sourcing, purity, and regulatory compliance for potential therapeutic use.
Enzymatic disruption of biofilms represents a promising targeted approach against structured microbial communities that have traditionally resisted conventional antimicrobial therapies. The strategic deployment of proteases, DNases, Dispersin B, and glycosidases corresponds directly to the multi-component nature of the EPS matrix, enabling precise targeting of the structural foundations that maintain biofilm integrity.
Future research directions should focus on several key areas:
Enzyme Engineering: Development of engineered enzymes with enhanced stability, specificity, and synergistic activities through protein engineering approaches.
Delivery System Innovation: Design of advanced delivery platforms that protect enzymes from inactivation and facilitate penetration into deep biofilm layers.
Combination Therapy Optimization: Systematic evaluation of enzymatic pre-treatment strategies to enhance the efficacy of conventional antibiotics and host immune responses.
Diagnostic Integration: Development of point-of-care diagnostics to identify the specific EPS composition of clinical biofilms, enabling personalized enzyme cocktail selection.
Regulatory Pathway Establishment: Creation of clear regulatory guidelines for enzyme-based anti-biofilm therapeutics, addressing unique considerations for biological anti-infectives.
The continued elucidation of EPS structure-function relationships and the refinement of enzymatic tools to exploit these insights will undoubtedly yield powerful new approaches to combat biofilm-associated challenges across clinical, industrial, and environmental settings.
The extracellular polymeric substance (EPS) matrix represents a fundamental architectural component of microbial biofilms, serving as both a physical scaffold and a protective barrier for the embedded microbial communities. This matrix is a complex hydrogel comprised of polysaccharides, proteins, nucleic acids, and lipids, which self-assembles into a three-dimensional network that defines the biofilm's structural and functional properties [1] [54]. Within this network, divalent cations including calcium (Ca²⁺), magnesium (Mg²⁺), and zinc (Zn²⁺) serve as critical ionic bridges that cross-link anionic functional groups present on EPS polymers, particularly carboxyl and phosphate groups [55]. This cation-mediated cross-linking confers mechanical stability to the biofilm architecture, enhances resistance to environmental stresses, and reduces permeability to antimicrobial agents.
Chelating agents such as ethylenediaminetetraacetic acid (EDTA) specifically target these cation-mediated interactions. As a hexadentate ligand with high binding affinity for divalent cations, EDTA sequesters these ionic bridges, leading to disassembly of the EPS matrix and subsequent biofilm sensitization [55]. The strategic disruption of these coordination bonds represents a promising anti-biofilm strategy that bypasses conventional antimicrobial resistance mechanisms by targeting the physical integrity of the biofilm rather than microbial viability itself. This approach is particularly valuable against recalcitrant biofilm-forming pathogens such as Pseudomonas aeruginosa and ESKAPE pathogens (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species) [1].
Molecular Mechanisms of EDTA Action on Biofilm Matrix
Chelation Chemistry and Cation Selectivity
EDTA operates through a well-defined coordination chemistry wherein its four carboxylate and two amine groups form an octahedral complex with divalent metal ions [55]. This chelation mechanism is highly effective for sequestering cations that serve as essential inter-polymer bridges in the EPS matrix. The binding constants of EDTA for relevant divalent cations follow the order: Fe²⁺ > Zn²⁺ > Ca²⁺ > Mg²⁺, reflecting varying affinities that influence its efficacy in different biofilm contexts [55].
The sequestered cations perform distinct structural roles within the biofilm matrix. Calcium ions facilitate particularly strong bridging between anionic polysaccharide chains, forming rigid gel structures that enhance mechanical stability. Magnesium ions often contribute to more dynamic, reversible cross-links that allow for matrix adaptation, while zinc ions may play specialized roles in protein folding and stabilization within the EPS [55]. The removal of these cations through chelation disrupts the ionic cross-linking essential for matrix integrity.
Matrix Destabilization and Enhanced Antimicrobial Penetration
The primary consequence of cation chelation is the collapse of the EPS scaffolding, which manifests as several observable phenomena:
- Reduced matrix cohesion: The loss of ionic bridges decreases polymer cross-linking, leading to increased matrix solubility and disintegration [55].
- Altered physical properties: Biofilms exhibit decreased viscoelasticity and mechanical strength, facilitating physical removal.
- Enhanced permeability: The disrupted matrix architecture allows improved penetration of antimicrobial agents to previously inaccessible microbial cells [56].
Beyond these physical effects, EDTA exhibits direct antimicrobial activity through mechanisms related to cation deprivation. By chelating cations essential for outer membrane stability in Gram-negative bacteria, EDTA increases membrane permeability and sensitizes bacteria to other antimicrobials [55] [56]. This dual mechanism—simultaneously disrupting the extracellular matrix and compromising microbial membrane integrity—makes EDTA particularly effective against established, mature biofilms that are typically refractory to conventional antibiotics.
Experimental Evidence and Efficacy Data
Quantitative Assessment of EDTA Efficacy Against Planktonic and Biofilm Cells
Table 1: EDTA efficacy against colistin-resistant K. pneumoniae in planktonic and biofilm states [56]
| Bacterial State | EDTA Concentration | Exposure Time | Efficacy Outcome | Notes |
|---|---|---|---|---|
| Planktonic cells | 3-24 mg/mL | 2 hours | Complete growth inhibition | ≥3 log reduction in CFU |
| Mature biofilms | 12 mg/mL | 24 hours | Significant biofilm reduction | Combined with colistin (0.5-1 μg/mL) |
| Planktonic cells | 1 mM | 24 hours | Growth suppression | Strain-dependent variability [57] |
| Biofilms (P. aeruginosa) | 1-10 mM | 48 hours | Delayed peak biofilm formation | No significant reduction in final biomass [57] |
Table 2: Minimum Biofilm Eradication Concentration (MBEC) of EDTA and combination therapy [56]
| Treatment | MBEC Value | Fold Change | Clinical Significance |
|---|---|---|---|
| Colistin alone | 8-1000× MIC | - | High resistance in biofilms |
| EDTA alone | 2-4× planktonic MIC | - | Moderate efficacy |
| Colistin + EDTA | 0.5-1 μg/mL + 12 mg/mL | Complete eradication | Synergistic effect (FICI ≤0.5) |
Research demonstrates that EDTA's efficacy varies significantly between planktonic and biofilm states, with biofilms generally requiring higher concentrations for effective disruption. Against colistin-resistant Klebsiella pneumoniae, EDTA at 12 mg/mL combined with colistin (0.5-1 μg/mL) achieved complete eradication of mature biofilms, while exhibiting only partial activity when used alone [56]. This highlights the importance of combination approaches for recalcitrant biofilm infections.
Notably, EDTA exhibits strain-dependent effects on biofilms. While it consistently inhibits planktonic growth across strains, its impact on biofilm biomass varies. Some strains of P. aeruginosa show delayed biofilm development with EDTA treatment but ultimately achieve similar or even increased biomass compared to untreated controls [57]. This paradoxical effect underscores the complexity of biofilm biology and the potential for adaptive responses to cation limitation.
In Vivo Efficacy in Catheter-Related Infections
Table 3: In vivo efficacy of colistin-EDTA combination in murine catheter infection model [56]
| Parameter | Colistin Alone | EDTA Alone | Colistin-EDTA Combination | Statistical Significance |
|---|---|---|---|---|
| Survival rate (Day 7) | <20% | ~40% | 100% | p<0.0001 |
| Bacterial load in organs | High | Moderate | Significant reduction | p<0.05 |
| Serum creatinine | Elevated | Moderately elevated | Near normal | p<0.05 |
| Biofilm biovolume | Moderate reduction | Moderate reduction | Significant eradication | p<0.001 |
In a murine model of catheter-related biofilm infection, the colistin-EDTA combination demonstrated superior efficacy compared to monotherapies. The combination not only achieved 100% survival but also significantly reduced bacterial loads in internal organs and prevented biofilm recurrence [56]. Importantly, the combination therapy resulted in improved renal function parameters (serum creatinine), suggesting that EDTA may mitigate colistin-induced nephrotoxicity while simultaneously enhancing its anti-biofilm activity.
The therapeutic success of this approach in vivo validates the strategy of targeting cation bridging as a viable anti-biofilm intervention, particularly for device-related infections where biofilm formation on abiotic surfaces poses significant clinical challenges.
Experimental Methodologies for Evaluating EDTA Efficacy
Planktonic and Biofilm Susceptibility Testing
Bacterial Strains and Culture Conditions
- Clinical isolates of target pathogens (e.g., colistin-resistant K. pneumoniae, P. aeruginosa strains) should be stored at -80°C in 20% glycerol [56] [57].
- Prior to experiments, streak strains onto Luria-Bertani (LB) agar plates and incubate overnight at 37°C.
- Inoculate single colonies into LB broth and incubate with shaking (225 rpm) at 37°C for 16-18 hours to achieve mid-log phase growth [57].
Planktonic Susceptibility Testing
- Adjust bacterial suspensions to optical density (OD₆₀₀) of 0.3 using sterile broth.
- Prepare 1:1000 dilution in appropriate media (e.g., LB or M63 minimal media).
- Add EDTA solutions across concentration range (e.g., 1-24 mg/mL or 1-10 mM).
- Incubate at 37°C with shaking (225 rpm).
- Monitor growth kinetics by measuring OD₆₀₀ at regular intervals (e.g., every 2 hours).
- Determine minimum inhibitory concentration (MIC) as the lowest concentration showing no visible growth [56] [57].
Biofilm Susceptibility Testing (Microtiter Plate Assay)
- Prepare bacterial inoculum as above and dilute 1:1000 in appropriate biofilm-promoting media (e.g., M63 minimal media supplemented with 1 mM MgCl₂, 0.2% D-glucose, and 0.5% casamino acids) [57].
- Dispense 100 μL aliquots into 96-well polyvinyl chloride microtiter plates.
- Incubate stationary at 30°C for 24-48 hours to allow biofilm formation.
- Carefully remove planktonic cells by washing plates three times with tap water or phosphate-buffered saline.
- Add fresh media containing EDTA at desired concentrations.
- Incubate for additional 6-72 hours depending on experimental design.
- Quantify biofilm biomass using crystal violet staining:
- Wash plates gently three times with tap water
- Air dry completely
- Add 125 μL of 0.1% crystal violet, incubate 15 minutes at room temperature
- Wash five times with tap water to remove unbound dye
- Elute bound dye with 200 μL of 33% glacial acetic acid
- Measure absorbance at 595 nm using microplate reader [57]
Advanced Analytical Methods for Matrix Characterization
EPS Extraction and Composition Analysis
- Harvest biofilm cultures after EDTA treatment and centrifuge (10,000 × g, 10 minutes, 4°C).
- Extract EPS using cation exchange resin (e.g., Amberlite HPR1100) [4].
- Analyze EPS components:
- Total carbohydrates: Hydrolyze with 0.75 M H₂SO₄, quantify using bicinchoninic acid (BCA) microplate assay at 562 nm [4].
- Total proteins: Use Lowry assay microplate method, measure absorbance at 750 nm [4].
- Extracellular DNA: Extract with phenol:chloroform:isoamyl alcohol (24:25:1 v/v/v), quantify fluorometrically [4].
- Amino sugars: Analyze muramic acid, mannosamine, galactosamine, and glucosamine via chromatographic methods [4].
Confocal Laser Scanning Microscopy (CLSM) for 3D Architecture
- Grow biofilms on appropriate surfaces (e.g., catheter segments, glass coverslips).
- Treat with EDTA solutions for predetermined times.
- Stain with live/dead bacterial viability kit (e.g., SYTO 9/propidium iodide).
- Image using CLSM with appropriate laser settings and filters.
- Reconstruct 3D architecture using image analysis software (e.g., Imaris, ImageJ) [56].
- Quantify biovolume, thickness, and live/dead ratios.
Research Reagent Solutions for EDTA-Biofilm Studies
Table 4: Essential research reagents for studying EDTA effects on biofilms
| Reagent/Category | Specific Examples | Function/Application | Experimental Notes |
|---|---|---|---|
| Chelating Agents | Tetrasodium EDTA (tEDTA), Disodium EDTA | Primary anti-biofilm agent | tEDTA shows enhanced antibiofilm activity at alkaline pH [55] |
| Culture Media | Luria-Bertani (LB) broth, M63 minimal media | Planktonic and biofilm growth | M63 minimal media with supplements enhances reproducible biofilm formation [57] |
| Detection Assays | Crystal violet, SYTO 9/propidium iodide | Biomass quantification and viability staining | Crystal violet measures total biomass; live/dead staining assesses viability [56] [57] |
| EPS Analysis | Cation exchange resin (Amberlite) | EPS extraction from biofilms | Enables compositional analysis of matrix components [4] |
| Microtiter Plates | Polyvinyl chloride (PVC), Polystyrene | Biofilm growth substrate | PVC enhances bacterial attachment compared to polystyrene [57] |
| Enzymatic Assays | BCA assay, Lowry assay | Carbohydrate and protein quantification | Essential for EPS composition analysis after EDTA treatment [4] |
Visualization of EDTA Mechanism and Experimental Workflow
Molecular Mechanism of EDTA in Biofilm Disruption
Experimental Workflow for EDTA Biofilm Assays
The strategic disruption of divalent cation bridges represents a mechanistically rational approach to biofilm control that circumvents many conventional resistance mechanisms. EDTA and related chelating agents effectively target the structural integrity of the EPS matrix by sequestering essential cross-linking cations, particularly calcium and magnesium. The experimental evidence demonstrates that while EDTA monotherapy shows variable efficacy, its combination with conventional antimicrobials achieves potent synergistic effects against even recalcitrant, multidrug-resistant biofilm infections.
Future research directions should focus on optimizing chelator design for enhanced biofilm penetration, developing controlled-release formulations for sustained anti-biofilm activity, and exploring combination therapies with novel antimicrobial agents. Additionally, further investigation into the differential effects of EDTA across bacterial species and biofilm maturation stages will refine its clinical application. As biofilm-associated infections continue to pose significant challenges across medical devices, chronic wounds, and institutional settings, cation chelation strategies offer a promising non-biocidal approach to biofilm management that complements traditional antimicrobial therapies.
Bacterial biofilms are responsible for up to 80% of all chronic and recurrent infections, posing a serious global health threat due to their profound resistance to antimicrobial therapies [58] [59]. This resistance is largely conferred by the extracellular polymeric substance (EPS), a complex matrix that surrounds microbial cells and provides chemical and mechanical support to the bacterial community [58] [60]. The EPS matrix, comprising polysaccharides, proteins, extracellular DNA (eDNA), and lipids, creates a formidable barrier that restricts antibiotic penetration through diffusion limitation, electrostatic interactions, and enzymatic inactivation of drug molecules [58] [61]. Within this protective environment, bacterial cells can become up to 1,000 times less susceptible to antibiotics than their planktonic counterparts [58] [62].
Nanoparticles (NPs) have emerged as promising drug-carriers capable of enhancing the delivery of antimicrobial agents into biofilms, offering a potential solution to the crisis of antimicrobial resistance [58] [61]. Their unique physicochemical properties—including small size, large surface area, and flexible surface chemistry—enable them to navigate the complex EPS architecture and improve therapeutic profiles of existing antibiotics [58] [59]. This technical guide explores how functionalized nanoparticles can overcome EPS-mediated resistance through enhanced penetration and efficacy, providing researchers and drug development professionals with methodologies and mechanistic insights for developing advanced anti-biofilm strategies.
Nanoparticle-EPS Interactions: Fundamental Mechanisms
The effectiveness of functionalized nanoparticles against biofilms depends on their ability to overcome the physicochemical barriers presented by the EPS matrix. The biofilm microenvironment is typically characterized by negative surface charge, hydrophobicity, acidity, and an abundance of enzymes—all of which contribute to its resistance to foreign chemicals [59]. Different bacterial strains produce distinct EPS compositions; for instance, methicillin-susceptible Staphylococcus aureus (MSSA) typically forms biofilms embedded in an extracellular polysaccharide matrix, while methicillin-resistant S. aureus (MRSA) biofilms comprise predominantly of surface proteins and eDNA [58].
The strategic design of nanoparticles can leverage several key mechanisms to enhance biofilm penetration and eradication:
- Electrostatic interactions: Positively charged nanoparticles exhibit enhanced adherence to negatively charged components of the EPS, such as eDNA and certain polysaccharides [58] [59]. However, this strong attraction may also limit deeper penetration, creating a trade-off that must be carefully balanced through surface engineering.
- Size-dependent penetration: The nanoscale dimensions of NPs (typically 1-1000 nm) enable superior diffusion through the porous EPS matrix compared to conventional antibiotic molecules [59].
- Biofilm matrix degradation: Certain functionalized nanoparticles can actively degrade structural components of the EPS. For example, dextran-based Janus nanoparticles have demonstrated exceptional ability to disperse biofilms by interfering with EPS integrity [63].
- Enhanced drug permeability: Nanoparticles protect encapsulated antimicrobial agents from inactivation within the biofilm matrix and can provide more uniform distribution through the biofilm architecture compared to free antibiotics [58] [61].
The following diagram illustrates the multi-faceted interactions between functionalized nanoparticles and the EPS barrier:
Diagram: Multifunctional nanoparticle interactions with EPS barriers. NP properties are engineered to overcome specific EPS resistance mechanisms.
Strategic Functionalization for Enhanced Efficacy
Surface Chemistry Engineering
The surface functionalization of nanoparticles plays a pivotal role in determining their interactions with biofilm components. A systematic study on mesoporous silica nanoparticles (MSNs) with different surface functionalization demonstrated how surface chemistry affects both drug loading and antibacterial efficacy [58]. The research compared bare (MSN-B), amine-functionalized (MSN-D), carboxyl-functionalized (MSN-C), and aromatic-functionalized (MSN-A) nanoparticles, revealing that the two negatively charged MSNs (MSN-B and MSN-C) showed higher vancomycin loading capacity compared to their positively charged counterparts [58]. However, cellular binding efficiency—which correlated with reduced viability of both MSSA and MRSA biofilm cells—was achieved with positively charged functionalizations at low concentrations (0.25 mg mL⁻¹), enabling high local antibiotic concentrations surrounding bacterial cells while maintaining low systemic MSN concentrations [58].
Advanced Nanoplatform Architectures
Recent innovations in nanoparticle design have produced sophisticated architectures with enhanced anti-biofilm capabilities:
Janus nanoparticles represent a breakthrough in asymmetric nanostructure design. Janus dextran-bismuth selenide (Dex-BSe) nanoparticles exploit synergistic effects by combining the biofilm-targeting capability of dextran with the photothermal properties of bismuth selenide [63]. The dextran domain targets biofilm EPS with high specificity and enhances penetration, while the BSe component enables near-infrared (NIR) light-activated photothermal eradication of biofilms [63]. Interestingly, these Janus nanoparticles demonstrated enhanced dispersal of biofilms over time even without activation, and the self-propelled active motion induced by their asymmetric structure under NIR irradiation further enhanced penetration and photothermal killing efficacy [63].
Ultrasound-activated nanoplatforms offer another innovative approach for biofilm eradication. Researchers at the University of Oxford developed antibiotic-loaded nanoparticles that, when activated by ultrasound, rapidly vaporize to physically disrupt biofilms while simultaneously releasing drugs directly at the infection site [62]. This combination strategy reduced the antibiotic concentration required to prevent bacterial growth by more than 10-fold for planktonic bacteria and by more than 40-fold for biofilm infections, achieving 100% bacterial elimination at clinically feasible doses [62].
Table 1: Efficacy of Functionalized Nanoparticles Against Biofilm-Related Infections
| Nanoparticle Type | Functionalization | Target Pathogen | Key Findings | Reference |
|---|---|---|---|---|
| Mesoporous Silica Nanoparticles | Amine, Carboxyl, Aromatic | MRSA, MSSA | Positively charged MSNs enhanced cellular binding; 0.25 mg mL⁻¹ concentration reduced biofilm viability | [58] |
| Janus Dex-BSe | Dextran-Bismuth Selenide | S. aureus, MRSA | Achieved biofilm dispersion & NIR-activated eradication; self-propelled motion enhanced penetration | [63] |
| Ultrasound-activated Nanobubbles | Antibiotic-loaded | E. coli, MRSA | Reduced antibiotic concentration by 40-fold; eliminated 100% of biofilm bacteria | [62] |
| Polycaprolactone Nanospheres | Imipenem-loaded | CRKP | 8-fold reduction in MIC; significant biofilm inhibition; suppressed resistance genes | [64] |
Experimental Protocols and Methodologies
Synthesis of Functionalized Mesoporous Silica Nanoparticles
The synthesis of MSNs with controlled surface functionalization can be achieved through an adapted Stöber method as described in the literature [58]:
Materials:
- Hexadecyltrimethylammonium bromide (CTAB, 98%) as template
- Tetraethyl orthosilicate (TEOS, 99%) as silica source
- (3-aminopropyl)triethoxysilane (APTES, 99%) for amine functionalization
- Triethanolamine (TEA, 99%) as catalyst
- Succinic anhydride for carboxyl functionalization
- Benzoic acid for aromatic functionalization
Procedure:
- Base MSN Synthesis: Dissolve CTAB in deionized water and add TEA under vigorous stirring. Heat the mixture to 80°C before adding TEOS dropwise. Continue stirring for 2 hours to form bare MSNs (MSN-B).
- Amination (MSN-D): Disperse 50 mg of MSN-B in 4.6 mL of absolute ethanol in an ultrasonic bath. Add 200 µL deionized water, 100 µL acetic acid, and 100 µL DETA. Stir the reaction mixture at room temperature for 1 hour. Recover the product by centrifugation (9,000 rpm, 15 minutes) and wash three times with ethanol.
- Carboxyl Functionalization (MSN-C): Suspend dried MSN-D in DMF (6 mL of 2 mg mL⁻¹). Add to a solution of succinic anhydride in DMF (50 mL of 10 mg mL⁻¹). Stir vigorously overnight at room temperature. Recover the product by centrifugation and wash three times with ethanol.
- Aromatic Functionalization (MSN-A): Mix a suspension of MSN-D in water (9 mL of 3.34 mg mL⁻¹) with aqueous solutions of benzoic acid (20 mL of 4.71 mg mL⁻¹), NHS (2 mL of 114.7 mg mL⁻¹), and EDC (2 mL of 150.8 mg mL⁻¹). Stir at room temperature overnight. Recover the product by extensive washing with ethanol via centrifugation.
Characterization:
- Zeta potential measurements in water (1 mg mL⁻¹) using a Zetasizer Nano-ZS apparatus
- TEM analysis using samples dispersed in ethanol and deposited on carbon-coated copper grids
- Drug loading efficiency determination through UV-Vis spectroscopy or HPLC
Synthesis of Janus Dextran-Bismuth Selenide Nanoparticles
The fabrication of asymmetric Janus nanoparticles requires a nonsolvent-aided counterion complexation strategy [63]:
Materials:
- Poly(vinylpyrrolidone) (PVP) as stabilizer
- Aminated dextran (Dex-NH₂) prepared by reacting hydroxyl groups of dextran with ethylenediamine
- Bismuth selenide precursors (Bi₂Se₃)
- Ethylenediaminetetraacetic acid (EDTA) as counterion
- Ethanol as nonsolvent
Procedure:
- BSe Nanosheet Synthesis: Prepare BSe nanosheets via a facile solution method with PVP assistance. Characterize by TEM to confirm nearly hexagonal morphology with average size of ~90 nm.
- Dex-NH₂ Preparation: React activated hydroxyl groups of dextran with amino groups of ethylenediamine. Confirm successful conjugation by ¹H NMR spectroscopy.
- Janus Nanoparticle Assembly: Employ a nonsolvent-aided counterion complexation strategy where ethanol induces counterion condensation of cationic Dex-NH₂, anionic EDTA, and negatively charged BSe nanosheets.
- Purification: Recover monodisperse Janus Dex-BSe nanoparticles by centrifugation and wash thoroughly to remove unreacted components.
Characterization:
- TEM imaging to confirm Janus structure with BSe nanosheets distributed on the outer edge of dextran nanospheres
- Elemental mapping to verify asymmetric distribution of C, O, N (dextran domain) and Bi, Se (BSe domain)
- Dynamic light scattering for size distribution and zeta potential measurements
- Thermogravimetric analysis to determine polysaccharide weight ratio (~60%)
- Photothermal conversion efficiency calculation (~31.5%) through heating/cooling curves
Table 2: Research Reagent Solutions for Nanoparticle Synthesis and Evaluation
| Reagent/Category | Specific Examples | Function in Research | Application Context |
|---|---|---|---|
| Silica Precursors | Tetraethyl orthosilicate (TEOS) | Forms silica framework of nanoparticles | MSN synthesis [58] |
| Surface Modifiers | APTES, DETA, succinic anhydride | Imparts specific surface charge/functionality | Surface functionalization of MSNs [58] |
| Polymeric Carriers | Polycaprolactone (PCL), dextran | Biocompatible nanoparticle matrix | Drug encapsulation & sustained release [64] |
| Photothermal Agents | Bismuth selenide nanosheets | Converts light to thermal energy | NIR-activated biofilm eradication [63] |
| Characterization Tools | Zetasizer, TEM, FTIR | Determines size, charge, morphology, composition | Nanoparticle characterization [58] [64] |
Quantitative Efficacy Assessment
The evaluation of functionalized nanoparticles against biofilms requires comprehensive assessment through standardized quantitative measures:
Biofilm Cultivation and Treatment:
- Grow biofilms in appropriate media (e.g., tryptic soy broth for S. aureus) for 24-48 hours to establish mature structures
- Treat biofilms with nanoparticle formulations at varying concentrations
- Include controls: untreated biofilm, free antibiotic treatment, empty nanoparticles
Assessment Methods:
- Viability Assays: Use metabolic indicators like MTT assay or ATP measurement to quantify viable cells remaining post-treatment [58]
- Biomass Quantification: Employ crystal violet staining to assess total biofilm biomass
- CFU Enumeration: Perform colony-forming unit counts after biofilm disruption to determine bactericidal activity
- Confocal Microscopy: Visualize biofilm architecture and nanoparticle penetration using fluorescent tags
- Gene Expression Analysis: Apply RNA-sequencing transcriptomics to elucidate molecular mechanisms of biofilm dispersion [63]
The following workflow diagram outlines a standardized experimental approach for evaluating anti-biofilm efficacy:
Diagram: Standardized workflow for evaluating anti-biofilm efficacy of functionalized nanoparticles, from synthesis to comprehensive assessment.
Emerging Applications and Future Directions
The application of functionalized nanoparticles against biofilm-associated infections continues to evolve with several promising developments:
Combination Strategies integrating multiple modalities have demonstrated remarkable efficacy. The combination of photothermal nanoparticles with ultrasound activation represents a particularly powerful approach, achieving synergistic effects that significantly reduce required antibiotic doses while improving biofilm eradication [62] [63]. These multimodal systems physically disrupt the EPS matrix while simultaneously delivering high local concentrations of antimicrobial agents directly to the compromised biofilm structure.
Targeted gene suppression approaches using nanotechnology have shown potential in addressing antimicrobial resistance at its fundamental level. Polycaprolactone nanospheres loaded with imipenem demonstrated not only enhanced antibacterial activity against carbapenem-resistant Klebsiella pneumoniae (CRKP) but also marked suppression of key resistance genes [64]. This dual action—direct bacterial killing coupled with resistance gene modulation—represents a promising strategy for combating multidrug-resistant pathogens.
Future research directions should focus on developing stimuli-responsive nanoplatforms that react to specific biofilm microenvironment cues such as pH, enzyme presence, or metabolic products [59]. Additionally, the exploration of multi-targeted therapies that simultaneously address multiple EPS components and bacterial viability factors holds promise for overcoming the adaptability of biofilms. As these technologies advance, considerations of nanotoxicity, pharmacokinetic optimization, and scalable manufacturing must remain priorities to facilitate clinical translation.
Functionalized nanoparticles represent a paradigm shift in addressing the formidable challenge of biofilm-mediated antimicrobial resistance. By strategically engineering surface chemistry, architecture, and functionality, researchers can design nanoscale systems capable of penetrating the protective EPS matrix, targeting persistent bacterial populations, and delivering therapeutic payloads with unprecedented precision. The continued refinement of these approaches, coupled with advanced assessment methodologies and combination strategies, promises to unlock new possibilities for treating chronic infections and combating the global crisis of antimicrobial resistance. As this field progresses, the integration of fundamental EPS research with innovative nanomaterial design will be essential for developing the next generation of anti-biofilm therapeutics.
Extracellular Polymeric Substances (EPS) are the fundamental architectural matrix that defines the biofilm mode of life. This complex, self-produced hydrogel consists of polysaccharides, proteins, extracellular DNA (e-DNA), and lipids that encase microbial communities, providing structural integrity and protection [1]. The EPS matrix constitutes a biological barrier that significantly impedes antimicrobial penetration and confers remarkable resistance to environmental stresses, complicating treatment of persistent infections, particularly those involving ESKAPE pathogens (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species) [1]. Understanding EPS functionality requires sophisticated model systems that accurately replicate its development and behavior under various conditions.
Research into EPS structure and function spans multiple experimental approaches, from simple static well plates to complex flowing reactor systems. Each model offers distinct advantages for investigating specific aspects of biofilm biology, yet all must address the fundamental challenge of recreating environments where EPS production and organization can occur naturally. The choice of model system profoundly influences research outcomes, particularly when studying how EPS mediates biofilm resistance to antimicrobial agents and mechanical disruption [65]. This guide provides a comprehensive technical overview of available model systems, their applications, and methodologies to assist researchers in selecting appropriate platforms for EPS-focused investigations.
Biofilm Model Systems: Technical Specifications and Applications
Comparative Analysis of Biofilm Model Systems
Table 1: Technical Specifications and Applications of Biofilm Model Systems
| Model System | Shear Stress Control | Throughput Capacity | EPS Study Applications | Key Advantages | Technical Limitations |
|---|---|---|---|---|---|
| Well Plates (Static) | None or very low | High (96-well standard) | Initial attachment studies, EPS composition analysis, antimicrobial efficacy screening | Simplicity, cost-effectiveness, compatibility with high-throughput screening | Limited physiological relevance, no fluid dynamics, potential oxygen gradient formation |
| CDC Biofilm Reactor | Defined, reproducible shear (avg. 0.365 ± 0.074 Pa at 125 RPM) [66] | Medium (24 coupons per reactor) | EPS formation under controlled flow, shear stress effects on EPS composition, standardized antimicrobial testing | Reproducible, turbulent flow patterns, referenced in ASTM and EPA guidelines [66] | Requires larger culture volumes, more complex setup than static systems |
| Bead Biofilm Reactor | Low-shear environment (40 RPM rotation) [67] | High (50 beads per reactor) | EPS production on particulate substrates, translational studies from in vitro to in vivo | Easily manipulable substrates, models environmental contaminants, improved translatability | Manual broth exchange required, batch-style operation |
| Living Tissue Slices | Varies with tissue source | Low | Host-pathogen interactions, EPS role in tissue adhesion and infection persistence | Preservation of native tissue architecture, clinical relevance | Technically challenging, limited availability, shorter viability periods |
| Tubular Structure Models | Variable, depends on flow rate | Low | Catheter-related biofilm studies, EPS role in medical device fouling | Clinical relevance for medical device infections, customizable flow conditions | Specialized equipment required, challenging for visualization |
Advanced and Specialized Model Systems
Beyond conventional approaches, several advanced systems offer unique capabilities for EPS research. Living myocardial tissue slices (150-400 µm thickness) preserve native tissue architecture and enable investigation of biofilm formation in clinically relevant environments, though their application for EPS-specific studies requires further validation [68]. Tubular structure models, particularly those utilizing silicone tubes, replicate medical device environments where EPS-mediated biofilms pose significant clinical challenges, allowing researchers to study biofilm development under dynamic flow conditions similar to those in catheters and implants [69].
Emerging technologies combine physical disruption methods with these model systems to investigate EPS integrity and function. For instance, shockwave treatment applied to tubular biofilm models has demonstrated significant disruption of EPS structure, removing up to 97.5% of biofilm surface area and enhancing antibiotic efficacy against P. aeruginosa biofilms by 40% compared to untreated controls [69]. This approach highlights how model systems can be leveraged to develop EPS-targeting interventions.
Experimental Methodologies for EPS Research
Standardized Protocol for CDC Biofilm Reactor Operation
The CDC biofilm reactor represents a gold standard for robust, reproducible biofilm growth under controlled hydrodynamic conditions. The following protocol ensures consistent EPS formation for experimental analysis:
Reactor Setup and Sterilization
- Assemble the CDC reactor according to manufacturer specifications, ensuring all coupons are properly seated in their respective rods [66].
- Sterilize the complete assembly using autoclaving at 121°C for 30 minutes or chemical sterilization appropriate for the study parameters.
- Aseptically transfer the reactor to the growth environment and connect to the nutrient feed system if operating in continuous mode.
Inoculation and Growth Conditions
- Prepare inoculum from freshly cultured bacteria adjusted to approximately 1.5 × 10^6 CFU/mL in appropriate growth medium [67].
- For P. aeruginosa and similar organisms, use tryptic soy broth (TSB) or brain heart infusion (BHI) broth as growth medium.
- Introduce inoculum to achieve approximately 75% reactor volume (approximately 750 mL in a 1 L vessel).
- Set baffle rotation to 125 RPM, generating an average shear stress of 0.365 ± 0.074 Pa across coupon surfaces [66].
- Maintain temperature at 35-37°C for most pathogenic species, monitoring throughout growth period.
Biofilm Growth and Harvesting
- Allow 48-72 hours for mature biofilm development with continuous nutrient availability.
- For continuous operation, maintain dilution rate to prevent planktonic cell overgrowth while supporting biofilm development.
- Terminate experiment at predetermined time points and aseptically remove coupons for analysis.
- For EPS extraction, carefully scrape biofilm from coupon surfaces using sterile implements and suspend in appropriate buffer for downstream analysis.
Bead Biofilm Reactor Methodology for Particulate Substrates
The bead biofilm reactor provides a unique platform for studying EPS formation on particulate materials with enhanced translational potential:
Reactor Assembly and Substrate Preparation
- Machine a custom stainless steel ring insert with 50 divots (3.5 mm diameter) to hold 4 mm glass beads in a crystallizing dish [67].
- Sand beads with 60-grit sandpaper for 30 seconds to increase surface roughness, enhancing bacterial attachment and EPS production.
- Clean sanded beads thoroughly with deionized water and sterilize by autoclaving at 121°C for 30 minutes.
- Aseptically place one bead in each divot within the sterile reactor vessel.
Biofilm Growth Under Low-Shear Conditions
- Prepare bacterial inoculum adjusted to 0.5 McFarland standard (~7.5 × 10^7 CFU/mL) in phosphate-buffered saline [67].
- Add 50 mL of 50% BHI broth and 1 mL of inoculum to achieve approximately 1.5 × 10^6 CFU/mL initial concentration.
- Secure aluminum foil lid and place reactor in incubator with rotation at 40 RPM and 37°C for 72 hours.
- Manually replace spent broth with fresh medium at 24-hour and 48-hour time points to maintain nutrient availability.
Biofilm Harvest and Analysis
- Aseptically remove beads at experimental endpoints using sterile forceps.
- For EPS extraction, place individual beads in buffer solution and vortex or sonicate to dislodge biofilm material.
- Process recovered biofilm for compositional analysis, including polysaccharide quantification, protein measurement, and e-DNA extraction.
Research Reagent Solutions for EPS Studies
Table 2: Essential Research Reagents for EPS Analysis
| Reagent/Chemical | Function in EPS Research | Application Examples | Technical Considerations |
|---|---|---|---|
| Crystal Violet (1% solution) | Biofilm biomass staining | Quantitative assessment of total biofilm formation, visualization of biofilm architecture on surfaces [69] | Dissolve in ethanol or water; effective for initial screening but not EPS-specific |
| SYTO9/PI Live-Dead Stain | Bacterial viability determination within EPS matrix | Confocal microscopy analysis of cell viability distribution in biofilms after treatment [69] | Requires specific excitation/emission filters; SYTO9 penetrates all cells, PI only membrane-compromised cells |
| Ciprofloxacin | Antibiotic challenge studies | Evaluating EPS-mediated protection against antimicrobial agents (typically 4 µg/mL for P. aeruginosa) [69] | Concentration must be optimized for specific bacterial species and growth conditions |
| Extracellular DNA Dyes | Visualization of e-DNA in EPS matrix | Assessing structural role of e-DNA in biofilm architecture | Multiple options available with different spectral properties |
| Polysaccharide-Specific Stains | EPS polysaccharide component labeling | Lectin-based staining for confocal microscopy, compositional analysis | Specificity varies by bacterial species and polysaccharide type |
| Glutaraldehyde (2.5%) | Biofilm fixation for electron microscopy | Preserving EPS ultrastructure for SEM analysis [69] | Requires careful handling; fixation time impacts preservation quality |
Quantitative Analysis and Data Visualization in EPS Research
EPS Disruption Efficacy: Shockwave Treatment Data
Table 3: Quantitative Assessment of Shockwave Efficacy Against P. aeruginosa Biofilms
| Analysis Method | Control Group Results | Shockwave + Antibiotic Results | Reduction Efficacy | Statistical Significance |
|---|---|---|---|---|
| SEM Surface Area Coverage | Full surface coverage | 2.5% residual coverage | 97.5% removal | p < 0.001 |
| CV Staining (OD600) | High biomass (reference) | OD600 = 0.14 | Significant reduction | p < 0.01 |
| CFU Enumeration (log10CFU/mL) | High viability (reference) | 40% reduction | Significant decrease | p < 0.01 |
| CLSM Live/Dead Analysis | Predominantly live cells | 67% dead bacteria | Significant killing | p < 0.001 |
Biofilm Reactor Performance Metrics
The CDC biofilm reactor demonstrates remarkable reproducibility, with studies reporting a viable cell log density for P. aeruginosa biofilms showing minimal variability (SD = 0.2442), where 78% of variability originated from among-laboratory sources and only 22% from within-laboratory sources [66]. This consistency makes it particularly valuable for standardized EPS research requiring comparison across multiple experiments or laboratories.
The bead biofilm reactor achieves consistent biofilm growth across multiple substrates, with average densities of 5.53 and 6.21 log10 colony-forming units per mm² for S. aureus and P. aeruginosa respectively [67]. This reproducibility enables high-throughput screening of anti-EPS compounds while maintaining physiological relevance.
Experimental Workflow and EPS Function Relationships
Research Workflow for EPS Studies
EPS Structure-Function Relationships in Biofilm Protection
EPS Structure-Function Relationships
The investigation of Extracellular Polymeric Substances requires careful matching of research questions with appropriate model systems. Static well plates offer unparalleled throughput for initial screening but lack the fluid dynamics that significantly influence EPS production and organization. The CDC biofilm reactor provides standardized, reproducible conditions with defined shear stress essential for studying mechanical influences on EPS structure. The bead biofilm reactor bridges translational gaps by enabling seamless transition from in vitro analysis to in vivo models, particularly for environmental and trauma-related biofilm infections.
Emerging technologies like shockwave disruption demonstrate how physical methods can compromise EPS integrity, potentially overcoming the barrier properties that render biofilms tolerant to conventional antibiotics. As research progresses, the integration of multiple model systems throughout the experimental continuum will provide the most comprehensive understanding of EPS function. This multifaceted approach will ultimately enable development of targeted strategies to disrupt the protective EPS matrix, potentially restoring susceptibility to conventional antimicrobials and improving outcomes for biofilm-associated infections.
Overcoming Biofilm Resistance: Strategic Inhibition and EPS Modulation
The structural and functional integrity of bacterial biofilms is largely determined by the extracellular polymeric substance (EPS), a complex matrix that encompasses microbial cells and provides protection against environmental threats and antimicrobial treatments [3] [2]. This matrix, primarily composed of polysaccharides, proteins, lipids, and extracellular DNA (eDNA), establishes a physical barrier that restricts drug diffusion and facilitates resistance mechanisms [70] [1]. The maturation of this robust structure is coordinately regulated by quorum sensing (QS), a bacterial cell-cell communication system that governs the expression of virulence factors, EPS production, and biofilm development in a cell-density-dependent manner [71] [72].
Targeting the QS system presents a novel therapeutic strategy to combat biofilm-related infections. Unlike conventional antibiotics that exert lethal pressure and drive resistance, QS inhibitors (QSIs) interfere with bacterial pathogenicity without impeding growth, thereby reducing selective pressure for resistance [71] [73]. Among the most promising QSIs are synthetic furanones and peptide-based agents, which disrupt signaling pathways to prevent biofilm maturation [70]. This whitepaper details the mechanisms, experimental methodologies, and therapeutic potential of these agents, framing their activity within the context of EPS structural research.
The Nexus of Quorum Sensing and EPS-Mediated Biofilm Maturation
The biofilm lifecycle begins with the initial attachment of planktonic cells to a surface, progressing to irreversible attachment and the development of a mature, three-dimensional structure. A pivotal feature of this mature biofilm is its EPS matrix, which confers mechanical stability and resilience [1] [54]. The composition of EPS is heterogeneous, but its fundamental components include:
- Exopolysaccharides (e.g., alginate, cellulose): Form the structural scaffold [2].
- Proteins and enzymes: Contribute to structural integrity and nutrient acquisition [3] [2].
- Extracellular DNA (eDNA): Promotes cell adhesion and structural cohesion [3] [54].
- Lipids and inorganic ions: Influence hydrophobicity and cross-link matrix components [2] [54].
The production of these EPS components is intricately regulated by the QS hierarchy. In Pseudomonas aeruginosa, a model opportunistic pathogen, the QS system comprises a las-rhl-pqs regulatory network [72] [73]. The LasI/LasR system sits at the top of this hierarchy, inducing the expression of the RhlI/RhlR and PQS (Pseudomonas Quinolone Signal) systems [73]. These systems collectively regulate the expression of over 300 genes, including those responsible for EPS production, virulence factor secretion, and biofilm maturation [72]. Consequently, the disruption of QS signaling directly impedes the development of the protective EPS matrix, rendering the bacterial community more susceptible to host immune responses and antimicrobial agents.
Synthetic Furanones as Potent Quorum Sensing Antagonists
Mechanism of Action: Molecular Targeting of QS Receptors
Synthetic furanones are structural analogs of native bacterial autoinducers, such as acyl-homoserine lactones (AHLs) [70] [73]. Their primary mechanism involves competitive binding to QS receptor proteins, thereby preventing the native signal from activating the transcription of QS-controlled genes.
Recent molecular docking and dynamics simulations reveal that furanones exhibit remarkable stability and strong binding affinity for LasR and RhlR receptors in P. aeruginosa [73]. The binding is characterized by:
- Hydrophobic Interactions: Furanones with shorter alkyl side chains bind strongly to the hydrophobic pocket of RhlR, while the presence of halogen substituents enhances binding across multiple receptors [73].
- Protein Inactivation: Binding to LasR often occurs at the AHL-binding domain, establishing extensive hydrophilic interactions that lead to protein inactivation and aggregation [73].
- Hierarchical Disruption: As LasR is at the top of the QS hierarchy, its inhibition has a cascading effect, downregulating the entire QS network and subsequent EPS production [70] [73].
The disruption of this signaling cascade directly impacts biofilm integrity by suppressing the synthesis of key EPS components, such as pel and psl polysaccharides in P. aeruginosa [70].
Experimental Protocols and Key Findings
Experimental Workflow for Evaluating Synthetic Furanones: The following diagram outlines a standard research pipeline for characterizing the anti-QS and anti-biofilm activity of synthetic furanones.
Key Quantitative Findings on Synthetic Furanones: Table 1: Summary of Experimental Data for Synthetic Furanones in P. aeruginosa Studies
| Target QS Receptor | Key Furanone Structural Features | Observed Biofilm Inhibition | Impact on Virulence Factors | Primary Experimental Method |
|---|---|---|---|---|
| LasR [73] | Presence of halogen substituents | Significant reduction in mature biofilm biomass [70] | Downregulation of elastase, pyocyanin [73] | Molecular Docking, Crystal Violet Assay [73] |
| RhlR [73] | Shorter alkyl side chains | Disruption of biofilm architecture & integrity [70] | Reduced rhamnolipid production [73] | Homology Modeling, Swarming Motility Assay [73] |
| PqsR [73] | Structurally distinct from native AI (NHQ) | Weaker biofilm inhibition compared to LasR/RhlR targeting [73] | Suppressed pyocyanin and HQNO production [73] | Molecular Dynamics Simulation, Pyocyanin Assay [73] |
Peptide-Based Quorum Sensing Inhibitors
Mechanism of Action: Disrupting Signal Synthesis and Reception
Peptide-based QSIs represent another potent class of anti-biofilm agents. Their mechanisms are diverse and include:
- Receptor Antagonism: Synthetic peptides can mimic or block the binding site of native autoinducer peptides (AIPs), particularly in Gram-positive bacteria, preventing receptor activation [70].
- Signal Synthase Inhibition: Certain peptides directly inhibit the enzymes (e.g., LuxI-type AHL synthases) responsible for producing QS signaling molecules [70] [71].
- QS Signal Degradation: Some engineered peptides are designed to cleave or degrade AHL molecules, effectively quenching the QS signal in the extracellular environment [70].
By interfering with QS circuitry, peptide-based agents suppress the transcription of genes encoding for EPS components like exopolysaccharides and extracellular DNA, thereby compromising the biofilm matrix [70].
Experimental Protocols and Key Findings
Experimental Workflow for Evaluating Peptide-Based QSIs: The evaluation of peptide-based inhibitors follows a multi-faceted approach to confirm target engagement and phenotypic impact.
Key Quantitative Findings on Peptide-Based QSIs: Table 2: Summary of Experimental Data for Peptide-Based QS Inhibitors
| Proposed Mechanism | Example Agent / Class | Observed Biofilm Inhibition | Impact on Virulence Factors | Gene Expression Changes (qPCR) |
|---|---|---|---|---|
| Receptor Antagonism [70] | Synthetic AIP mimics | >70% reduction in biofilm biomass in S. aureus [70] | Reduced toxin production | Downregulation of hla (alpha-toxin gene) |
| Signal Interference [70] [71] | Engineered lactonase peptides | Degradation of AHLs, preventing biofilm maturation [70] | Attenuated virulence in animal models [71] | Broad downregulation of QS-regulated regulon |
| Multi-target Inhibition [70] | Synthetic peptides (unspecified) | Disruption of biofilm architecture & enhanced antibiotic penetration [70] | Synergistic effect with conventional antibiotics [70] | Downregulation of pelA, pslB (EPS genes) [70] |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents for QSI and Biofilm Research
| Reagent / Material | Function / Application | Specific Example / Note |
|---|---|---|
| Recombinant QS Receptors (e.g., LasR, RhlR) [73] | Target protein for molecular docking and in vitro binding assays. | Purified LasR (PDB ID: 6V7X) used in furanone studies [73]. |
| Crystal Violet [72] | A dye that stains biomass; used for quantitative biofilm assessment. | Standard protocol for static biofilm assays in microtiter plates [72]. |
| Calcein-AM / Propidium Iodide (PI) [72] | Fluorescent live/dead stains for Confocal Laser Scanning Microscopy (CLSM) of biofilms. | Allows visualization of 3D biofilm architecture and cell viability [72]. |
| Chromobacterium violaceum CV017 [73] | A biosensor strain for detecting AHL inhibition; violacein pigment is QS-controlled. | Used for high-throughput screening of potential QSIs [73]. |
| DNase I [3] | An enzyme that degrades extracellular DNA (eDNA), a key EPS component. | Used to study the role of eDNA in biofilm stability and resistance [3]. |
| Specific Hydrolytic Enzymes (e.g., Serratiopeptidase, α-amylase) [3] | Disrupt specific EPS components (proteins, polysaccharides) to weaken biofilm matrix. | Serratiopeptidase enhances ofloxacin activity against sessile cells [3]. |
Synthetic furanones and peptide-based agents represent a paradigm shift in antimicrobial strategy, moving from bactericidal activity to anti-virulence therapy. Their primary action—disrupting QS to prevent the formation of a mature, EPS-encased biofilm—directly addresses the root cause of biofilm-associated antibiotic tolerance [70] [71]. The efficacy of these agents, as demonstrated through rigorous computational and experimental methodologies, underscores their potential as either standalone therapies or synergistic partners with conventional antibiotics [70].
Future research should prioritize overcoming translational challenges, such as improving the bioavailability and pharmacokinetics of these inhibitors [70] [71]. The development of multi-targeted therapies that simultaneously disrupt multiple QS systems and EPS components, potentially leveraging nanocarrier-based delivery systems, is a promising avenue [70]. By integrating these advanced QSIs into the broader context of EPS research, the scientific community can accelerate the development of effective clinical interventions to combat persistent biofilm-mediated infections.
The rise of antimicrobial resistance (AMR) represents one of the most critical global health threats of the 21st century, with biofilm-associated infections constituting approximately 80% of all microbial infections in the human body [74]. Bacterial biofilms are structured communities of microbial cells encased within a self-produced matrix of extracellular polymeric substances (EPS) that confer significant tolerance to antibiotics, disinfectants, and host immune responses [74] [3]. The EPS matrix forms a protective shield around bacterial cells, creating a physical barrier that limits antimicrobial penetration and provides a stable environment for microbial persistence [75] [76].
2-Aminoimidazoles (2-AIs) have emerged as a promising class of anti-biofilm agents that specifically target EPS production without affecting bacterial viability [74] [77]. These compounds are analogues of oroidin family alkaloids derived from marine sponges and represent a novel anti-virulence approach that may be less likely to select for resistance compared to conventional antibiotics [74] [76]. This review examines the molecular mechanisms of 2-AI-mediated EPS inhibition, structure-activity relationships, and potential clinical applications, with particular emphasis on their evolutionarily robust nature that counter-selects for resistant mutants [78] [76].
Extracellular Polymeric Substances (EPS): The Architectural Core of Biofilms
Composition and Functional Diversity
The EPS matrix is a complex, hydrated polymer network primarily composed of polysaccharides, proteins, nucleic acids (eDNA), and lipids [3] [75] [24]. These components interact through weak physicochemical forces to form a cohesive, viscoelastic structure that encompasses microbial cells and provides mechanical stability to the biofilm architecture [3]. The composition of EPS varies significantly between bacterial species and is influenced by environmental conditions, growth stage, and nutrient availability [24] [4].
Table 1: Major Components of Bacterial EPS and Their Functional Roles
| EPS Component | Primary Functions | Representative Analytical Methods |
|---|---|---|
| Polysaccharides | Structural integrity, adhesion, cohesion, nutrient retention | FT-IR spectroscopy, carbohydrate assays [3] |
| Proteins | Enzymatic activity, structural support, surface attachment | Lowry assay, FT-IR spectroscopy [3] [4] |
| Extracellular DNA (eDNA) | Genetic exchange, structural stability, cation chelation | Phenol-chloroform extraction, fluorescence assays [3] [4] |
| Lipids | Hydrophobicity modulation, signaling | FT-IR spectroscopy, lipid extraction [3] |
| Amino Sugars (MurN, ManN, GalN, GlcN) | Structural reinforcement, microbial marker | Acid hydrolysis, chromatographic techniques [4] |
EPS as a Public Good: Ecological Implications
From a social evolution perspective, EPS production represents a cooperative trait where the metabolic cost of production is borne by individual cells, while the protective benefits are shared across the microbial community [76]. This "public good" characteristic creates an ecological dynamic where non-producing "cheater" strains can exploit the EPS produced by others, potentially outcompeting producers under certain conditions [76]. This evolutionary principle forms the theoretical foundation for 2-AI-based anti-biofilm strategies, as inhibiting public goods should select against resistance development [78] [76].
2-Aminoimidazoles: Molecular Mechanisms of EPS Inhibition
Transcriptional Regulation of EPS Biosynthesis
2-AIs specifically target the transcriptional machinery responsible for EPS production in Gram-positive and Gram-negative pathogens. In Salmonella Typhimurium, 2-AIs strongly repress the expression of csgD, the master regulator of biofilm formation [78] [76]. CsgD activates the transcription of the csgBAC operon (responsible for curli fimbriae production) and adrA (encoding a diguanylate cyclase that produces the secondary messenger c-di-GMP for cellulose synthesis) [76]. By downregulating csgD, 2-AIs simultaneously inhibit both major EPS components in Salmonella – curli and cellulose – thereby preventing the formation of the characteristic rdar (red, dry, and rough) colony morphotype associated with robust biofilm formation [76].
Figure 1: 2-AI Inhibition of Salmonella EPS Biosynthesis Pathway. 2-Aminoimidazoles target the master regulator CsgD, subsequently inhibiting both curli fimbriae and cellulose production essential for biofilm formation [78] [76].
Structure-Activity Relationships of 2-AI Analogues
Extensive structure-activity relationship (SAR) studies have identified key molecular features that optimize 2-AI anti-biofilm activity while minimizing toxicity [74]. The 5-aryl-2-aminoimidazole (5-Ar-2-AI) core structure provides the fundamental pharmacophore, with specific substitutions dramatically influencing potency and spectrum of activity:
- N1-substitutions: Alkyl chains of varying lengths (e.g., octyl groups) enhance activity against Gram-positive species [74] [77]
- 2N-substitutions: Cycloalkyl groups (e.g., cyclopentyl) improve selectivity and safety profiles [74]
- 5-aryl modifications: Electron-withdrawing groups (e.g., bromo, chloro) at the para position significantly enhance potency [74] [77]
- Amination strategies: Introduction of amino groups at the 2N-position broadens activity against Gram-negative species, particularly P. aeruginosa [74]
Recent optimization efforts have focused on 2N-substituted-aminated 5-Ar-2-AIs, which demonstrate improved activity spectra against both Gram-positive and Gram-negative pathogens while maintaining favorable toxicity profiles [74].
Table 2: Efficacy Spectrum of Optimized 2-AI Compounds Against Clinical Pathogens
| 2-AI Compound Class | E. coli | P. aeruginosa | S. aureus | S. epidermidis | MRSA | Key Structural Features |
|---|---|---|---|---|---|---|
| 2N-substituted-aminated | +++ | +++ | +++ | +++ | ++ | Amino group incorporation [74] |
| N1-2N-disubstituted | ++ | - | +++ | +++ | +++ | Dual alkyl substitution [74] |
| 2N-substituted (non-aminated) | ++ | + | + | + | + | Cycloalkyl groups [74] |
| N1-substituted | + | + | ++ | ++ | ++ | Long alkyl chains [74] |
Legend: +++ = strong inhibition (≥70% biofilm reduction); ++ = moderate inhibition (40-69% biofilm reduction); + = weak inhibition (<40% biofilm reduction); - = no significant activity
Experimental Assessment of 2-AI Efficacy
Standardized Biofilm Inhibition Assays
Quantification of 2-AI anti-biofilm activity typically employs 96-well peg-lid assays with crystal violet staining for biomass quantification [74] [78]. Key protocol parameters include:
- Bacterial strains: Clinical isolates of target pathogens (e.g., S. aureus JAR060131, S. Typhimurium ATCC14028) [77] [76]
- Growth conditions: Diluted nutrient media (e.g., TSB 1/20) to induce biofilm formation [77] [79]
- Treatment duration: 48-hour incubation with serial dilutions of 2-AI compounds [78]
- Control groups: Include untreated controls and EPS-deficient mutants (e.g., ΔcsgD) [76]
- Dual-readout assessment: Simultaneous measurement of biofilm biomass (OD570) and planktonic growth (OD600) to confirm specific anti-biofilm activity [78]
Orthopedic Implant Coating Applications
Advanced coating methodologies have been developed to covalently conjugate 2-AI compounds to titanium implant surfaces [77] [79]. The optimized protocol involves:
Figure 2: Titanium Implant Coating Workflow. Schematic representation of the covalent conjugation process for applying 2-AI coatings to orthopedic implants [77] [79].
This coating methodology achieves a 5-fold higher drug loading compared to previous methods and demonstrates resilience to standard sterilization procedures [77]. In vitro testing shows approximately 1 log reduction in S. aureus biofilm cells on coated titanium surfaces without affecting planktonic growth [77] [79].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents and Materials for 2-AI and EPS Research
| Reagent/Material | Specification | Research Application | Functional Role |
|---|---|---|---|
| LC0024-NH2 | 1-(8-aminooctyl)-5-(4-bromophenyl)-N-cyclopentyl-1H-imidazol-2-amine | Implant coating studies [77] [79] | Standard 2-AI for surface functionalization |
| RC41 | 5-(4-chlorophenyl)-N-cyclopentyl-1H-imidazol-2-amine | Salmonella EPS inhibition [78] | CsgD repression studies |
| Fmoc-APTES | Fmoc-protected 3-aminopropyltriethoxy silane | Titanium functionalization [77] [79] | Surface amination linker |
| HMDI | Hexamethylene diisocyanate | Covalent conjugation [77] [79] | Cross-linking agent |
| Cation Exchange Resin | Amberlite HPR1100 | EPS extraction [4] | Polymer separation from cells |
| Congo Red Agar | CR-containing growth medium | Morphotype assessment [76] | Visual EPS detection |
Evolutionary Robustness: Resistance Characteristics
A critical advantage of 2-AI-based EPS inhibition is its evolutionarily robust nature. Long-term serial passage experiments with Salmonella Typhimurium demonstrate that despite 40 days of continuous 2-AI exposure, populations remain fully susceptible to biofilm inhibition without developing resistance [78] [76].
The ecological explanation for this phenomenon lies in the public goods character of EPS. When a resistant mutant emerges that produces EPS despite 2-AI presence, it bears the metabolic cost of production while neighboring susceptible cells benefit from the shared protection without paying the cost [76]. In competitive assays, EPS-deficient mutants (ΔcsgD) outcompete wild-type producers under 2-AI treatment, demonstrating strong counter-selection against resistance [76].
While mutations in efflux pump regulators (e.g., ramR) and RNA polymerase components (rpoC, rpoS) may occur under prolonged 2-AI exposure, these adaptations do not restore EPS production and may even reduce fitness in untreated environments [78]. This contrasts sharply with conventional antibiotics, where resistance typically emerges rapidly and confers a selective advantage [78] [76].
2-Aminoimidazoles represent a mechanistically novel class of anti-biofilm agents that specifically target EPS production through transcriptional regulation of key matrix components. Their unique action against cooperative virulence traits, favorable toxicity profiles, and evolutionarily robust characteristics make them promising candidates for clinical development, particularly as coatings for medical implants [77] [79]. Future research directions should focus on expanding their spectrum of activity, optimizing pharmacokinetic properties for systemic applications, and exploring synergistic combinations with conventional antibiotics to address the persistent challenge of biofilm-associated infections.
The extracellular polymeric substance (EPS) matrix is a fundamental determinant of the biofilm lifestyle, contributing significantly to the recalcitrance of chronic infections to conventional antimicrobial therapy. This self-produced, protective matrix encompasses microbial cells in a complex scaffold of biopolymers, creating a formidable physical and chemical barrier that restricts antibiotic penetration, facilitates immune evasion, and promotes the emergence of resistance [80]. Comprising polysaccharides, proteins, extracellular DNA (eDNA), and lipids, the EPS can constitute over 90% of the total biofilm biomass, with its unique physicochemical properties, such as its polyanionic nature, enabling the binding and neutralization of antimicrobial agents [81] [80]. Within this heterogeneous structure, bacteria can adopt a dormant, metabolically inactive state, further reducing their susceptibility to antibiotics that target active cellular processes [80]. The clinical imperative to address this challenge is clear; biofilm-associated infections place a significant burden on healthcare systems, complicating treatments involving medical devices and chronic wounds [80]. Consequently, strategies aimed at disassembling the EPS matrix represent a promising frontier for restoring the efficacy of existing antimicrobials. This review examines the rationale for synergistic approaches that combine EPS-disrupting agents with conventional antibiotics, detailing the mechanisms, agents, and experimental methodologies that are advancing this field.
Composition and Function of the EPS Matrix
The biofilm EPS matrix is a sophisticated assemblage of macromolecules that provides structural integrity and protection. Its composition is dynamic, varying with microbial species, strain, and environmental conditions, but typically includes a few key classes of components [81] [4].
- Polysaccharides: Often the most abundant component, these exopolysaccharides form a hydrated polymer network that acts as a primary scaffold. They facilitate initial surface attachment, mediate cell-cell cohesion, and contribute to the matrix's gel-like properties and water retention [80]. Their charged residues can also sequester ions and antimicrobial molecules.
- Proteins: The proteinaceous component includes both structural proteins, which reinforce the three-dimensional architecture, and enzymes, which participate in matrix remodeling by degrading other EPS constituents (e.g., polysaccharides, proteins, and eDNA) to provide nutrients and enable biofilm dispersal [80].
- Extracellular DNA (eDNA): eDNA is a crucial structural component in many biofilms, contributing to biofilm stability through electrostatic interactions. It also facilitates horizontal gene transfer, accelerating the spread of antibiotic resistance genes within the microbial community [81] [80].
- Lipids and Biosurfactants: These components can influence hydrophobicity and surface tension, playing roles in initial adhesion, matrix structuring, and potentially in competitive interactions within microbial communities [82] [80].
The protective function of the EPS is multifaceted. It acts as a diffusion barrier, slowing the penetration of antimicrobial agents and leading to sub-lethal exposure for embedded cells [80]. Furthermore, antibiotics can be directly inactivated within the matrix through binding (e.g., aminoglycosides to eDNA), complexation, or enzymatic degradation [81] [80]. This combination of physical shielding and chemical inactivation, coupled with the altered physiology of biofilm-resident cells, is the cornerstone of biofilm-specific tolerance and resistance.
Table 1: Major Constituents of the EPS Matrix and Their Functional Roles
| EPS Constituent | Primary Functions | Impact on Antimicrobial Efficacy |
|---|---|---|
| Polysaccharides | Structural scaffolding, adhesion, water retention, ion exchange | Limits drug diffusion; can bind and neutralize antimicrobials |
| Proteins | Structural support, enzymatic matrix remodeling | Enzymatic degradation of some antimicrobials; structural barrier |
| Extracellular DNA (eDNA) | Structural stability, horizontal gene transfer, cation chelation | Binds cationic antimicrobials (e.g., aminoglycosides); spreads resistance genes |
| Lipids & Biosurfactants | Modulate surface hydrophobicity, adhesion | Can contribute to barrier properties and competitive exclusion |
Mechanisms of EPS Disruption to Potentiate Antimicrobials
Disrupting the EPS matrix targets the foundational basis of biofilm resistance. By degrading or altering key matrix components, these agents can enhance the penetration and activity of co-administered antimicrobials. The primary mechanisms of disruption include enzymatic degradation, chelation, and the inhibition of matrix synthesis or assembly.
Enzymatic Degradation of Matrix Components
Enzymes are highly specific tools for dismantling the EPS. Their use can mechanically weaken the biofilm structure, force cells into a more susceptible planktonic state, and improve antibiotic access [80].
- Glycoside Hydrolases (Dispersin B, α-Amylase): These enzymes target polysaccharide components. For instance, Dispersin B hydrolyzes poly-N-acetylglucosamine (PNAG), a key polysaccharide in staphylococcal biofilms. α-Amylase has been shown to detach Staphylococcus aureus biofilms in a concentration-dependent manner [3] [81].
- DNases: Degradation of eDNA by DNase I enzymes compromises the structural integrity of biofilms from species like Pseudomonas aeruginosa and can dissociate certain S. aureus biofilms. This also reduces the available pool of eDNA for antibiotic binding and gene transfer [81] [80].
- Proteases: Serine proteases (e.g., Savinase, Subtilisin A) and metalloproteases (e.g., Serratiopeptidase) are effective against protein-dependent biofilms. They can cleave structural proteins and adhesion factors, leading to significant reductions in biofilm biomass. Serratiopeptidase, for example, enhances the activity of ofloxacin against sessile cultures of P. aeruginosa and Staphylococcus epidermidis [3].
Chelating Agents and Metal Ion Sequestration
Divalent cations such as calcium (Ca²⁺) and magnesium (Mg²⁺) play a critical role in cross-linking anionic EPS components, particularly polysaccharides, thereby stabilizing the matrix [83]. Chelating agents like EDTA (Ethylenediaminetetraacetic acid) and citrate bind these metal ions, disrupting ionic bridges and leading to a loss of matrix cohesion and integrity. This action can increase porosity and enhance the diffusion of antimicrobials into the biofilm depths.
Inhibition of Quorum Sensing and Matrix Synthesis
Quorum sensing (QS) is a cell-cell communication system that regulates biofilm maturation and EPS production in response to population density [84]. Quorum quenching (QQ) strategies interfere with QS by degrading signaling molecules (e.g., acyl-homoserine lactones in Gram-negative bacteria) or blocking their receptors. By disrupting this coordination, QQ agents can prevent the formation of robust, mature biofilms and render established communities more susceptible to antimicrobial attack [85].
The following diagram illustrates how these different disruption strategies synergize with antibiotics to eradicate biofilms.
Quantitative Evidence of Synergistic Efficacy
The synergy between EPS-disrupting agents and conventional antimicrobials has been demonstrated across numerous in vitro studies against key pathogens. The data, often quantified by reductions in biofilm biomass, bacterial viability (log CFU), or minimum biofilm eradication concentrations (MBEC), provides compelling evidence for this approach.
Table 2: Documented Synergistic Effects Between EPS-Disrupting Agents and Antimicrobials
| EPS-Disrupting Agent | Target Pathogen & Biofilm Model | Conventional Antimicrobial | Synergistic Outcome | Reference Context |
|---|---|---|---|---|
| Serratiopeptidase (Protease) | P. aeruginosa, S. epidermidis | Ofloxacin | Absence of sessile cells when combined with MBC of ofloxacin | [3] |
| α-Amylase | S. aureus | Not Specified | Concentration- and time-dependent biofilm detachment | [3] |
| Dispersin B | Staphylococcal biofilms | Various antibiotics | Enhanced penetration and efficacy against PNAG-dependent biofilms | [80] |
| DNase I | P. aeruginosa (with NETs) | Tobramycin | Restored susceptibility by degrading protective eDNA/NET shield | [81] |
| Glycoside Hydrolases | P. aeruginosa, S. aureus (wound models) | Standard antimicrobials | Induced dispersal; enhanced killing of dispersed cells | [81] |
| 2-hydroxy-4-methoxybenzaldehyde (HMB) | Staphylococcus epidermidis | Not Specified | Targeted initial cell adhesion, inhibiting biofilm formation | [85] |
| Selenium-containing Indole Derivative | Staphylococcus aureus (via ica operon) | Not Specified | Downregulated icaA/D, inhibiting PIA/PNAG production | [85] |
Experimental Protocols for Evaluating Synergy
Robust and standardized experimental models are essential for quantifying biofilm formation and assessing the efficacy of anti-biofilm strategies. Below are detailed protocols for common assays used in this field.
In Vitro Biofilm Cultivation Models
Microtiter Plate (96-well) Assay: This is a high-throughput, cornerstone method for biofilm studies.
- Inoculation: Dilute an overnight bacterial culture in fresh, appropriate broth (e.g., Tryptic Soy Broth, Luria-Bertani) to a standardized optical density (e.g., OD600 ~0.1).
- Adhesion & Growth: Dispense 200 µL of the bacterial suspension into the wells of a sterile, flat-bottom 96-well plate. Include negative control wells with broth only. Incubate the plate statically for a specific duration (e.g., 24-48 hours) at the optimal growth temperature (e.g., 37°C for human pathogens).
- Biofilm Maturation: After incubation, carefully remove the planktonic cells by inverting and flicking the plate. Wash the adherent biofilms twice with 200 µL of phosphate-buffered saline (PBS) to remove loosely attached cells.
Biofilm Reactors & Flow-Cell Systems: For more complex and mature biofilms, systems like the Calgary Biofilm Device (MBEC Assay), drip-flow reactors, or continuous-flow capillary systems can be used. These models better simulate the shear forces and nutrient availability found in vivo, producing biofilms with characteristics more akin to clinical infections [81].
EPS Extraction and Composition Analysis
Understanding EPS composition is key to developing targeted disruptors. The CER (Cation Exchange Resin) method is a common extraction technique [4].
- Harvesting: Collect a known volume of biofilm culture (e.g., 10 mL).
- CER Addition: Add a predetermined amount of CER (e.g., Amberlite IRP-69) to the sample. The optimal resin dose should be determined empirically for a given biofilm (e.g., 60-80 g resin/g volatile solids for Pseudomonas putida).
- Extraction: Stir the mixture slowly (e.g., 600 rpm) for a defined period (e.g., 1-2 hours) at 4°C to protect heat-labile components.
- Separation: Centrifuge the suspension (e.g., 20,000 × g, 30 minutes, 4°C) to pellet cells and resin.
- Recovery: Carefully collect the supernatant, which contains the solubilized EPS. Filter through a 0.22 µm membrane to remove residual cells and store at -20°C until analysis.
Assessment of Anti-Biofilm and Antimicrobial Efficacy
Biomass Quantification - Crystal Violet (CV) Staining:
- Fix the washed biofilms from the 96-well plate by air-drying or using methanol.
- Stain each well with 200 µL of a 0.1% (w/v) crystal violet solution for 15-20 minutes.
- Wash the plate thoroughly with water to remove unbound dye.
- Destain the bound CV with 200 µL of 30% acetic acid or ethanol for 10-15 minutes.
- Transfer 125 µL of the destaining solution to a new plate and measure the absorbance at 550-600 nm. Higher absorbance correlates with greater biofilm biomass.
Viability Assessment - Colony Forming Units (CFU) Enumeration:
- After treatment, aspirate the medium from the biofilm wells and wash gently with PBS.
- Add a known volume of PBS or saline to the wells and scrape the bottom of the wells thoroughly with a pipette tip or a sterile scraper to dislodge the biofilm.
- Vortex the suspension vigorously to homogenize it.
- Perform serial 10-fold dilutions in PBS.
- Plate appropriate dilutions onto agar plates and incubate. Count the resulting colonies and calculate the Log CFU/mL or Log CFU/well.
Synergy Testing - Checkerboard Assay:
- Prepare 2-fold serial dilutions of the antibiotic in one direction of a 96-well plate containing biofilm.
- Prepare 2-fold serial dilutions of the EPS-disrupting agent in the perpendicular direction.
- Incubate the plate and determine the fractional inhibitory concentration index (FICI). A FICI of ≤0.5 is generally considered synergistic.
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Reagents for EPS and Biofilm Synergy Research
| Reagent / Material | Primary Function in Research | Example Application |
|---|---|---|
| Cation Exchange Resin (CER) | Extraction of EPS from biofilm cultures | Mild extraction of EPS components for compositional analysis [4] |
| Dispersin B | Glycoside hydrolase; degrades PNAG polysaccharide | Disruption of staphylococcal biofilms; synergy studies with antibiotics [80] |
| DNase I | Enzyme that degrades extracellular DNA (eDNA) | Destabilization of eDNA-dependent biofilms (e.g., P. aeruginosa); reduction of antibiotic binding [81] [80] |
| Proteinase K / Savinase | Broad-spectrum proteases; degrade proteinaceous matrix components | General disruption of protein-rich EPS; used to study matrix contribution to resistance [3] |
| Crystal Violet | Dye for staining and quantifying adherent biofilm biomass | Standard, high-throughput assessment of total biofilm biomass in microtiter plates |
| Calgary Biofilm Device (MBEC Assay) | High-throughput cultivation of uniform, mature biofilms | Standardized screening of antibiotic and anti-biofilm agent efficacy against biofilm-grown cells |
The strategic disruption of the EPS matrix represents a paradigm shift in combating biofilm-associated infections. By targeting the physical barrier that underpins biofilm resilience, EPS-disrupting agents—ranging from specific enzymes to chelators and quorum quenchers—can effectively resensitize embedded bacterial cells to conventional antimicrobials. The synergistic efficacy of this approach, as demonstrated in a growing body of in vitro evidence, offers a promising path to extend the utility of our existing antibiotic arsenal. Future work must focus on optimizing delivery mechanisms, such as nanotechnology-based systems, and validating these strategies in complex in vivo models and clinical settings to translate this potent synergy into tangible therapeutic advancements.
Optimizing Environmental and Nutritional Conditions to Modulate EPS Production and Composition
Within the scope of research on extracellular polymeric substances (EPS) function in biofilm structure, the deliberate manipulation of their production and composition represents a critical avenue for controlling biofilm functionality. EPS constitutes the foundational matrix of biofilms, a complex mixture of polymers that provides structural integrity, mediates adhesion, and protects embedded microbial communities from environmental stresses and antimicrobial agents [86]. The composition of EPS—primarily polysaccharides, proteins, DNA, and lipids—is not static; rather, it is dynamically influenced by environmental and nutritional cues [87] [18]. Understanding and optimizing these conditions is therefore paramount for applications ranging from mitigating biofilm-related infections to harnessing biofilms for industrial and environmental biotechnology. This guide synthesizes current research to provide a technical framework for modulating EPS through precise control of cultivation parameters.
Core EPS Components and Their Functional Roles
The extracellular polymeric substance is a sophisticated hydrogel whose functionality is derived from its specific composition. The primary constituents include:
- Polysaccharides: These form the scaffolding of the biofilm, providing overall structural integrity and influencing the matrix's porosity and mechanical stability [87] [18].
- Proteins: Enzymatic proteins facilitate metabolic processes within the biofilm, while structural proteins often mediate surface adhesion and cohesion between cells [18]. The protein-to-carbohydrate ratio is a critical indicator of biofilm characteristics [88].
- Extracellular DNA (eDNA): This component contributes to biofilm architecture through its structural role and facilitates horizontal gene transfer, promoting genetic adaptation and increased antimicrobial resistance [89] [87].
- Lipids: Though less abundant, lipids contribute to the matrix's hydrophobicity and may act as surfactants [88] [90].
- Amino Sugars: Recent studies identify amino sugars like mannosamine (ManN) and galactosamine (GalN) as exclusive EPS constituents, potentially playing specific structural or signaling roles [18].
The properties of a biofilm—whether its hydraulic resistance, antimicrobial tolerance, or mechanical robustness—are direct consequences of the interactions between these components [87] [86].
Quantitative Optimization of Environmental Conditions
Environmental factors exert profound influence on both the quantity and quality of EPS produced by microbial consortia and pure cultures. Systematic optimization of these parameters is fundamental to directing biofilm formation.
Table 1: Optimal Environmental Conditions for Maximum EPS Production
| Parameter | Optimal Condition | Organism/System | Observed EPS Yield | Key Experimental Notes |
|---|---|---|---|---|
| Temperature | 35°C | Bacterial Consortium (Klebsiella pneumoniae, Pseudomonas aeruginosa, Burkholderia cepacian) | 12.60 g/L [88] | Incubation period of 8 days. |
| 32°C | Pseudomonas aeruginosa AG01 | Not Specified | Static incubation in nutrient broth [90]. | |
| pH | 7.5 | Bacterial Consortium | 15.5 g/L [88] | Measured after 8 days of incubation. |
| 6.0 | Pseudomonas aeruginosa AG01 | Not Specified | Identified as ideal for EPS production [90]. | |
| Incubation Time | 96 hours (4 days) | Pseudomonas aeruginosa AG01 | Not Specified | For highest EPS production [90]. |
| 8 days | Bacterial Consortium | 15.5 g/L (at pH 7.5) [88] | Peak production detected. |
Experimental Protocol: Determining Optimal Temperature and pH
- Culture Inoculation: Prepare a standardized inoculum of the target microorganism(s) in an appropriate liquid growth medium.
- Parameter Variation:
- Temperature Gradient: Dispense equal volumes of the culture into multiple flasks. Incubate these flasks at different temperatures (e.g., 25°C, 30°C, 35°C, 40°C) under static or shaking conditions as required.
- pH Gradient: Prepare the growth medium and adjust aliquots to a range of pH values (e.g., 6.0, 6.5, 7.0, 7.5, 8.0) using sterile acid/base solutions. Inoculate each pH-adjusted medium and incubate at the previously determined optimal temperature.
- Harvesting: After the predetermined incubation period (e.g., 96 hours for P. aeruginosa [90]), harvest the cultures.
- EPS Extraction and Quantification: Separate cells from the culture broth via centrifugation (e.g., 20 min at 5,000 rpm). Precipitate EPS from the cell-free supernatant using multiple volumes of chilled ethanol (e.g., 4 volumes of 95% ethanol), incubate at 4°C for 24 hours, and recover the precipitate by centrifugation [88] [90]. The resulting EPS pellet can be lyophilized and weighed to determine yield.
Nutritional Regulation of EPS Yield and Composition
The availability and type of carbon and nitrogen sources are pivotal drivers of EPS biosynthesis, directly influencing metabolic fluxes toward polymer production.
Table 2: Impact of Nutritional Sources on EPS Production
| Nutrient Type | Optimal Source | Organism/System | Observed EPS Yield | Compositional Impact |
|---|---|---|---|---|
| Carbon | Glucose | Bacterial Consortium | 21.07 g/L [88] | Commonly used for high yield. |
| Glucose / Galactose | Pseudomonas aeruginosa AG01 | Not Specified | Identified as most efficient carbon source [90]. | |
| Nitrogen | Ammonium Sulfate | Bacterial Consortium | 17.84 g/L [88] | Inorganic nitrogen source. |
| Yeast Extract / Peptone | Pseudomonas aeruginosa AG01 | Not Specified | Organic nitrogen sources found most efficient [90]. | |
| Substrate Quality | Starch (over Glycerol) | Mixed Soil Bacteria & Fungi | Higher EPS-Carbohydrate/Protein Ratio [18] | Shifts EPS composition, increasing polysaccharide proportion. |
Furthermore, the presence of mineral surfaces can stimulate EPS production. Studies show that culturing microorganisms in the presence of a quartz matrix significantly increases the EPS-carbohydrate/protein ratio, indicating that surface attachment itself is a key environmental trigger for modulating EPS composition [18].
- Basal Medium Preparation: Prepare a basal culture medium lacking a carbon or nitrogen source.
- Nutrient Supplementation:
- Carbon Source Test: Supplement the basal medium with different carbon sources (e.g., glucose, galactose, glycerol, starch) at equimolar carbon concentrations while keeping the nitrogen source constant.
- Nitrogen Source Test: Supplement the basal medium with different nitrogen sources (e.g., ammonium sulfate, yeast extract, peptone, sodium nitrate) at equimolar nitrogen concentrations while keeping the carbon source constant.
- Culture and Analysis: Inoculate each medium, incubate under optimal physical conditions, and harvest. Proceed with EPS extraction and gravimetric analysis as described in Section 3.1. For deeper analysis, quantify total carbohydrate [90] and protein [88] [90] content of the purified EPS.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful EPS research requires a suite of specific reagents and methodologies for growth, extraction, and characterization.
Table 3: Key Research Reagent Solutions for EPS Studies
| Reagent/Material | Function in EPS Research | Exemplary Application |
|---|---|---|
| Cation Exchange Resin (CER) | Extracts EPS from microbial aggregates by disrupting ionic bonds between polymers and cells. | Standardized EPS extraction from bacterial/fungal cultures [18]. |
| Ethanol (95%, chilled) | Precipitates polysaccharides and other polymers from aqueous solution for isolation and quantification. | Used in 4:1 ratio with supernatant to precipitate crude EPS [88] [90]. |
| Phenol-Sulfuric Acid Reagent | Quantifies total carbohydrate content via colorimetric reaction with sugars. | Determination of carbohydrate concentration in EPS extracts [90]. |
| Folin-Ciocalteu / Lowry Reagent | Quantifies total protein content through colorimetric reaction with peptide bonds. | Measurement of protein concentration in EPS extracts [88] [18]. |
| SYTO-9 Green Fluorescent Stain | Stains nucleic acids, allowing visualization of cells and eDNA within the biofilm matrix. | Fluorescence imaging of biofilm structure [89]. |
| Quartz Matrix | Provides a sterile, inert surface to study the effects of solid interfaces on biofilm formation and EPS production. | Used in shake flasks to force microbial growth in a matrix, simulating soil environments [18]. |
Analytical Techniques for EPS Characterization
Beyond quantification, a comprehensive analysis of EPS requires characterizing its chemical and structural properties.
- Fourier Transform Infrared Spectroscopy (FTIR): Identifies functional groups and chemical bonds within the EPS. Studies consistently use FTIR to confirm the presence of characteristic groups of carbohydrates, proteins, and lipids (e.g., C=O, COOH, NH, OH) [88] [90].
- Chemical Ratio Analysis: Calculating ratios such as protein-to-carbohydrate provides insight into the EPS matrix's nature. A high ratio (e.g., 11.65:1) suggests a protein-dominated matrix, which can have different physical properties compared to a polysaccharide-rich one [88].
- Advanced Imaging and Spectroscopy: Techniques like Optical Coherence Tomography (OCT) reveal the meso-scale physical structure (porosity, thickness) of intact biofilms, while 2D-confocal Raman microscopy provides non-invasive chemical characterization and spatial mapping of EPS components [87].
Visualizing EPS Workflows and Pathways
The following diagrams summarize the key experimental and biosynthetic pathways involved in EPS research, adhering to the specified style and color guidelines.
EPS Optimization and Analysis Workflow
Key Pathways in EPS Biosynthesis
The targeted modulation of EPS production and composition through controlled environmental and nutritional parameters is a powerful strategy in biofilm research. As evidenced by contemporary studies, optimal yields are achieved by carefully balancing temperature, pH, and incubation time, while the choice of carbon and nitrogen sources can dramatically shift the biochemical profile of the EPS matrix. The emerging recognition of specific components like amino sugars and their response to environmental stimuli further deepens the complexity and opportunity in this field. By employing the standardized protocols, analytical techniques, and reagents outlined in this guide, researchers and drug development professionals can systematically engineer biofilm matrices to either combat their detrimental effects or harness their beneficial potential, thereby advancing the frontiers of microbial science and therapeutic development.
Multi-Targeted Strategies for Effective Biofilm Eradication in Clinical Settings
Bacterial biofilms are structured communities of microbial cells encased in a self-produced matrix of extracellular polymeric substances (EPS) that are responsible for up to 80% of all human microbial infections [16]. This EPS matrix constitutes over 90% of the biofilm's dry mass, creating a formidable barrier against antimicrobial treatments and host immune responses [91] [43]. The EPS provides structural integrity through a complex network of biopolymers while functioning as a protective shield that significantly contributes to antimicrobial tolerance—often making biofilm-associated infections thousands of times more resistant to antibiotics than their planktonic counterparts [16]. Understanding the composition and functional dynamics of the EPS matrix is therefore fundamental to developing effective eradication strategies against biofilm-mediated infections in clinical settings.
The EPS matrix is composed of a complex mixture of polysaccharides, proteins, extracellular DNA (eDNA), lipids, and other biopolymers that together create a hydrated, three-dimensional polymer network [91] [3] [2]. This matrix is not merely a physical scaffold but represents a dynamically functional component of microbial communities, facilitating cell-cell communication, nutrient trapping, and horizontal gene transfer [91] [16]. The composition of EPS varies significantly depending on the microbial species, environmental conditions, and biofilm age, which contributes to the heterogeneity and adaptability of biofilms in clinical environments [43] [1]. This review examines multi-targeted therapeutic approaches that specifically address the structural and functional complexity of the EPS matrix to achieve effective biofilm eradication.
EPS Composition and Functional Architecture in Biofilms
Structural Components of the EPS Matrix
The EPS matrix is primarily composed of macromolecular components that collectively determine the physicochemical and mechanical properties of biofilms. The table below summarizes the key constituents and their functional roles in biofilm integrity and pathogenicity.
Table 1: Major EPS Components and Their Functional Roles in Clinical Biofilms
| EPS Component | Primary Functions | Clinical Significance |
|---|---|---|
| Exopolysaccharides (e.g., dPNAG, alginate, Pel, Psl) | Structural scaffolding, adhesion, cohesion, barrier formation | dPNAG is produced by diverse pathogens including S. aureus and E. coli; alginate is characteristic of P. aeruginosa in cystic fibrosis infections [16] |
| Proteins (including enzymes and amyloid fibers) | Structural support, enzymatic activity, adhesion, matrix stability | Extracellular proteases contribute to virulence; amyloid fibers enhance structural integrity and resistance [3] [2] |
| Extracellular DNA (eDNA) | Structural integrity, cation chelation, genetic exchange, adhesion | Network formation in P. aeruginosa; cell lysis-derived in S. aureus; contributes to antibiotic tolerance [91] |
| Lipids and Surfactants | Hydrophobicity modulation, surface activity, architectural adaptation | Influence biofilm morphology and dispersal mechanisms [2] |
| Minerals (e.g., Ca²⁺, Mg²⁺) | Matrix cross-linking, structural enhancement, ionic bridging | Divalent cations strengthen EPS cohesion; calcite contributes to matrix integrity in several pathogens [43] [2] |
Architectural Organization and Mechanical Properties
The three-dimensional architecture of biofilms features heterogeneous microcolonies interspersed with fluid channels that facilitate nutrient transport and waste removal [91] [16]. This organized structure results from a sophisticated developmental process beginning with initial attachment and progressing through microcolony formation, maturation, and eventual dispersal [1] [16]. The mechanical stability of biofilms—encompassing properties such as cohesion, adhesion, and viscoelasticity—is predominantly governed by EPS composition and the interactions between its various components [43] [3].
The spatial organization within biofilms creates gradients of nutrients, oxygen, and metabolic activity that contribute to physiological heterogeneity among embedded cells [16]. This heterogeneity includes subpopulations of dormant cells and persisters with dramatically reduced metabolic activity, which are notoriously tolerant to conventional antibiotics [16]. The EPS matrix further impedes antibiotic penetration through a combination of physical barrier functions, chemical interactions with antimicrobial agents, and reduced diffusion rates, collectively enabling persistent infections that are difficult to eradicate [91] [16].
Figure 1: EPS Components and Their Primary Functional Relationships in Biofilm Matrix
Enzymatic Strategies for EPS Disruption
Glycoside Hydrolases and Exopolysaccharide Degradation
Glycoside hydrolases target the carbohydrate components of the EPS matrix, which often serve as primary structural elements. Dispersin B is a particularly notable glycoside hydrolase that specifically degrades poly-N-acetylglucosamine (dPNAG), a key exopolysaccharide produced by numerous clinically significant pathogens including Staphylococcus aureus, Escherichia coli, and Klebsiella pneumoniae [16]. This enzyme catalyzes the hydrolysis of β-(1,6)-N-acetyl-D-glucosamine linkages, effectively disrupting the structural integrity of biofilms dependent on dPNAG for matrix stability [16]. Similarly, alginate lyase targets alginate-producing Pseudomonas aeruginosa biofilms—particularly relevant in cystic fibrosis lung infections—by cleaving the glycosidic linkages between mannuronic and guluronic acid residues [16].
The efficacy of glycoside hydrolases can be significantly influenced by chemical modifications of the target polysaccharides. For instance, the N-deacetylation and O-succinylation of dPNAG affect enzyme accessibility and catalytic efficiency [16]. The table below summarizes key glycoside hydrolases with demonstrated anti-biofilm potential against clinically relevant pathogens.
Table 2: Glycoside Hydrolases for Targeting Biofilm Exopolysaccharides
| Enzyme | Target Polysaccharide | Mechanism of Action | Clinically Relevant Pathogens |
|---|---|---|---|
| Dispersin B | Poly-β-(1,6)-N-acetyl-D-glucosamine (dPNAG) | Hydrolyzes β-(1,6) glycosidic linkages | S. aureus, E. coli, K. pneumoniae, A. baumannii [16] |
| Alginate Lyase | Alginate | Cleaves glycosidic bonds in alginate polymer | P. aeruginosa (particularly in cystic fibrosis) [16] |
| Amylases | Starch-like polysaccharides | Hydrolyzes α-(1,4) glycosidic bonds | S. aureus [3] |
| Cellulases | Cellulose | Degrades β-(1,4) glucose polymers | Various biofilm-forming species [16] |
Proteases and Nucleases for Matrix Disruption
Proteases target the proteinaceous components of the EPS matrix, including structural proteins and functional enzymes that contribute to biofilm integrity. Serratiopeptidase and subtilisin A have demonstrated efficacy in degrading protein-based EPS components, with studies showing enhanced antibiotic penetration when used in combination with fluoroquinolones against P. aeruginosa and S. epidermidis biofilms [3]. The metalloprotease serratiopeptidase at concentrations of 10 U/mL combined with ofloxacin at minimal inhibitory concentrations resulted in substantial reduction of sessile cells, while thermal inactivation of the enzyme abolished this effect, confirming the enzymatic mechanism of action [3].
Deoxyribonucleases (DNases) degrade extracellular DNA (eDNA), which serves as a crucial structural component in many clinical biofilms. eDNA functions in cell-cell adhesion, matrix stability, and cation chelation within the biofilm architecture [91] [16]. In P. aeruginosa biofilms, eDNA forms grid-like structures that provide architectural integrity, while in S. aureus, it is released through controlled cell lysis and integrates into the matrix [91]. DNase treatment disrupts these eDNA-dependent structural networks, facilitating biofilm disintegration and enhanced susceptibility to antimicrobial agents [16].
Experimental Protocol: Enzymatic Biofilm Disruption Assay
Objective: Evaluate the efficacy of glycoside hydrolases, proteases, and DNases in disrupting pre-established biofilms of clinically relevant pathogens.
Materials and Reagents:
- Biofilm Formation: Culture tubes or 96-well plates; appropriate growth media (e.g., Tryptic Soy Broth); bacterial suspensions standardized to 10⁶ CFU/mL.
- Enzyme Preparations: Dispersin B (100 µg/mL in PBS); Proteinase K (1 mg/mL in PBS); DNase I (100 U/mL in PBS).
- Staining Solutions: Crystal violet (0.1% for biomass quantification); fluorescent stains (SYTO9/propidium iodide for viability assessment).
- Equipment: Microplate reader; confocal laser scanning microscope; atomic force microscope for mechanical properties assessment.
Methodology:
- Biofilm Development: Inoculate surfaces with bacterial suspension and incubate for 24-48 hours at appropriate temperature to establish mature biofilms.
- Enzyme Treatment: Gently wash established biofilms with sterile PBS to remove non-adherent cells. Apply enzyme solutions at optimized concentrations and incubate for 2-24 hours at 37°C.
- Assessment of Biofilm Disruption:
- Biomass Quantification: Fix biofilms with methanol, stain with 0.1% crystal violet for 15 minutes, destain with ethanol-acetone mixture, measure absorbance at 595 nm.
- Viability Assessment: Stain with LIVE/DEAD BacLight bacterial viability kit, visualize with confocal microscopy.
- Mechanical Properties: Use atomic force microscopy to measure changes in biofilm cohesive strength following enzyme treatment [43].
- Synergy Testing: Combine sub-effective concentrations of enzymes with conventional antibiotics to evaluate enhanced susceptibility.
Data Analysis: Compare treated and untreated biofilms using statistical analysis (t-test or ANOVA with post-hoc testing). Synergy is determined when combination treatment results in ≥2-log reduction in viable counts compared to most effective single agent.
Combination Therapies for Enhanced Biofilm Eradication
Enzyme-Antibiotic Combinations
The combination of biofilm-disrupting enzymes with conventional antibiotics represents a promising strategy to overcome the protective barrier function of the EPS matrix. This approach leverages enzymes to degrade structural EPS components, thereby enhancing antibiotic penetration and efficacy against embedded bacterial cells. A notable example includes the combination of α-amylase with antibiotics, which has demonstrated significant biofilm disruption against S. aureus in a concentration- and time-dependent manner [3]. Similarly, the combination of serratiopeptidase with ofloxacin achieved near-complete eradication of P. aeruginosa and S. epidermidis biofilms when the enzyme was used at 10 U/mL alongside the antibiotic at minimal bactericidal concentration [3].
The sequential application of EPS-degrading enzymes followed by antibiotics often proves more effective than concurrent administration, as the enzymatic disruption must precede antibiotic exposure to maximize penetration and target access. This strategy is particularly relevant for biofilms characterized by extensive EPS production that creates a significant diffusion barrier against antimicrobial agents [3] [16].
Anti-Cancer Agents and Unconventional Combinations
Interestingly, certain anti-cancer agents have demonstrated anti-biofilm properties, suggesting potential for drug repurposing in biofilm management. Cisplatin, a platinum-based chemotherapeutic, has shown efficacy against P. aeruginosa biofilms in a murine keratitis model, while 5-Fluorouracil, a uracil analog used in cancer treatment, suppressed biofilm formation and virulence gene expression in E. coli in a dose-dependent manner [92]. These findings highlight the potential for cross-disciplinary therapeutic approaches targeting fundamental cellular processes shared between cancer proliferation and biofilm development.
Additional combination strategies include the use of N-acetylcysteine (NAC) with ciprofloxacin, which demonstrated synergistic effects against P. aeruginosa biofilms in cystic fibrosis patients. NAC is thought to inhibit EPS matrix production, thereby compromising biofilm structural integrity and enhancing ciprofloxacin penetration [92]. Similarly, macrolides combined with carbapenems or glycopeptides have shown promise against Gram-negative and Gram-positive biofilm infections, respectively, through targeting of alginate components in the EPS matrix [92].
Table 3: Promising Combination Therapies for Biofilm Eradication
| Combination Approach | Components | Proposed Mechanism | Target Pathogens |
|---|---|---|---|
| Enzyme-Antibiotic | Dispersin B + β-lactams | EPS degradation enhances antibiotic penetration | dPNAG-producing pathogens [16] |
| Antibiotic-Adjuvant | N-acetylcysteine + Ciprofloxacin | Inhibition of EPS production; enhanced penetration | P. aeruginosa (cystic fibrosis) [92] |
| Drug Repurposing | Cisplatin + Conventional antibiotics | Anti-biofilm activity through undefined mechanisms | P. aeruginosa [92] |
| Dual Antibiotic | Clarithromycin + Vancomycin | Alginate targeting combined with cell wall inhibition | Pseudomonas and Staphylococcus spp. [92] |
Advanced and Emerging Anti-Biofilm Strategies
Nanoparticle-Based Approaches
Nanoparticles represent a promising frontier in biofilm eradication due to their ability to penetrate the EPS matrix and target embedded bacterial cells. Metal oxide nanoparticles such as TiO₂, CuO, ZnO, and Fe₃O₄ have demonstrated significant efficacy against bacterial biofilms across diverse species [91]. These nanomaterials function through multiple mechanisms, including direct interaction with bacterial cell walls and membranes, disruption of vital molecular processes, and generation of reactive oxygen species that damage cellular components [91]. The small size and high surface area-to-volume ratio of nanoparticles facilitate enhanced diffusion through the EPS matrix, potentially bypassing the limitations of conventional antimicrobial agents.
Polymer-based nanoparticles offer additional advantages of biocompatibility and biodegradability while functioning as effective drug delivery systems for targeted antimicrobial therapy [91]. These nanocarriers can be engineered to encapsulate antibiotics, enzymes, or other anti-biofilm agents and release them in a controlled manner within the biofilm microenvironment. The surface properties of nanoparticles can be modified to interact specifically with EPS components, further enhancing their penetration and retention within biofilms [91] [92].
Quorum Sensing Inhibition and EPS Modulation
Quorum sensing (QS) represents a promising target for anti-biofilm strategies as it regulates key processes in biofilm development, including EPS production and maturation. QS inhibitors (QSIs) interfere with bacterial cell-to-cell communication systems, potentially disrupting biofilm formation without exerting direct bactericidal pressure [91] [93]. By targeting the signaling networks that coordinate biofilm development, QSIs can prevent the transition from planktonic to sessile lifestyles and reduce the production of EPS components that constitute the structural framework of biofilms [91].
Experimental approaches to QS inhibition include:
- Signal analog development: Designing molecules that structurally resemble natural autoinducers but function as competitive antagonists.
- Signal degradation: Utilizing enzymes such as acyl-homoserine lactonases that degrade QS signaling molecules.
- Receptor blockade: Developing compounds that interfere with signal reception and subsequent gene expression regulation.
Combining QS inhibitors with EPS-degrading enzymes or conventional antibiotics may provide synergistic effects by simultaneously disrupting biofilm coordination mechanisms and structural integrity [91] [93].
Figure 2: Multi-Targeted Strategic Approaches for Biofilm Eradication
The Scientist's Toolkit: Essential Research Reagents and Methodologies
Table 4: Essential Research Reagents for EPS and Biofilm Studies
| Reagent/Category | Specific Examples | Primary Research Application |
|---|---|---|
| EPS-Degrading Enzymes | Dispersin B, Alginate lyase, Proteinase K, DNase I | Targeted disruption of specific EPS components; mechanistic studies of matrix function [43] [16] |
| Analytical Tools for EPS Characterization | FT-IR spectroscopy, CLSM, AFM | Composition analysis; structural visualization; mechanical properties assessment [43] [3] |
| Biofilm Culture Systems | CDC biofilm reactor, Calgary device, 96-well plate models | Controlled biofilm growth under standardized or shear stress conditions [43] |
| Staining and Visualization | Crystal violet, SYTO9/propidium iodide, FITC-conjugated lectins | Biomass quantification; viability assessment; EPS component localization [43] [18] |
| EPS Modification Agents | Periodic acid, EDTA, Dispersin B, Divalent cations | Selective targeting of EPS components; studying matrix cross-linking [43] |
| Antimicrobial Agents | Conventional antibiotics, Quaternary ammonium compounds, Antimicrobial peptides | Efficacy assessment alone and in combination with EPS-targeting agents [92] |
Experimental Protocol: EPS Extraction and Composition Analysis
Objective: Extract and characterize the compositional profile of EPS from bacterial biofilms to understand matrix contributions to biofilm properties.
Materials and Reagents:
- Extraction Resin: Cation exchange resin (CER; Amberlite HPR1100) [18]
- Analysis Reagents: Sulfuric acid (0.75 M) for carbohydrate hydrolysis; copper sulfate solution with Folin-Ciocalteu reagent for protein assay; phosphate saline buffer (PBS) [18]
- Culture Conditions: Appropriate growth media; carbon sources (glycerol, starch); quartz matrix for surface attachment [18]
Methodology:
- Biofilm Growth: Cultivate bacterial strains in appropriate media for 4 days with shaking (100 rpm) at 30°C, with or without quartz matrix to evaluate surface attachment effects [18].
- EPS Extraction:
- Collect 10 mL aliquots from cultures using graduated cylinder.
- Add predetermined amount of cation exchange resin (optimized for specific bacterial species).
- Mix thoroughly for specified duration at controlled temperature.
- Separate extract by centrifugation or filtration [18].
- EPS Component Analysis:
- Total Carbohydrates: Hydrolyze with 0.75 M H₂SO₄ (autoclave at 100°C for 10 min), dilute with PBS, quantify using bicinchoninic acid (BCA) microplate assay measuring absorbance at 562 nm [18].
- Total Proteins: Use Lowry assay microplate method with copper sulphate and Folin-Ciocalteu reagent, measure absorbance at 750 nm [18].
- Amino Sugars: Analyze muramic acid, mannosamine, galactosamine, and glucosamine content using appropriate chromatographic or colorimetric methods [18].
- Data Normalization and Interpretation: Express results relative to total biomass or cell counts; compare compositional profiles across growth conditions and bacterial strains.
Technical Considerations: Extraction efficiency varies by bacterial species and growth conditions; methodological consistency is critical for comparative studies. Colorimetric assays may exhibit cross-interference between different EPS components, requiring appropriate controls and validation [86].
The multifaceted nature of biofilm-associated infections demands equally sophisticated eradication strategies that specifically target the structural and functional complexity of the EPS matrix. The approaches discussed herein—enzymatic disruption, combination therapies, nanoparticle technology, and quorum sensing inhibition—represent promising avenues for overcoming the formidable challenges posed by clinical biofilms. Future research directions should focus on optimizing delivery mechanisms for EPS-targeting agents, developing personalized approaches based on pathogen-specific EPS composition, and exploring synergistic combinations that address multiple aspects of biofilm biology simultaneously.
Advancements in analytical techniques for EPS characterization will be crucial for deepening our understanding of matrix architecture and function. Moving beyond reliance on colorimetric methods to more comprehensive analytical approaches will enable researchers to identify novel targets within the EPS matrix and develop increasingly precise interventions [86]. As our knowledge of biofilm biology expands, so too will our capacity to design effective, targeted strategies to combat these resilient microbial communities, ultimately improving outcomes for patients suffering from persistent biofilm-associated infections.
Evaluating Anti-EPS Strategies: Efficacy, Resistance Profiles, and Clinical Potential
The crisis of antimicrobial resistance necessitates the development of innovative anti-infective strategies with reduced potential for resistance evolution. Anti-virulence drugs that specifically target bacterial pathogenicity without impacting growth present a promising alternative to conventional antibiotics. A cornerstone of bacterial virulence is biofilm formation, which enhances tolerance to antimicrobials and host immune defenses. Biofilms are structured communities of bacteria encased in a self-produced matrix of extracellular polymeric substances (EPS) [94]. This case study examines the long-term efficacy and evolutionary consequences of inhibiting EPS production in Salmonella biofilms, situating this approach within the broader thesis that understanding EPS function is critical for developing evolutionarily robust anti-biofilm strategies. Unlike conventional antibiotics, which impose strong selective pressures for resistance, EPS inhibition leverages social evolution theory to potentially counter-select resistant mutants, offering a sustainable therapeutic paradigm [76].
The Public Good Nature of Salmonella EPS
EPS Composition and Function in Salmonella Biofilms
Salmonella biofilms rely on a complex EPS matrix whose primary components include curli fimbriae, cellulose, colanic acid, and the Vi antigen in typhoidal serovars [95]. This matrix is not merely structural; it is functionally critical for attachment, community stability, and virulence. The production of these components is centrally regulated by the transcriptional activator CsgD, which acts as the master regulator of biofilm formation [76]. From a social evolution perspective, these EPS components function as public goods—costly products synthesized by individual cells that confer benefits to the entire local population, including non-producing "cheater" cells [76].
Experimental evidence confirms that EPS-deficient Salmonella mutants (e.g., ΔcsgD) exhibit severely compromised biofilm formation and significantly reduced tolerance to antimicrobials and disinfectants [76]. Specifically, EPS-deficient mutants show a 72-76% reduction in biofilm biomass and increased susceptibility to hydrogen peroxide and ciprofloxacin compared to their wild-type counterparts [76]. This protective function establishes EPS as a virulence factor whose inhibition could therapeutically disarm the pathogen.
Evolutionary Rationale for Targeting Public Goods
The public goods character of EPS creates a unique evolutionary scenario when targeted for inhibition. Social evolution theory predicts that in a mixed population of EPS producers and non-producers, non-producers gain a fitness advantage by benefiting from the shared EPS without bearing the metabolic cost of its production [76]. When an EPS inhibitor is introduced, resistant mutants that resume EPS production would incur this metabolic cost while their produced EPS becomes a public good that also benefits nearby inhibitor-sensitive cells. This creates a net fitness disadvantage for resistant mutants, leading to their counter-selection within the population [76]. This theoretical framework provides the foundation for developing evolutionarily robust anti-biofilm strategies.
Experimental Investigation of Long-Term EPS Inhibition
Compound Selection and Validation
The core experimental work utilized a 5-aryl-2-aminoimidazole-based (2-AI) EPS inhibitor designated RC41 (5-(4-chlorophenyl)-N-cyclopentyl-1H-imidazol-2-amine) [96] [78]. This compound specifically represses the expression of csgD, thereby preventing the biosynthesis of key EPS components like curli fimbriae and cellulose without directly affecting bacterial viability [96]. Initial dose-response characterization established that a concentration of 50 µM optimally inhibited biofilm formation (approximately 75% reduction) without impacting planktonic growth, thereby fulfilling the criteria for a pure anti-virulence agent [96].
Table 1: Dose-Response Profile of EPS Inhibitor RC41 in Salmonella Typhimurium
| Concentration (µM) | Effect on Biofilm Biomass | Effect on Planktonic Biomass |
|---|---|---|
| <20 | No effect | No effect |
| 20-80 | Dose-dependent inhibition | Increase (shift to planktonic phase) |
| 50 (selected) | ~75% inhibition | No reduction |
| ≥100 | ≥99% inhibition | Reduction (growth delay side effect) |
Long-Term Evolution Experiment Protocol
To assess evolutionary responses, a serial passage evolution experiment was conducted, mirroring realistic long-term treatment scenarios [96] [78].
- Biofilm Culture Conditions: Biofilms were grown statically on the bottom of Petri dishes containing diluted Tryptic Soy Broth, simulating nutrient-limited industrial surfaces [96].
- Passage Schedule: After 48 hours of growth, biofilms were harvested and used to inoculate fresh medium for the next cycle. This process was repeated for 20 cycles (40 days) [96].
- Experimental Lineages: The study included three independent RC41-treated lineages (50 µM) and three control lineages (0 µM) to distinguish adaptations specific to inhibitor treatment from general adaptations to the biofilm lifestyle [96].
- Assessment Points: Evolved populations were evaluated for resistance development (defined as the ability to resume biofilm formation during treatment), genomic alterations, and phenotypic changes.
Key Findings on Efficacy and Resistance
The long-term treatment demonstrated remarkable durability of the anti-biofilm effect. After 40 days of continuous exposure, the RC41-evolved populations remained as susceptible to biofilm inhibition as the ancestral strain, with biofilm biomass reduction of approximately 73% compared to 77% for the wild-type [96]. Crucially, no resistance to the anti-biofilm activity was observed, validating the social evolution hypothesis that EPS inhibition does not select for resistance [96] [78].
Table 2: Summary of Evolutionary Outcomes After Long-Term EPS Inhibitor Exposure
| Evolutionary Aspect | Finding | Implication |
|---|---|---|
| Resistance to Biofilm Inhibition | Not observed | Anti-biofilm efficacy remains robust over time |
| Biofilm Biomass Post-Treatment | Remained low (0.047 OD₅₇₀ₙₘ) comparable to treated ancestor | No restoration of virulence trait |
| Genomic Adaptation | Mutations in ramR and RNA polymerase genes | Adaptation to unintended side effects |
| Phenotypic Outcome | Reduced basal biofilm formation, increased efflux activity | Trade-offs may reduce environmental fitness |
Unintended Evolutionary Adaptations and Mitigation
Genomic and Phenotypic Changes
Despite the absence of classical resistance, whole-genome sequencing revealed that the RC41-evolved populations had accumulated mutations, particularly in genes encoding the RamR transcriptional regulator and components of the RNA polymerase complex [96] [78].
- RamR Mutations: These mutations occurred at high frequency and are known to derepress the AcrAB-TolC efflux pump system, leading to increased efflux activity [96].
- Fitness Advantage: The selection for these mutations was attributed to a slight growth-delaying side effect of RC41 at the employed concentration. Enhanced efflux activity provided a fitness advantage by mitigating this growth delay, not by overcoming EPS inhibition [96].
- Phenotypic Trade-Off: The evolved lineages exhibited a reduced basal biofilm-forming capacity in the absence of the inhibitor, indicating an evolutionary cost to adaptation [96].
These findings highlight a crucial distinction: bacterial populations can adapt to drug treatment without necessarily developing resistance to the drug's intended mechanism of action.
Specificity Enhancement Through Chemical Modification
Following the discovery that unintended side effects drove adaptation, researchers chemically modified the RC41 compound to enhance its specificity [96] [78]. The modified inhibitor demonstrated:
- Retained Anti-Biofilm Efficacy: Preservation of potent CsgD repression and biofilm inhibition.
- Reduced Growth Delay: Partial alleviation of the unintended effect on planktonic growth.
- Eliminated Selection Pressure: The modified compound no longer selected for mutations leading to increased efflux pump activity [96].
This successful refinement underscores the importance of drug specificity in anti-virulence strategies and demonstrates that chemical optimization can mitigate unintended evolutionary consequences.
Comparative Analysis with Conventional Antibiotics
The evolutionary trajectory of biofilms under EPS inhibition starkly contrasts with that observed under conventional antibiotic pressure. When Salmonella biofilms are exposed to sub-inhibitory concentrations of antibiotics like ciprofloxacin, azithromycin, or cefotaxime, resistance evolves rapidly through distinct genetic mechanisms [97]. Furthermore, antibiotic-resistant lineages often exhibit collateral trade-offs, including reduced biofilm-forming capacity and attenuated virulence [97]. This contrasts with EPS inhibitor-evolved populations, which maintained stable susceptibility to the anti-biofilm effect while accumulating different types of adaptive mutations. The table below summarizes these critical differences.
Table 3: Comparison of EPS Inhibitors vs. Conventional Antibiotics on Salmonella Biofilms
| Parameter | EPS Inhibitor (2-AI) | Conventional Antibiotics |
|---|---|---|
| Primary Target | Virulence regulator CsgD (non-essential) | Essential cellular processes (e.g., DNA replication, cell wall synthesis) |
| Resistance Development | Not observed after 40 days of treatment [96] | Rapid evolution of resistance [97] |
| Evolutionary Mechanism | Social counter-selection of resistant mutants [76] | Direct selection for survival mutations [97] |
| Primary Adaptation | Mutations in efflux regulators (e.g., ramR) [96] | Drug-specific target mutations [97] |
| Fitness Consequence | Reduced basal biofilm formation [96] | Variable trade-offs in biofilm formation and virulence [97] |
| Therapeutic Strategy | Anti-virulence | Bactericidal/Bacteriostatic |
Experimental Protocols for EPS Inhibition Studies
Biofilm Cultivation and EPS Inhibitor Treatment
Protocol for Serial Passage Evolution Experiment [96] [78]:
- Inoculum Preparation: Grow Salmonella Typhimurium overnight in diluted Tryptic Soy Broth (dTSB).
- Biofilm Initiation: Inoculate Petri dishes with dTSB containing 50 µM RC41 (for treated lineages) or vehicle control (for control lineages).
- Growth Conditions: Incubate statically for 48 hours at 30°C to allow biofilm formation on the bottom of the dishes.
- Passage: Harvest biofilms by scraping, homogenize, and use to inoculate fresh medium for the next cycle.
- Repetition: Repeat this process for 20 cycles (40 days), maintaining separate lineages.
- Analysis: Assess resistance development, genomic changes, and phenotypic characteristics in evolved populations.
Assessment Methods for Biofilm Phenotypes
- Biofilm Biomass Quantification: Use crystal violet staining measured at OD₅₇₀ₙₘ to assess total biofilm biomass [96] [97].
- Planktonic Growth Monitoring: Measure optical density of the culture supernatant to detect effects on growth [96].
- Morphotype Analysis: Plate isolates on Congo Red agar to assess curli and cellulose production based on colony color and morphology (rdar morphotype indicates EPS production) [76].
- Antimicrobial Tolerance Testing: Challenge biofilms with disinfectants (e.g., hydrogen peroxide) or antibiotics to evaluate the protective role of EPS [76].
Research Reagent Solutions
Table 4: Essential Research Reagents for Salmonella Biofilm and EPS Studies
| Reagent/Condition | Function in Research | Specific Example/Application |
|---|---|---|
| 5-aryl-2-aminoimidazoles (2-AI) | EPS inhibitors targeting CsgD expression | RC41 at 50 µM for specific biofilm inhibition without growth impact [96] |
| Diluted Tryptic Soy Broth (dTSB) | Nutrient-limited biofilm growth medium | Mimics industrial conditions; promotes biofilm formation over planktonic growth [96] |
| Congo Red (CR) Agar | EPS component detection | Differentiates EPS producers (rdar) from non-producers (saw) based on colony morphotype [76] |
| Crystal Violet (CV) Stain | Biofilm biomass quantification | Stains EPS and attached cells; absorbance measurement correlates with biomass [97] |
| Cholesterol-Coated Surfaces | Gallstone biofilm mimic | For studying chronic S. Typhi biofilms relevant to carrier state [95] |
| Cation Exchange Resin (CER) | EPS extraction from cultures | Standardized method for isolating EPS for compositional analysis [4] |
Signaling Pathways and Experimental Workflows
CsgD Regulatory Pathway and Inhibitor Mechanism
Diagram 1: CsgD Regulatory Pathway and Inhibitor Mechanism. The 2-AI inhibitor RC41 represses CsgD expression, preventing biosynthesis of key EPS components curli and cellulose.
Evolutionary Selection Dynamics Under EPS Inhibition
Diagram 2: Evolutionary Selection Dynamics. Without treatment, non-producers exploit producers. With treatment, resistant producers bear metabolic and inhibitory costs, giving susceptible non-producers a competitive advantage.
This case study demonstrates that long-term EPS inhibition in Salmonella biofilms represents an evolutionarily robust anti-virulence strategy. The core finding—that resistance to biofilm inhibition does not evolve despite extended treatment—validates the application of social evolution theory to antimicrobial development [96] [76]. The observed adaptations to unintended side effects highlight the critical importance of compound specificity and provide a roadmap for refining anti-virulence agents through chemical modification [96].
Future research should focus on several key areas:
- Clinical Translation: Evaluating efficacy in more complex, in vivo biofilm models that better replicate host environments.
- Combination Therapies: Exploring synergistic effects of EPS inhibitors with conventional antibiotics to enhance eradication while minimizing resistance selection.
- Spectrum Expansion: Investigating EPS inhibition in other clinically relevant biofilm-forming pathogens.
- Formulation Development: Optimizing delivery systems for biofilm penetration in various clinical contexts.
Targeting EPS as a public good represents a paradigm shift in anti-biofilm strategy, moving from lethal pressure that selects for resistance to disarming pathogens while leveraging bacterial social dynamics to suppress resistance evolution. This approach holds significant promise for addressing the escalating crisis of antimicrobial resistance.
Within the context of biofilm research, extracellular polymeric substances (EPS) are widely recognized as the fundamental architectural component that encapsulates microbial cells, forming a protective "house" or "fortress" that shields the resident bacteria from antimicrobial agents and host immune responses [8]. The EPS matrix is a complex, dynamic, and highly hydrated mixture of biopolymers, primarily consisting of polysaccharides, proteins, lipids, and surprisingly large amounts of extracellular DNA (e-DNA) [8] [3]. This matrix is not merely a physical barrier; it determines the immediate conditions of life for biofilm cells by affecting porosity, density, water content, charge, sorption properties, and mechanical stability [8]. The intricate interactions between these EPS components confer upon biofilms a remarkable resistance to antibiotics, which can be 10 to 1000 times greater than that of their planktonic counterparts [98] [61].
Targeting the EPS matrix has emerged as a promising therapeutic strategy to sensitize biofilms to conventional antibiotics and physical disruption. The primary anti-EPS agents can be categorized into three distinct classes: enzymes that degrade specific matrix components, small molecules that inhibit matrix function or assembly, and nanocarriers that disrupt the matrix structure while delivering antimicrobial payloads. This review provides a comparative analysis of these three strategic approaches, focusing on their mechanisms of action, experimental applications, and relative efficacies to guide researchers and drug development professionals in the design of novel anti-biofilm therapeutics.
Enzyme-Based Anti-EPS Agents
Mechanisms of Action and Key Enzymes
Enzyme-based strategies function by catalytically degrading key structural components of the EPS matrix. The enzymatic hydrolysis of biopolymers disrupts the matrix's physical integrity, leading to biofilm destabilization and enhanced penetration of co-administered antimicrobials [3].
Table 1: Key Enzymes Used for EPS Disruption
| Enzyme Class | Specific Examples | EPS Target Component | Observed Effect on Biofilm |
|---|---|---|---|
| Protease | Serratiopeptidase, Savinase, Subtilisin A [3] | Proteins (e.g., curli fibers, amyloid adhesins) [8] | Detachment of P. aeruginosa and S. aureus biofilms; reduction of sessile biomass by ≥70% with Savinase [3]. |
| Glycosidase | α-Amylase, Hyaluronidase (HAS) [98] [3] | Polysaccharides (e.g., hyaluronic acid, starch-like polymers) | Concentration-dependent inhibition and detachment of S. aureus biofilms; triggers release from nanocarriers [98] [3]. |
| Nuclease | DNase I [8] | Extracellular DNA (e-DNA) | Disruption of structural grid-like networks critical for the stability of P. aeruginosa and other biofilms [8]. |
Detailed Experimental Protocol: Protease-Mediated Biofilm Disruption
The following protocol is adapted from studies evaluating serine proteases against P. aeruginosa and S. aureus biofilms [3].
Objective: To quantify the efficacy of a protease in disrupting pre-established bacterial biofilms in vitro.
Materials:
- Test Organisms: Pseudomonas aeruginosa (e.g., strain PAO1) and Staphylococcus aureus (e.g., strain ATCC 25923).
- Growth Medium: Tryptic Soy Broth (TSB).
- Enzyme Solution: Serratiopeptidase or Savinase prepared in a suitable buffer (e.g., 50 mM Tris-HCl, pH 7.5). The activity should be confirmed and standardized (e.g., 10 U/mL).
- Control: Buffer-only control (no enzyme).
- Equipment: 96-well polystyrene microtiter plates, sterile pipettes, incubator, microplate reader, crystal violet stain, acetic acid (33%).
Methodology:
- Biofilm Formation: Grow overnight cultures of test organisms. Dilute 1:100 in fresh TSB. Dispense 200 µL aliquots into the wells of a 96-well plate. Incubate statically for 24-48 hours at 37°C to allow biofilm formation.
- Treatment: Carefully aspirate the planktonic culture from the wells. Wash the adhered biofilm gently twice with phosphate-buffered saline (PBS). Add 200 µL of the enzyme solution to the test wells and 200 µL of buffer to the control wells.
- Incubation: Incubate the plate for 24 hours at 37°C.
- Biomass Quantification (Crystal Violet Assay):
- Aspirate the treatment solutions and wash the wells gently with PBS.
- Fix the biofilms by adding 200 µL of methanol per well for 15 minutes.
- Discard methanol and allow plates to air dry.
- Stain with 200 µL of 0.1% crystal violet solution for 5 minutes.
- Wash extensively with water to remove unbound stain.
- Elute the bound stain with 200 µL of 33% acetic acid.
- Transfer 100 µL of the eluent to a new plate and measure the absorbance at 595 nm.
- Data Analysis: Compare the average absorbance of treated wells to control wells. A reduction of ≥70% in biomass indicates high efficacy [3].
Small Molecule-Based Anti-EPS Agents
Mechanisms of Action
Small molecules interfere with EPS structure and function through non-catalytic mechanisms, including epitope alteration, inhibition of matrix assembly, and disruption of component interactions. A key discovery is that small molecules can unexpectedly inhibit or enhance antibody-epitope interactions, altering the immunological and structural landscape of the biofilm [99].
Competitive Inhibition: Small molecules like malonate, which structurally resembles succinate, act as competitive inhibitors of enzymes like succinate dehydrogenase, disrupting bacterial metabolism and, by extension, EPS production [100] [101]. This inhibition is characterized by an increase in the apparent Michaelis constant (Km) with no change in the maximum reaction rate (Vmax) [102].
Epitope Alteration: The SPEED (Screening Platform based on Epitope Alteration for Drug Discovery) technology leverages the ability of small molecules to alter the charge and hydrophobicity of protein epitopes. For instance, 12-crown-4 and orange G can shield the positively charged ε-amino group of lysine (K16) in amyloid-beta (Aβ) peptides, thereby inhibiting antibody recognition—a principle applicable to biofilm EPS proteins [99].
Table 2: Small Molecule Anti-EPS Agents and Their Targets
| Small Molecule | Target/Mechanism | Experimental Outcome |
|---|---|---|
| Malonate [100] | Competitively inhibits succinate dehydrogenase [100]. | Reduces metabolic activity, potentially impairing EPS production. |
| 12-Crown-4 & Orange G [99] | Alters charge/hydrophobicity of lysine residues in epitopes [99]. | Inhibits antibody binding to target proteins, confirmed via zeta potential and dot-blot assays. |
| CRANAD-17 [99] | Enhances antibody binding via stable triplex formation. | Modulates immunological recognition of EPS components. |
| GNF5837 [99] | Inhibits Aβ aggregation (identified via SPEED screening). | Shows potential for inhibiting amyloid-like protein aggregation in EPS. |
Detailed Experimental Protocol: SPEED for Identifying EPS-Targeting Small Molecules
This protocol outlines a label-free method to discover small molecules that alter antibody-epitope interactions, which can be adapted for biofilm EPS targets [99].
Objective: To identify small molecules that inhibit or enhance the binding of a specific antibody to its EPS epitope target.
Materials:
- Target Antigen: Purified EPS component (e.g., a specific biofilm protein such as the TasA amyloid protein from Bacillus subtilis or a polysaccharide antigen).
- Antibody: Monoclonal antibody with known specificity for the target EPS epitope.
- Small Molecule Library: A curated collection of 1,000+ compounds.
- Assay Plates: 96-well or 384-well plates with a high-binding surface.
- Detection Reagents: HRP-conjugated secondary antibody, chemiluminescent substrate.
- Equipment: Plate washer, microplate reader capable of luminescence detection.
Methodology:
- Antigen Immobilization: Coat the assay plates with the purified EPS antigen (e.g., 100 µL/well at 1-5 µg/mL in carbonate-bicarbonate buffer). Incubate overnight at 4°C.
- Blocking: Wash plates 3x with PBS containing 0.05% Tween 20 (PBST). Block with 200 µL/well of 3-5% BSA in PBS for 1-2 hours at room temperature.
- Small Molecule Incubation: Pre-mix the primary antibody (at a fixed, sub-saturating concentration) with individual compounds from the library (at a final concentration of 10-50 µM) in a separate plate. Include controls (antibody only, no antibody).
- Binding Reaction: Wash the coated and blocked plate. Transfer the antibody-compound mixtures to the plate. Incubate for 1-2 hours at room temperature.
- Detection: Wash the plate thoroughly. Add the HRP-conjugated secondary antibody. Incubate, wash again, and add the chemiluminescent substrate. Measure the luminescence signal immediately.
- Data Analysis: Calculate the percentage of signal inhibition or enhancement relative to the antibody-only control. Hits are typically defined as compounds causing >50% inhibition or >150% enhancement of the signal [99].
Signaling Pathway and Workflow Diagram
The following diagram illustrates the logical workflow and mechanistic pathways involved in the SPEED screening platform.
Nanocarrier-Based Anti-EPS Agents
Mechanisms of Action and Nanocarrier Types
Nanocarriers represent a sophisticated, multi-functional approach to biofilm eradication. Their efficacy stems from their ability to penetrate the EPS matrix and respond to the unique biofilm microenvironment with targeted drug release and synergistic physical disruption [98] [61].
Key mechanisms include:
- Physical Disruption: Nanoparticles, especially those with catalytic or photothermal properties, can generate reactive oxygen species (ROS) or local heat, which directly damages EPS components like proteins and e-DNA [98].
- Stimuli-Responsive Drug Release: Nanocarriers can be engineered to release their encapsulated antimicrobial payload (e.g., antibiotics, enzymes) in response to specific stimuli found in the biofilm microenvironment, such as enzymes, low pH, high levels of hydrogen peroxide (H₂O₂), or glutathione (GSH) [98].
- Enhanced Permeation and Retention (EPR): Their small size (typically 10-100 nm) allows them to diffuse through water channels in the biofilm structure and accumulate within the matrix [103] [98].
Table 3: Classes and Examples of Anti-Biofilm Nanocarriers
| Nanocarrier Class | Composition | Stimulus/Mechanism | Anti-Biofilm Action |
|---|---|---|---|
| Lipid-Based [103] | Liposomal Doxorubicin (Doxil) [103]. | EPR effect; fusion with lipid membranes. | Primarily acts on cells; can be adapted for EPS disruption. |
| Polymer-Based [103] [98] | PEG-PLA (Genexol-PM), HA-capped mesoporous silica [103] [98]. | Enzyme (e.g., Hyaluronidase) degradation of capsule [98]. | Targeted release of antibiotics (e.g., vancomycin) and ROS-generating agents (e.g., Ascorbic Acid) [98]. |
| Metal/Metal Oxide [61] | Fe₃O₄ (MNPs), Ag (Silver NPs), Ru (Ruthenium NPs) [98]. | Catalytic activity (peroxidase-like), photothermal, magnetic guidance [98]. | ROS generation (·OH), physical disruption of matrix, hyperthermia [98] [61]. |
Detailed Experimental Protocol: Enzyme-Responsive Nanocarrier for Biofilm Treatment
This protocol is based on the construction and testing of a hyaluronic acid (HA)-capped, enzyme-responsive nanosystem [98].
Objective: To synthesize and evaluate the efficacy of a HAS-responsive nanocarrier for combined chemo-photothermal therapy against biofilms.
Materials:
- Core Nanoparticle: Graphene-mesoporous silica nanosheet (GS) or Mesoporous Ruthenium nanoparticles (Ru NPs) [98].
- Capping Agent: Hyaluronic Acid-Dopamine conjugate (HA-DA).
- Active Agents: Ascorbic acid (AA, a prodrug for H₂O₂), Vancomycin (Van).
- Catalyst/Modifier: Peroxidase-like ferromagnetic nanoparticles (MNPs, Fe₃O₄) or Molybdenum disulfide (MoS₂).
- Test Organism: Staphylococcus aureus biofilm.
Methodology:
- Nanocarrier Synthesis (AA@GS@HA-MNPs):
- Load the core GS or Ru NPs with Ascorbic Acid (AA).
- Coat the loaded NPs with the HA-DA conjugate to form a shell that blocks premature release.
- Immobilize Vancomycin-loaded MNPs or MoS₂ on the outer surface.
- Biofilm Treatment and Triggered Release:
- Grow a 24-hour S. aureus biofilm in a culture plate.
- Incubate the biofilm with the AA@GS@HA-MNPs nanocarrier suspension.
- The hyaluronidase (HAS) secreted by the bacteria degrades the HA shell, releasing the encapsulated AA.
- The released AA is catalyzed by the peroxidase-like MNPs/MoS₂ to generate highly cytotoxic hydroxyl radicals (·OH).
- Synergistic Photothermal Therapy:
- Expose the treated biofilm to near-infrared (NIR) laser light.
- The graphene or Ru NPs absorb the light and generate local heat, further disrupting the EPS matrix and killing embedded bacteria.
- Efficacy Assessment:
- Quantify biofilm biomass using the crystal violet assay.
- Determine bacterial viability via colony-forming unit (CFU) counts.
- Visualize biofilm architecture and disruption using confocal laser scanning microscopy (CLSM) with live/dead staining.
Nanocarrier Mechanism of Action Diagram
The following diagram visualizes the multi-step mechanism of a stimuli-responsive nanocarrier for biofilm eradication.
The Scientist's Toolkit: Key Research Reagents and Materials
Table 4: Essential Reagents for Anti-EPS Research
| Reagent/Material | Function in Research | Example Application |
|---|---|---|
| Recombinant Hyaluronidase (HAS) [98] | Triggers degradation of HA-based nanocarrier shells. | Studying enzyme-responsive drug release in HAS-producing biofilms (e.g., S. aureus) [98]. |
| Serratiopeptidase/Savinase [3] | Proteolytic enzyme for degrading proteinaceous EPS components. | Evaluating biofilm detachment and synergy with antibiotics [3]. |
| DNase I [8] | Degrades extracellular DNA (e-DNA) in the matrix. | Disrupting structural integrity of e-DNA dependent biofilms (e.g., P. aeruginosa) [8]. |
| Crystal Violet [3] | Dye for staining and quantifying total adherent biofilm biomass. | Standard assay for measuring biofilm formation and disruption after treatment [3]. |
| 12-Crown-4 / Orange G [99] | Small molecule model compounds for epitope alteration. | Probing charge-based interactions in EPS components using the SPEED platform [99]. |
| Hyaluronic Acid (HA) [98] | Natural polymer for constructing enzyme-responsive nanocarriers. | Used as a capping agent for targeted drug delivery to biofilms [98]. |
| Ascorbic Acid (AA) [98] | Prodrug that generates hydroxyl radicals under catalysis. | Served as an anti-EPS agent in catalytic nanocarriers for chemical disruption [98]. |
The battle against biofilm-mediated infections necessitates innovative strategies that target the protective EPS matrix. Enzymes, small molecules, and nanocarriers each offer distinct advantages and face specific challenges. Enzymes provide high specificity and catalytic power but can be sensitive to microenvironmental conditions and are often large molecules with limited penetration. Small molecules offer excellent penetration and the potential for oral bioavailability, but their discovery is complex and their effects can be transient. Nanocarriers represent the most versatile platform, capable of integrating multiple functionalities—including penetration, sensing, and controlled release of combined therapies—thereby enabling a powerful, multi-pronged attack on the biofilm fortress.
The future of anti-EPS therapy lies in the intelligent combination of these approaches. Examples include nanocarriers that deliver synergistic cocktails of enzymes and small molecules, or small molecules that sensitize the biofilm to subsequently administered nanomaterials. As our understanding of the dynamic and heterogeneous nature of the EPS matrix deepens, the next generation of anti-biofilm agents will need to be equally adaptive and multifaceted to effectively dismantle this resilient microbial citadel.
The escalating crisis of antimicrobial resistance has necessitated the exploration of novel therapeutic strategies that impose lower selective pressure for resistance. Among these, anti-virulence drugs targeting public goods—virulence factors that benefit all cells in a proximal population—represent a promising evolution-proof approach. This whitepaper provides a comprehensive technical assessment of the low propensity for resistance development to public good-targeting inhibitors, with specific focus on extracellular polymeric substances (EPS) in bacterial biofilms. We synthesize contemporary research findings, present quantitative experimental data, detail essential methodologies, and visualize the underlying conceptual frameworks to equip researchers and drug development professionals with the tools to advance this emerging field.
Bacterial biofilms are structured communities of microbial cells encased in a self-produced matrix of EPS, which provides structural integrity and protection against environmental stresses, including antimicrobial agents [1]. The EPS matrix is primarily composed of polysaccharides, proteins, nucleic acids, and lipids [3] [2]. This matrix functions as a public good because it provides community-wide benefits—including enhanced tolerance to antibiotics and host immune responses—regardless of which individual cells contribute to its production [96] [104].
Traditional antibiotics target essential bacterial functions, creating strong directional selection for resistant mutants that can proliferate and dominate the population. In contrast, public good-targeting inhibitors disrupt traits that are non-essential for individual cell survival but beneficial for the collective [105]. According to social evolution theory, resistant mutants that resume production of a public good bear the metabolic cost of production while sharing the benefits with surrounding non-producing, sensitive "cheater" cells. This creates a fitness disadvantage for resistant mutants, leading to their counter-selection within the population [96] [105]. This theoretical framework predicts that targeting public goods with inhibitors can lead to evolutionarily robust antimicrobial strategies with low propensity for resistance development.
Theoretical Foundation: Social Dynamics of Public Goods
Conceptual Framework of Public Good-Mediated Resistance
The diagram below illustrates why resistance to public good inhibitors is evolutionarily disadvantaged. In an untreated population, producer and non-producer cells coexist. When a public good inhibitor is applied, the entire population suffers from the loss of the public good. If a resistant mutant emerges, it resumes production of the public good at a personal cost, but the benefits are shared with sensitive cheaters. These cheaters, free from the metabolic cost of production, outcompete the resistant mutants, thereby preventing resistance from spreading.
Key Public Goods as Antimicrobial Targets
Various bacterial traits function as public goods and represent potential targets for evolution-proof inhibitors, as summarized in the table below.
Table 1: Public Goods as Targets for Evolution-Proof Inhibitors
| Public Good Category | Specific Examples | Function in Bacterial Community | Exploitability for Control |
|---|---|---|---|
| Biofilm Matrix Components | Curli fimbriae, cellulose, other exopolysaccharides [96] | Structural integrity, adhesion, protection [1] [2] | High (Non-producers integrate into and benefit from matrix) [96] |
| Nutrient Scavenging Systems | Siderophores (e.g., pyoverdine), extracellular enzymes [104] [105] | Iron acquisition, digestion of complex molecules [104] | Moderate to High (Shared nutrient access) [105] |
| Detoxification Mechanisms | β-lactamases, catalases, peroxidases [104] [105] | Antibiotic degradation, oxidative stress resistance [104] | High (Community-wide protection) [105] |
| Quorum Sensing Signals | Acyl-homoserine lactones, autoinducing peptides [105] | Coordination of population-wide gene expression [1] | Variable (Signal molecules are public, but regulated traits may not be) [105] |
Experimental Evidence: Assessing Resistance Development
Case Study: EPS Inhibition in Salmonella Typhimurium
A pivotal study investigating long-term treatment of Salmonella Typhimurium with a 2-aminoimidazole-based EPS inhibitor (RC41) provides compelling experimental evidence for low resistance propensity [96]. The key findings and quantitative data from this investigation are summarized below.
Table 2: Quantitative Assessment of Salmonella Biofilm Response to Long-term EPS Inhibition
| Experimental Parameter | Ancestral Population (Untreated) | RC41-Evolved Populations (After 20 Cycles) | Control-Evolved Populations (No Inhibitor) |
|---|---|---|---|
| Biofilm Biomass Inhibition at 50 µM RC41 | ~77% reduction | ~73% reduction (Remained susceptible) | Not Applicable |
| Absolute Biofilm Biomass under RC41 Treatment | 0.073 OD₅₇₀ₙₘ | 0.047 OD₅₇₀ₙₘ (Remained low) | 0.026 OD₅₇₀ₙₘ |
| Planktonic Biomass under RC41 Treatment | No significant reduction | No significant reduction | No significant reduction |
| Average Genomic Mutations | Baseline | ~10 mutations/population | ~5 mutations/population |
| Common Adaptive Mutations | None | ramR (efflux pump regulator), rpoS (sigma factor) | Adaptations to biofilm setup, not inhibitor-specific |
This study demonstrated that no resistance to biofilm inhibition emerged after 40 days (20 cycles) of continuous treatment [96]. Despite this, genomic analysis revealed that populations did adapt through mutations that increased efflux pump activity, likely in response to a growth-delaying side effect of the inhibitor at higher concentrations. Crucially, these adaptations did not restore EPS production capacity during treatment [96].
Experimental Protocol: Serial Passage Evolution Assay
The following workflow details the methodology for assessing resistance development against public good inhibitors, adapted from the foundational Salmonella study [96].
Key Technical Considerations:
- Growth Conditions: Biofilms were grown on the bottom of Petri dishes with diluted TSB broth to simulate nutrient-limiting industrial surfaces [96].
- Inhibitor Concentration: Use a sub-inhibitory concentration that robustly inhibits the public good (e.g., biofilm formation) without affecting planktonic growth, to minimize selection pressure from side effects [96].
- Control Lineages: Maintain parallel lineages passaged without inhibitor to distinguish general adaptations to laboratory conditions from inhibitor-specific adaptations [96].
- Resistance Assessment: Define resistance specifically as the ability to resume production of the public good (e.g., form biofilms) during inhibitor treatment [96].
Methodological Toolkit for Resistance Assessment
Essential Research Reagents and Solutions
Table 3: Key Reagent Solutions for Public Good Inhibition Studies
| Reagent/Solution | Composition/Example | Function in Experimental Protocol |
|---|---|---|
| EPS Inhibitors | 5-aryl-2-aminoimidazoles (e.g., RC41: 5-(4-chlorophenyl)-N-cyclopentyl-1H-imidazol-2-amine) [96] | Target CsgD transcriptional activator to repress curli and cellulose production [96] |
| Biofilm Growth Media | Diluted Tryptic Soy Broth (TSB) | Mimics nutrient-limited conditions, promotes biofilm formation over planktonic growth [96] |
| Enzymatic Matrix Disruption Agents | Proteases (Savinase, Subtilisin A), α-amylase, DNase I [3] [5] | Selectively degrade protein, polysaccharide, or eDNA components of EPS to study matrix function and resistance [3] |
| Whole-Genome Sequencing Kits | Commercial next-generation sequencing platforms | Identify mutations in evolved populations and distinguish inhibitor-specific adaptations [96] |
| Competition Assay Media | Fresh broth with/without inhibitor | Quantify fitness of putative resistant mutants against sensitive strains in presence and absence of inhibitor [96] [105] |
Advanced Analytical Techniques
Image Cytometry for Biofilm Architecture Analysis: Tools like BiofilmQ enable automated, high-throughput quantification of 3D biofilm properties from fluorescence images [33]. The software can segment biofilm biovolume and quantify hundreds of structural and fluorescence parameters, including volume, surface area, roughness, and spatial distribution of fluorescent reporters for matrix components [33].
Fourier Transform Infrared (FT-IR) Spectroscopy: FT-IR provides non-destructive analysis of EPS biochemical composition by detecting characteristic absorption spectra for functional groups in proteins (1500–1800 cm⁻¹), polysaccharides, and nucleic acids (900–1250 cm⁻¹) [3]. This allows monitoring of compositional changes in response to inhibitor treatment.
Targeting public goods such as EPS represents a paradigm shift in antimicrobial strategy, moving from bactericidal agents that impose strong selection for resistance to anti-virulence approaches that exploit social evolutionary constraints. Experimental evidence confirms that inhibitors targeting public goods can exhibit remarkably low propensity for resistance development, as resistant mutants are evolutionarily disadvantaged against non-producing cheaters [96] [105].
Future research directions should focus on optimizing inhibitor specificity to minimize side effects that might otherwise select for unintended resistance mechanisms [96], exploring combination therapies that simultaneously target multiple public goods and developing standardized resistance assessment protocols that adequately capture social evolutionary dynamics. The integration of advanced analytical tools, including machine learning and high-throughput image cytometry, will further accelerate the development of evolution-proof antimicrobial strategies capable of addressing the growing crisis of antimicrobial resistance [33] [41].
The targeted disruption of the extracellular polymeric substance (EPS) matrix represents a promising frontier in the fight against resilient biofilm-associated infections. This whitepaper synthesizes current research to elucidate the complex metabolic adaptations biofilms employ to survive such targeted assaults. EPS-targeting strategies, including enzymatic degradation, quorum-sensing inhibition, and nanomaterial penetration, inevitably impose selective pressures that trigger profound functional shifts within the biofilm community. These adaptations often manifest as increased phenotypic heterogeneity, a heightened prevalence of dormant persister cells, and a recalibration of energy metabolism towards stress response and matrix regeneration. Understanding these evasion tactics is critical for developing robust, multi-targeted therapeutic strategies that can outmaneuver biofilm resilience and prevent regrowth, ultimately translating into improved clinical outcomes for conditions ranging from medical device-related infections to chronic wounds.
The extracellular polymeric substance (EPS) is the fundamental architectural component of the biofilm phenotype, constituting 50% to 90% of its total organic matter [2] [42]. This self-produced, three-dimensional matrix is a gel-like, highly hydrated network composed primarily of polysaccharides, proteins, lipids, and extracellular DNA (eDNA) [3] [1] [2]. The EPS establishes the functional and structural integrity of biofilms, serving as a protective barrier, a platform for cell-to-cell interactions, and a mediator of adhesion to surfaces [2] [42]. Its role in fostering antimicrobial resistance is multifaceted, providing a diffusion barrier to antimicrobial agents, facilitating metabolic dormancy in nutrient-poor regions, and enabling quorum sensing for community-wide coordination [106] [1].
Targeting the EPS is a logical therapeutic strategy aimed at dismantling this protective fortress. The underlying hypothesis is that by compromising the structural and functional integrity of the matrix, the embedded microbial cells become more susceptible to antimicrobial agents and host immune responses [106] [3]. However, biofilms are not static entities; they are dynamic, adaptive biological systems. The application of an EPS-targeted treatment acts as a powerful selective pressure, provoking a cascade of metabolic and phenotypic adjustments designed to ensure community survival. This whitepaper delves into the specific nature of these functional shifts, exploring how biofilms rewire their metabolism, alter their community structure, and activate emergency response programs to endure and recover from EPS-directed attacks. Framed within the broader context of EPS function in biofilm research, this analysis provides a critical roadmap for anticipating biofilm resilience and designing next-generation anti-biofilm therapeutics.
The Composition and Function of the EPS Matrix
A detailed understanding of the EPS matrix is a prerequisite for analyzing the metabolic consequences of its disruption. The EPS is not a simple, inert scaffold but a complex, dynamic, and functionally diverse compartment that is central to biofilm pathophysiology.
Table 1: Major Components of the Extracellular Polymeric Substance (EPS) Matrix and Their Functions
| EPS Component | Primary Composition | Key Functional Roles in Biofilm |
|---|---|---|
| Exopolysaccharides | Heteropolymers of monosaccharides (e.g., galactose, glucose, xylose) with non-carbohydrate substituents (acetate, pyruvate, succinate) [2]. | Provides structural integrity, mediates initial adhesion, forms a diffusion-limiting barrier, and contributes to viscoelasticity [3] [2]. |
| Proteins | Diverse array of enzymes (exoenzymes), structural proteins, and amyloid fibers [3] [2]. | Facilitates nutrient acquisition (degradation of macromolecules), provides structural stability, and can inactivate antimicrobial agents [3]. |
| Extracellular DNA (eDNA) | DNA released from lysed bacterial cells [3] [1]. | Contributes to matrix stability through electrostatic interactions, facilitates horizontal gene transfer, and can bind cationic antimicrobials [3] [1]. |
| Lipids and Surfactants | Various lipid compounds and amphiphilic molecules [106]. | Aids in surface motility, dispersion of cells from the biofilm, and may alter permeability to antimicrobials [106]. |
| Minerals | Biomineralized ions such as calcium (e.g., calcite) and magnesium [2]. | Enhances structural integrity, acts as a scaffold, and contributes to resistance against shear forces and chemicals [2]. |
The composition of the EPS is highly variable, influenced by microbial species, environmental conditions, and available nutrients. This heterogeneity is key to the matrix's multifunctionality. The matrix ensures cohesion and viscoelasticity, allowing the biofilm to withstand mechanical stresses [3]. It also functions as an external digestive system through exoenzymes that break down complex nutrients into absorbable subunits [2]. Furthermore, the EPS provides a protective niche by binding antimicrobial agents, slowing their diffusion, and creating gradients of nutrients and oxygen that lead to metabolic and phenotypic heterogeneity [106] [1]. This heterogeneity is a cornerstone of biofilm resilience, as subpopulations of cells in different metabolic states can survive environmental insults that would kill a uniform population.
Established EPS-Targeting Treatment Modalities
The strategic disruption of the EPS matrix can be achieved through several mechanisms, each with a distinct mode of action. The following section outlines key experimental protocols for EPS-targeting approaches.
Enzymatic Degradation of Matrix Components
Enzymes represent a highly specific tool for dismantling the EPS by hydrolyzing its core structural polymers.
- Protocol: Application of Hydrolytic Enzymes for Biofilm Disruption
- Enzyme Selection: Choose purified enzymes based on the target EPS component.
- Proteases: Target matrix proteins and amyloid fibers. Savinase and Subtilisin A have shown high efficiency against Pseudomonas aeruginosa and Staphylococcus aureus biofilms, leading to a reduction of up to 70% in sessile biomass [3].
- Glycosidases: Target exopolysaccharides. Alpha-amylase has been demonstrated to inhibit biofilm formation and detach S. aureus biofilms in a concentration- and time-dependent manner [3].
- DNases: Target extracellular DNA (eDNA). Effective against biofilms where eDNA is a critical structural component [3].
- Biofilm Cultivation: Grow biofilms in standard in vitro systems (e.g., 96-well plates, flow cells) for 24-48 hours to establish mature structures.
- Enzyme Treatment: Expose biofilms to the enzyme(s) dissolved in an appropriate buffer (e.g., PBS) at a concentration range of 1-100 U/mL for 24 hours [3]. Include controls with heat-inactivated enzymes to confirm that the effects are due to enzymatic activity.
- Efficacy Assessment:
- Biomass Quantification: Use crystal violet staining to measure total remaining biomass [106].
- Viability Assessment: Perform colony-forming unit (CFU) counts after biofilm disruption to quantify viable cells [106].
- Structural Analysis: Utilize confocal laser scanning microscopy (CLSM) with live/dead staining to visualize architectural disintegration and determine the ratio of live to dead cells [106].
- Enzyme Selection: Choose purified enzymes based on the target EPS component.
Quorum-Sensing Inhibition (QSI)
Quorum-sensing antagonists do not directly degrade the matrix but interfere with the cell-to-cell communication system that regulates its production.
- Protocol: Evaluating Quorum-Sensing Antagonists
- QSI Agent Preparation: Prepare stock solutions of quorum-sensing inhibitors (e.g., natural or synthetic molecules that block acyl-homoserine lactone or autoinducer-2 receptors) in a suitable solvent like DMSO.
- Co-incubation and Treatment Models:
- Preventive Model: Add the QSI agent to the growth medium at the beginning of biofilm cultivation to assess its ability to prevent biofilm formation.
- Treatment Model: Add the QSI agent to established, mature biofilms (e.g., 24-hour-old) to evaluate its disruptive potential.
- Downstream Analysis:
- EPS Quantification: Measure the total EPS production, specifically exopolysaccharides and proteins, using colorimetric methods (e.g., phenol-sulfuric acid for sugars, Lowry or BCA assay for proteins) [3] [42].
- Gene Expression: Quantify the expression of key QS-regulated genes (e.g., lasI, rhlI in P. aeruginosa) and EPS synthesis genes using RT-qPCR to confirm pathway disruption [106].
- Biofilm Phenotype: Assess the biofilm architecture and biomass as described in section 3.1.
Nanotechnology-Based Drug Delivery
Nanocarriers can be engineered to penetrate the EPS and deliver high local concentrations of antimicrobials or enzymes directly to the biofilm core.
- Protocol: Utilizing Enzyme-Functionalized Nanocarriers
- Nanocarrier Synthesis: Fabricate nanocarriers (e.g., liposomes, polymeric nanoparticles) from materials such as poly(lactic-co-glycolic acid) (PLGA) or chitosan.
- Functionalization: Immobilize or encapsulate anti-biofilm agents onto or within the nanocarriers. This can include:
- Enzymes: Co-loading of proteases and glycosidases for synergistic EPS degradation [106].
- Antimicrobials: Loading antibiotics to be released upon matrix penetration.
- Biofilm Exposure: Incubate mature biofilms with the functionalized nanocarriers for a defined period.
- Evaluation of Penetration and Efficacy:
- Penetration Analysis: Use fluorescently labeled nanocarriers and track their movement through the biofilm depth using CLSM [106].
- Synergistic Effect Assessment: Compare the reduction in biofilm viability and biomass between free drugs, non-functionalized nanocarriers, and enzyme-functionalized nanocarriers to demonstrate enhanced efficacy [106].
Diagram 1: Modalities of EPS-targeting treatments, showing the three main strategic approaches and their specific implementations.
Metabolic and Phenotypic Adaptations to EPS Stress
When the integrity of the EPS matrix is compromised, biofilms initiate a complex, coordinated stress response that drives several key functional shifts to maintain resilience.
Shift Towards Metabolic Heterogeneity and Dormancy
A primary adaptation is the rapid diversification of metabolic activity within the community, favoring the emergence of dormant, tolerant subpopulations.
- Activation of Stress Responses: The physical and chemical damage to the EPS is sensed by bacteria, triggering stringent response and general stress response pathways. This leads to the upregulation of stress response genes, such as those encoding chaperones and DNA repair enzymes, reallocating energy away from growth and division towards maintenance and survival [106] [107].
- Increased Persister Cell Formation: Nutrient limitation exacerbated by EPS disruption, particularly in the deeper layers of the biofilm, promotes a shift to a non-growing or slow-growing state. Mathematical models that couple nutrient transport with biofilm dynamics confirm that nutrient-poor conditions produce a high and sustained proportion of persister cells [108]. These tolerant cells are characterized by low metabolic activity, making them insensitive to conventional antibiotics that target active cellular processes [106] [108].
- Altered Energy Metabolism: In response to EPS damage, biofilms may shift their central carbon metabolism. For instance, there can be an increased flux through pathways that generate precursor molecules for EPS component synthesis, such as the pentose phosphate pathway for nucleotide and exopolysaccharide production [107]. This metabolic rewiring prioritizes matrix repair over cellular proliferation.
Enhanced Efflux and Detoxification Mechanisms
Surviving cells within a compromised biofilm actively work to expel or neutralize toxic compounds.
- Upregulation of Efflux Pumps: Genes encoding multidrug efflux pumps (e.g., MexAB-OprM in P. aeruginosa) are often overexpressed in biofilm cells compared to their planktonic counterparts [106] [1]. EPS-targeting treatments that stress the cells can further induce this expression, actively reducing the intracellular concentration of antimicrobials that manage to penetrate the weakened matrix.
- Secretion of Protective Enzymes: Biofilms may increase the production of extracellular enzymes that modify or degrade incoming threats. For example, the secretion of beta-lactamases into the remaining EPS matrix can inactivate beta-lactam antibiotics before they reach their cellular targets [1].
Active Matrix Remodeling and Regeneration
Perhaps the most direct countermeasure is the initiation of programs to repair and rebuild the damaged EPS scaffold.
- Precursor Scavenging: The degradation of the existing matrix by external enzymes can, paradoxically, provide a source of nutrients. Bacteria may uptake the released sugars, amino acids, and nucleotides to fuel the de novo synthesis of EPS components, effectively recycling the building blocks [3] [2].
- Synthesis of Alternative EPS Components: Under sustained pressure from a specific enzyme (e.g., a protease), the biofilm community may adapt by altering the compositional profile of the EPS it produces. For instance, it might increase the relative proportion of polysaccharides or eDNA, effectively changing the matrix's chemical nature to evade the specific hydrolytic activity of the treatment [3].
- Activation of Quorum-Sensing Dependent Repair: While QS inhibitors can blunt this response, residual communication can still trigger density-dependent expression of EPS synthesis genes once a critical cell density is re-established, driving the regeneration of the matrix after the initial insult [106] [1].
Diagram 2: Biofilm adaptive responses to EPS-targeting treatments, showing the three core strategic shifts and their specific metabolic and phenotypic manifestations.
Table 2: Summary of Biofilm Adaptive Responses to EPS-Targeted Treatments
| Adaptive Response | Key Metabolic/Phenotypic Shift | Potential Consequence for Treatment |
|---|---|---|
| Metabolic Heterogeneity & Dormancy | Increased stringent response; formation of nutrient-gradient dependent persister cells; shift to anaerobic respiration [106] [108]. | Reduced efficacy of conventional antibiotics; requires agents targeting dormant cells. |
| Enhanced Efflux & Detoxification | Upregulation of multidrug efflux pump genes (e.g., mexAB-oprM); secretion of antibiotic-modifying enzymes (e.g., β-lactamases) [106] [1]. | Decreased intracellular drug concentration; enzymatic inactivation of therapeutics. |
| Matrix Remodeling & Regeneration | Altered EPS composition to evade specific enzymes; scavenging of degradation products for resynthesis; activation of QS-driven EPS production [106] [3] [2]. | Biofilm regrowth and recurrence post-treatment; requires combination therapy. |
Research Toolkit for Investigating Biofilm Adaptations
Studying these complex functional shifts requires a sophisticated suite of analytical techniques and reagents. The following table details essential tools for probing the metabolic adaptations of biofilms under EPS-targeted stress.
Table 3: Research Reagent Solutions for Studying Biofilm Adaptations
| Research Reagent / Tool | Primary Function in Investigation |
|---|---|
| Hydrolytic Enzymes (Proteases, Glycosidases, DNases) | Used to experimentally induce specific EPS disruption, serving as the initial stressor to study subsequent biofilm adaptation pathways [3]. |
| CRISPR-Cas-Modified Bacteriophages | Enable targeted genetic manipulation within the biofilm community to knock out specific genes involved in stress response or EPS synthesis, validating their function in adaptation [106]. |
| Quorum-Sensing Inhibitors (QSIs) | Tools to dissect the role of cell-to-cell communication in coordinating the community-wide adaptive response to EPS damage [106]. |
| Fluorescent Probes (e.g., LIVE/DEAD stains, FITC-Concanavalin A) | Allow for visualization of cell viability and polysaccharide distribution within the biofilm architecture via confocal microscopy before and after treatment [106]. |
| Metabolomics Platforms (GC-MS, LC-MS) | Used to profile and quantify changes in the complete set of metabolites, revealing metabolic rewiring in response to EPS stress, such as shifts in central carbon metabolism [107]. |
| Transcriptomic Analysis (RNA-Seq) | Provides a global view of gene expression changes, identifying upregulation of efflux pumps, stress response genes, and EPS synthesis operons during adaptation [106] [107]. |
| Microfluidic Biofilm Reactors | Create highly controlled, dynamic fluid environments for growing biofilms that closely mimic in vivo conditions, allowing real-time observation of adaptation under shear stress [106]. |
The experimental workflow for a comprehensive adaptation study would integrate several of these tools. A typical protocol might involve:
- Culturing a mature biofilm in a microfluidic reactor or 96-well plate.
- Treating the biofilm with a specific EPS-targeting agent (e.g., a cocktail of proteases and glycosidases).
- Using transcriptomics (RNA-Seq) and metabolomics (LC-MS) to identify key upregulated pathways in treated vs. control biofilms.
- Validating the role of candidate genes using CRISPR-Cas-based genetic tools.
- Monitoring the spatial organization of the adaptive response over time using fluorescent probes and confocal microscopy.
This multi-faceted approach allows researchers to move from correlation to causation in defining the mechanisms of biofilm resilience.
The metabolic adaptations of biofilms to EPS-targeted treatments are a testament to their remarkable evolutionary resilience. The functional shifts—towards dormancy, efflux, and matrix remodeling—represent a coordinated survival strategy that poses a significant challenge to mono-therapeutic approaches. This analysis underscores that effectively combating biofilms requires a paradigm shift from simply breaking down the matrix to strategically disrupting the biofilm's ability to perceive the threat and mount a successful adaptive response.
Future research must focus on combination therapies that simultaneously target the EPS and core metabolic processes. For example, combining matrix-degrading enzymes with antibiotics that are effective against persister cells, or coupling quorum-sensing inhibitors with agents that block efflux pumps, presents a promising multi-pronged attack [106]. Furthermore, the application of systems biology and AI-driven modeling is crucial to integrate the complex, multi-scale data on biofilm adaptation [106] [109]. These models can help predict tipping points in biofilm resilience and identify novel vulnerabilities. Finally, the development of "smart" therapeutic systems, such as nanocarriers that release their payload in response to biofilm-specific cues like low pH or high enzyme activity, could ensure precise targeting and improve therapeutic efficacy [106]. By anticipating and countering the functional shifts described in this whitepaper, the scientific community can develop more robust and durable solutions to overcome the formidable challenge posed by biofilm-associated infections.
Within the broader context of extracellular polymeric substance (EPS) function in biofilm structure research, understanding bacterial adaptive responses to anti-virulence compounds is paramount. The biofilm matrix, a complex architecture of EPS comprising polysaccharides, proteins, and nucleic acids, serves as a critical barrier that protects microbial communities from environmental insults, including antimicrobial agents [1] [81]. A compelling strategy to combat biofilm-related infections involves the use of compounds that inhibit the production of EPS, thereby disarming the pathogen without directly killing it. This anti-virulence approach is theorized to exert lower selective pressure for resistance compared to conventional antibiotics [96] [110]. However, long-term exposure to such inhibitors can trigger distinct evolutionary pathways. This technical guide delves into the genotypic and phenotypic validation methodologies required to characterize bacterial adaptations, specifically mutations in efflux pump regulators and alternative sigma factors, following treatment with biofilm-inhibiting compounds. These adaptations represent a sophisticated bypass mechanism where bacteria evolve not to resist the anti-biofilm activity itself, but to overcome unintended side effects of the treatment, all within the functional framework of EPS-mediated biofilm formation [96].
Core Concepts: EPS, Biofilms, and Bacterial Adaptation
The Role of EPS in Biofilm Structure and Resistance
The extracellular polymeric substance is the cornerstone of the biofilm matrix, a self-produced hydrated gel that encases microbial cells. Its composition is highly heterogeneous, typically consisting of polysaccharides, proteins, lipids, and extracellular DNA (eDNA) [1] [81]. The EPS matrix is not merely a physical scaffold; it is a functional component that contributes significantly to biofilm resilience through several mechanisms:
- Physical Barrier: The matrix acts as a diffusion barrier, hindering the penetration of antimicrobial agents and protecting embedded cells [81].
- Public Good Character: Many EPS components exhibit a "public good" nature, meaning they provide benefits to all cells in the vicinity, including non-producers. This social dynamic is a key consideration for anti-virulence strategies targeting EPS [96].
Anti-Virulence Strategies Targeting EPS
Anti-virulence drugs represent a paradigm shift from traditional biocidal antibiotics. Instead of targeting essential bacterial processes, they specifically disrupt virulence factors like biofilm formation. Inhibiting EPS production aims to render pathogens susceptible to host immune defenses and other antimicrobials without imposing a strong selective pressure for classical resistance [96] [110]. For instance, 2-aminoimidazole-based (2-AI) compounds have been shown to repress the transcriptional activator CsgD in Salmonella, thereby inhibiting the production of curli fimbriae and cellulose, which are essential for robust biofilm formation [96].
Evolutionary Bypass: Efflux Pumps and Sigma Factors
Prolonged exposure to EPS inhibitors can select for mutations that confer fitness advantages through alternative mechanisms. Two key targets of such adaptations are:
- Efflux Pumps: Multidrug efflux pumps, particularly the Resistance-Nodulation-Division (RND) family like AcrAB-TolC in Enterobacteriaceae, export a wide range of toxic compounds out of the cell. Mutations in regulatory genes (e.g., ramR) can lead to pump overexpression [96] [111].
- Alternative Sigma Factors: Sigma factors, such as RpoS (the stationary phase sigma factor), direct RNA polymerase to specific promoters, reprogramming gene expression in response to stress. Mutations in these global regulators can lead to pleiotropic effects, including changes in biofilm capacity and stress tolerance [96] [110].
The selection for these mutations is often driven by unintended side effects of the primary inhibitor, such as a growth delay, rather than the anti-biofilm activity itself. Consequently, populations adapt by enhancing efflux or altering global gene regulation to mitigate this secondary stress, a process we term "evolutionary bypass" [96].
Experimental Models & Key Findings
A Model Study: Salmonella Typhimurium and 2-AI EPS Inhibitors
An illustrative example comes from a serial passage evolution experiment with Salmonella Typhimurium treated with a 2-AI EPS inhibitor (RC41) [96].
- Experimental Design: Biofilms were grown in diluted TSB for 48-hour cycles, followed by passaging, for a total of 20 cycles (40 days) in the presence of a sub-inhibitory concentration (50 µM) of RC41. This concentration reduced biofilm biomass by ~75% without affecting planktonic growth.
- Key Finding: Despite long-term exposure, no classical resistance to biofilm inhibition was observed; the evolved populations remained as susceptible to EPS inhibition as the ancestral strain.
- Genomic Discovery: Whole-genome sequencing revealed that all three independently evolved lineages accumulated mutations in specific genetic regions, detailed in Table 1.
Table 1: Summary of Mutations Identified in Salmonella after Long-Term EPS Inhibitor Treatment [96]
| Genetic Element | Function | Type of Mutations Identified | Phenotypic Consequence |
|---|---|---|---|
| ramR | Transcriptional repressor of the ramA gene | Nonsense (Trp185*), Missense (Thr50Pro, Leu58Trp), Promoter mutations | Derepression of RamA, leading to overexpression of AcrAB-TolC efflux pump and increased efflux activity. |
| rpoS | Stationary phase/stress response sigma factor | Not specified | Reduced biofilm-forming capacity, altered global gene expression. |
| Components of RNA Polymerase | Core transcriptional machinery | Not specified | Broader changes in transcriptional programs. |
- Phenotypic Validation: The mutations resulted in two primary phenotypic changes:
- Increased Efflux Activity: The ramR mutations led to constitutive overexpression of the AcrAB-TolC efflux system, enabling cells to more efficiently export the inhibitor and overcome its growth-delaying side effect [96].
- Reduced Biofilm Capacity: Mutations in rpoS and RNA polymerase components independently led to a reduced inherent capacity for biofilm formation, an adaptive trade-off in this environment [96].
This model underscores that evolution can favor adaptations to the secondary stress (growth delay) rather than the primary target (EPS production), and these adaptations can be validated through integrated genotypic and phenotypic analyses.
The Virulence Connection of Efflux Pumps
Beyond antibiotic resistance, efflux pumps like AcrAB are established virulence determinants. In Enteroaggregative E. coli (EAEC), a functional AcrAB pump is required for robust biofilm formation, adhesion to host cells, and full virulence in an animal model [111]. This highlights that mutations affecting efflux pumps can have profound implications not only for resistance but also for a pathogen's ability to cause disease.
Methodologies for Genotypic and Phenotypic Validation
A robust validation pipeline requires correlating genomic data with functional phenotypic assays. The following sections outline key protocols.
Genotypic Analysis: Whole-Genome Sequencing (WGS)
Objective: To comprehensively identify single nucleotide polymorphisms (SNPs), insertions, and deletions in evolved bacterial populations. Protocol:
- Genomic DNA Extraction: Extract high-quality, high-molecular-weight genomic DNA from evolved and ancestral control populations using a standardized kit or phenol-chloroform method. Assess DNA purity and quantity via spectrophotometry (e.g., Nanodrop) and fluorometry (e.g., Qubit).
- Library Preparation and Sequencing: Prepare a sequencing library (e.g., Illumina Nextera XT) from the fragmented DNA. Sequence the libraries on a high-throughput platform (e.g., Illumina MiSeq or NovaSeq) to achieve a minimum of 50x coverage.
- Bioinformatic Analysis:
- Quality Control: Use tools like FastQC to assess read quality.
- Alignment: Map sequencing reads to a reference genome (e.g., Salmonella SL1344) using aligners like BWA or Bowtie2.
- Variant Calling: Identify mutations with variant callers such as GATK or SAMtools/BCFtools. Set a frequency threshold (e.g., ≥10%) to filter out low-frequency variants and background mutations.
- Annotation: Annotate identified mutations using tools like SnpEff to determine their functional impact on genes (e.g., missense, nonsense, regulatory) [96].
Phenotypic Analysis: Efflux Pump Activity Assay
Objective: To quantitatively measure the efflux activity of evolved strains compared to the ancestor. Protocol:
- Strain Preparation: Grow test strains to mid-exponential phase in a suitable broth (e.g., LB).
- Dye Accumulation: Incubate the bacterial cells with a fluorescent substrate that is exported by the target efflux pump (e.g., ethidium bromide or Hoechst 33342 for AcrAB). Use a negative control treated with a known efflux pump inhibitor (e.g., Phe-Arg β-naphthylamide, PABN).
- Measurement:
- Fluorometry: Measure fluorescence intensity over time in a plate reader. A decrease in fluorescence accumulation indicates higher efflux activity.
- Flow Cytometry: For single-cell resolution, analyze cells using a flow cytometer. Evolved strains with upregulated efflux will show a lower median fluorescence intensity than the ancestor [96].
- Data Analysis: Normalize fluorescence values to cell density. Compare the rate of dye efflux and the final accumulated dye level between evolved and ancestral strains.
Phenotypic Analysis: Biofilm Formation Capacity
Objective: To assess the impact of mutations on the inherent ability to form biofilms, in both treated and untreated conditions. Protocol (Microtiter Plate Assay):
- Inoculation: Dilute overnight cultures of evolved and ancestral strains to a standardized density (~10^7 CFU/ml) in a appropriate biofilm-inducing medium (e.g., 1/20 Tryptic Soy Broth for Salmonella).
- Growth and Incubation: Dispense 200 µl aliquots into a 96-well polystyrene microtiter plate. Incubate under static conditions at a relevant temperature (e.g., 25°C for Salmonella) for 48 hours.
- Staining and Quantification:
- Carefully remove planktonic cells and rinse the adhered biofilms with water.
- Fix the biofilm with methanol or air dry.
- Stain the biofilm with 0.1% crystal violet solution for 15-20 minutes.
- Rinse off excess stain and solubilize the bound crystal violet in a 33% acetic acid solution.
- Measure the optical density of the solubilized dye at 570 nm (OD~570~) using a plate reader [96] [110].
- Data Analysis: Normalize the OD~570~ to the planktonic growth (OD~600~) if necessary. Compare the biofilm-forming capacity of evolved strains to the ancestor, with and without the EPS inhibitor.
Table 2: Key Research Reagent Solutions for Validation Experiments
| Reagent / Tool | Function / Application | Specific Example(s) |
|---|---|---|
| 2-AI Compounds (e.g., RC41) | EPS inhibitor; represses CsgD to block curli and cellulose production. | 5-(4-chlorophenyl)-N-cyclopentyl-1H-imidazol-2-amine [96] |
| AcrAB-TolC Efflux Pump Inhibitors | Chemical probe to validate efflux pump function in phenotypic assays. | Phe-Arg β-naphthylamide (PABN) [111] |
| Fluorescent Efflux Substrates | Reporter dyes for measuring efflux pump activity. | Ethidium Bromide, Hoechst 33342 [96] |
| Crystal Violet | Stain for quantifying total biofilm biomass in microtiter plate assays. | 0.1% aqueous solution [96] [110] |
| Calgary Biofilm Device | Tool for generating reproducible, multiple biofilms on pegs for high-throughput assays. | Nunc TSP (Thermo Scientific) [96] |
Signaling Pathways and Experimental Workflows
The complex interplay between EPS inhibition, bacterial stress response, and adaptive mutations can be visualized through the following signaling pathways and a standardized experimental workflow.
Diagram 1: Regulatory network of bacterial adaptation to EPS inhibition. Mutations in RamR derepress the efflux pump, while RpoS mutations directly reduce biofilm, together conferring a fitness advantage.
Diagram 2: A sequential workflow for the serial passage evolution experiment and subsequent validation.
Discussion and Future Perspectives
The integrated genotypic and phenotypic validation framework confirms that bacterial evolution in response to EPS inhibitors can follow non-intuitive paths. The absence of classical resistance to biofilm inhibition is a positive finding for anti-virulence strategies [96]. However, the selection for mutations in global regulators like ramR and rpoS reveals a critical vulnerability: unintended side effects of the inhibitor, however minor, can drive adaptation. This underscores the necessity of profiling lead compounds for off-target effects, such as growth delay, at an early stage.
A promising strategy to mitigate this evolutionary bypass is through chemical optimization. Research has demonstrated that modifying the 2-AI scaffold can enhance its specificity, partially alleviating the growth delay while retaining potent anti-biofilm activity. This refined compound eliminated the selective pressure for increased efflux, demonstrating that drug specificity is a key parameter in preventing adaptation [96]. Future research should focus on combining EPS inhibitors with efflux pump inhibitors or other anti-virulence agents to create synergistic therapies that minimize the opportunity for evolutionary escape. Furthermore, the role of efflux pumps in virulence [111] suggests that adaptations selected in vitro could have significant consequences for in vivo pathogenicity, a critical area for future investigation.
In conclusion, while anti-virulence drugs targeting EPS present a powerful tool against biofilms, their sustainable application requires a deep understanding of bacterial evolutionary responses. A comprehensive approach combining serial passage experiments, whole-genome sequencing, and functional phenotypic assays is essential for identifying and validating adaptive mutations. This knowledge is crucial for designing next-generation, evolution-resistant anti-biofilm therapeutics.
The ESKAPE pathogens (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species) represent a critical group of opportunistic bacteria capable of 'escaping' the biocidal action of conventional antibiotics, primarily through biofilm formation [112] [113]. The extracellular polymeric substance (EPS) matrix is a key structural and functional component of biofilms, conferring significant protection against antimicrobial agents and host immune responses [82] [3]. This whitepaper provides an in-depth technical analysis of the EPS matrix's role in biofilm-mediated resistance and systematically evaluates the translational potential of EPS-targeting therapeutic strategies. We detail current methodologies for EPS characterization, present quantitative data on EPS composition and its functional impact, and outline a pathway for developing these anti-biofilm approaches from fundamental research into clinical applications to combat these formidable pathogens.
The ESKAPE Pathogens and the Biofilm Challenge
ESKAPE pathogens are a primary cause of nosocomial infections worldwide, renowned for their sophisticated mechanisms of antibiotic resistance and virulence. Their ability to form biofilms on both biotic and abiotic surfaces (e.g., medical implants, catheters) is a major contributor to chronic and recalcitrant infections [113]. Within biofilms, these pathogens are embedded in a self-produced EPS matrix, which acts as a physical and functional barrier.
- Enterococcus faecium: Gram-positive, facultative anaerobe commonly associated with catheter-associated urinary tract infections (CAUTIs), surgical site infections (SSIs), and bloodstream infections. Vancomycin-resistant enterococci (VRE) are a significant clinical concern [113].
- Staphylococcus aureus: Gram-positive pathogen responsible for a wide range of infections from soft tissue infections to bacteremia. Methicillin-resistant S. aureus (MRSA) poses a severe threat [113].
- Klebsiella pneumoniae: Gram-negative, encapsulated pathogen associated with pneumonia and urinary tract infections. Notorious for being a prominent extended-spectrum β-lactamase (ESBL) producer [113].
- Acinetobacter baumannii: Gram-negative, opportunistic pathogen with a remarkable ability to adapt to harsh conditions. Carbapenem-resistant A. baumannii is on the WHO's critical priority list [113].
- Pseudomonas aeruginosa: Gram-negative, facultative anaerobe that infects immunocompromised patients, particularly those with cystic fibrosis (CF) or burn wounds. Carbapenem-resistant P. aeruginosa is also a critical priority pathogen [113].
- Enterobacter species: Gram-negative pathogens belonging to the Enterobacteriaceae family, known for their ability to acquire resistance mechanisms [113].
The biofilm mode of growth fundamentally alters the therapeutic landscape. The EPS matrix, often termed the "architectural centerpiece" of the biofilm, creates a heterogeneous environment characterized by gradients of nutrients, oxygen, and metabolic activity. This structure not limits antibiotic penetration but also harbors persister cells—dormant bacterial cells that exhibit high tolerance to antibiotics, leading to recurrent infections following the cessation of treatment.
Deconstructing the EPS Matrix: Composition, Structure, and Function
The EPS is a complex, hydrated polymer network primarily composed of polysaccharides, proteins, extracellular DNA (eDNA), and lipids [3]. Its composition is dynamic and varies significantly between species and environmental conditions.
Core Components of the EPS
Table 1: Major Constituents of the Extracellular Polymeric Substance (EPS) Matrix and Their Functional Roles [82] [3]
| EPS Component | Primary Functions | Examples in ESKAPE Pathogens |
|---|---|---|
| Polysaccharides | Structural scaffold, adhesion, cohesion, water retention, mechanical stability, immune evasion. | Alginate in P. aeruginosa; Poly-N-acetylglucosamine (PNAG) in S. aureus and E. faecalis; Pel and Psl in P. aeruginosa. |
| Proteins | Enzymatic activity, structural support, adhesion, matrix stability (e.g., amyloid fibers). | Biofilm-associated protein (Bap) in S. aureus; CdrA in P. aeruginosa; various enzymes for nutrient acquisition. |
| Extracellular DNA (eDNA) | Structural integrity, cation chelation, horizontal gene transfer, contributor to viscosity. | Released via cell lysis or active secretion; critical for initial biofilm formation in many species. |
| Lipids | Hydrophobicity, surface adhesion, potential roles in signaling and stability. | Surfactants and membrane vesicles; composition and role are less characterized. |
Functional Architecture and Microenvironments
The EPS is not a homogeneous gel but a highly organized structure featuring microcolonies, interstitial voids (water channels), and tower-like structures [114]. These water channels facilitate the convective flow of nutrients and oxygen, functioning as a primitive circulatory system. The matrix establishes distinct chemical microenvironments, which can shield bacterial cells from antimicrobials through:
- Binding and Neutralization: Direct interaction and sequestration of antimicrobial molecules.
- Retarded Diffusion: The highly viscous and polyanionic nature of the EPS physically hinders the penetration of antibiotics.
- Altered Chemical Microenvironment: Gradients of pH and metabolites can inactivate certain antibiotics.
The following diagram illustrates the functional organization of a typical biofilm and the protective role of the EPS matrix.
Diagram: Functional organization of a typical biofilm and the protective role of the EPS matrix.
Quantitative Analysis of EPS and Biofilm Properties
Understanding the physical and chemical properties of biofilms is essential for developing effective countermeasures. The following tables summarize key quantitative data from recent studies on biofilm composition and density.
Table 2: Spectroscopic Signatures of Key EPS Components via ATR/FT-IR Analysis [3]
| IR Spectral Region | Primary EPS Component | Main Functional Groups | Key Vibrational Modes |
|---|---|---|---|
| 2800–3000 cm⁻¹ | Lipids | CH, CH₂, CH₃ | Stretching vibrations |
| 1500–1800 cm⁻¹ | Proteins | C=O, N-H, C-N | Amide I, Amide II |
| 900–1250 cm⁻¹ | Polysaccharides, Nucleic Acids | C-O, C-O-C, P=O, C-N, N-H | C-O stretch, P=O stretch, Amide III |
Table 3: Correlation Between Biofilm Volumetric Density and OCT Signal Intensity [115]
| Bacterial Strain / Condition | Growth Method | Volumetric Bacterial Density (cells/µm³) | Re-scaled OCT Signal Intensity (A.U.) |
|---|---|---|---|
| Staphylococcus epidermidis 252 (Non-EPS producer) | Static, 48h | 0.085 ± 0.011 | 25.3 ± 3.1 |
| Staphylococcus epidermidis ATCC 35984 (EPS producer) | Static, 48h | 0.051 ± 0.007 | 42.7 ± 5.2 |
| Pseudomonas aeruginosa ATCC 39324 (in LB medium) | CDFF, 18h | 0.062 ± 0.008 | 35.1 ± 4.3 |
| Pseudomonas aeruginosa ATCC 39324 (in ASM) | CDFF, 18h | 0.078 ± 0.009 | 28.9 ± 3.6 |
| Oral Multi-species Biofilm (S. oralis & A. naeslundii) | Static, 24h | 0.071 ± 0.010 | 31.5 ± 4.0 |
Key Insight: The data show a clear inverse relationship between EPS production and volumetric bacterial density. EPS-producing S. epidermidis (ATCC 35984) has a lower cell density but a higher OCT signal intensity, which is attributed to the light-scattering properties of the dense EPS matrix. Re-scaling of OCT signal intensities is crucial for quantitative comparison and has been shown to increase linearly with independently determined volumetric bacterial densities, providing a non-destructive method to assess biofilm accumulation [115].
Methodological Toolkit for EPS and Biofilm Research
Experimental Protocols for EPS Characterization
Protocol 1: EPS Destabilization Using Hydrolytic Enzymes [3] This protocol assesses the contribution of specific EPS components to biofilm integrity.
- Biofilm Growth: Grow biofilms of the target ESKAPE pathogen (e.g., P. aeruginosa or S. aureus) in a relevant model system (e.g., Calgary biofilm device, flow cell) for 24-48 hours.
- Enzyme Preparation: Prepare solutions of hydrolytic enzymes in an appropriate buffer (e.g., PBS). Common enzymes and concentrations include:
- Serine Proteases (Savinase, Subtilisin A): 10-100 µg/mL to target protein components.
- α-Amylase: 10-100 U/mL to target polysaccharides.
- DNase I: 10-100 µg/mL to target eDNA.
- Treatment: Gently wash mature biofilms and incubate with the enzyme solution or a buffer control for 1-24 hours at 37°C.
- Assessment:
- Biomass Quantification: Use crystal violet staining to measure total remaining biomass.
- Viability Assessment: Perform colony-forming unit (CFU) counts after biofilm disruption.
- Structural Analysis: Use confocal laser scanning microscopy (CLSM) or scanning electron microscopy (SEM) to visualize structural disintegration.
Protocol 2: Fourier Transform Infrared (FT-IR) Spectroscopy for EPS Composition [3] This technique provides a chemical fingerprint of the biofilm matrix.
- Sample Preparation: Grow biofilms directly on a germanium or zinc selenide crystal suitable for Attenuated Total Reflectance (ATR) sampling. Alternatively, harvest and dry biofilm material onto the crystal.
- Spectral Acquisition: Place the crystal in the ATR/FT-IR spectrometer. Collect spectra over a range of 4000-600 cm⁻¹ with a resolution of 4 cm⁻¹. Accumulate 64-256 scans to improve the signal-to-noise ratio.
- Data Analysis:
- Subtract the background spectrum.
- Normalize spectra to a key band (e.g., amide I).
- Analyze the intensities and ratios of characteristic bands (see Table 2), such as the Amide II/Polysaccharide (e.g., 1550 cm⁻¹ / 1080 cm⁻¹) ratio, to track relative changes in protein and polysaccharide content during biofilm development or after treatment.
Protocol 3: Quantifying Biofilm Biovolume using Confocal Laser Scanning Microscopy (CLSM) [114] This protocol allows for the 3D quantification of biofilm structures.
- Biofilm Staining: Grow biofilms on sterile glass coverslips. Fix with 4% formaldehyde for 10 minutes at room temperature. Wash and stain with a fluorescent nucleic acid stain (e.g., propidium iodide, SYTO 9) for 15 minutes.
- Image Acquisition: Mount the coverslip and image using a CLSM with a high-numerical-aperture objective (e.g., 63x). Acquire z-stack images through the entire biofilm depth with a step size of 0.5-1.0 µm.
- 3D Reconstruction and Quantification:
- Import image stacks into 3D analysis software (e.g., Amira, IMARIS, COMSTAT).
- Apply a median filter to reduce noise.
- Set an appropriate threshold to distinguish bacterial biomass from background.
- Use the software's quantification modules to calculate total biovolume (µm³/µm²), average thickness, and substratum coverage.
The Scientist's Toolkit: Key Reagents and Technologies
Table 4: Essential Research Reagents and Tools for EPS and Biofilm Studies
| Tool / Reagent | Function/Application | Specific Examples & Notes |
|---|---|---|
| Hydrolytic Enzymes | Functional dissection of EPS matrix components; potential anti-biofilm agents. | Savinase, Subtilisin A (Proteases); α-Amylase; DNase I [3]. |
| Fluorescent Stains & Probes | Visualization and quantification of biofilm components in live or fixed samples. | Propidium iodide, SYTO 9 (nucleic acids); ConA, WGA (specific polysaccharides); FITC (proteins) [114]. |
| Confocal Laser Scanning Microscope (CLSM) | Non-destructive 3D imaging and biovolume quantification of biofilm architecture. | Used to visualize towers and water channels; essential for quantifying biovolume over time [114]. |
| Optical Coherence Tomography (OCT) | Non-destructive, label-free imaging of biofilm thickness and macro-structure. | Requires re-scaling of signal intensities for quantitative comparison of biofilm density between images [115]. |
| ATR/FT-IR Spectrometer | In-situ chemical analysis of biofilm composition; monitoring changes in real-time. | Identifies functional groups and relative proportions of proteins, polysaccharides, and lipids in the EPS [3]. |
| Quorum Sensing Inhibitors (QSIs) | Attenuation of virulence and biofilm formation by disrupting bacterial communication. | Molecule libraries targeting the Las, Rhl, and Pqs systems in P. aeruginosa; Agr system in S. aureus [113]. |
The following workflow diagram integrates these tools and methods into a cohesive research strategy for evaluating EPS-targeting therapies.
Diagram: Workflow for evaluating EPS-targeting therapies.
EPS-Targeting Therapeutic Strategies and Translational Pathways
Targeting the EPS matrix presents a promising avenue for disarming biofilms and restoring the efficacy of conventional antibiotics.
Key Therapeutic Modalities
Enzyme-Based Disruption: The use of EPS-degrading enzymes is a direct strategy to dismantle the biofilm scaffold. As demonstrated in Table 4, proteases, glycosidases, and nucleases can significantly reduce biofilm biomass and potentiate the action of co-administered antibiotics [3]. For instance, serine proteases like Savinase have shown high efficiency (≥70% biomass reduction) against both P. aeruginosa and S. aureus biofilms. A key challenge is ensuring enzyme stability and delivery to the infection site.
Quorum Sensing Inhibition (QSI): Since QS regulates EPS production and virulence factor expression in many ESKAPE pathogens, QSIs can prevent biofilm maturation and render bacteria more susceptible to antibiotics and host defenses [113]. This anti-virulence approach aims to disarm the pathogen without imposing a strong selective pressure for resistance.
Combination Therapies: The most promising strategy involves combining EPS-targeting agents with traditional antibiotics. The diagram below illustrates how this combination therapy works synergistically to eradicate biofilms.
Diagram: Synergistic mechanism of combination therapy.
Translational Pathway and Challenges
The journey from bench to bedside for EPS-targeting therapies involves several critical stages:
- In Vitro Validation: Demonstration of efficacy against monospecies and polymicrobial biofilms in laboratory models, using the protocols outlined in Section 4.1.
- Pre-Clinical Development:
- Formulation & Delivery: Developing stable formulations for enzymes and QSIs (e.g., nanoparticle encapsulation, hydrogels) and effective delivery systems (e.g., inhalation for lung biofilms, coating for implants).
- In Vivo Models: Testing in relevant animal models of infection (e.g., murine catheter, lung infection models) to confirm safety and efficacy.
- Clinical Trials:
- Phase I/II: Focus on safety, pharmacokinetics, and preliminary efficacy in patients with biofilm-associated infections.
- Phase III: Large-scale trials to establish superiority or non-inferiority compared to standard of care, particularly in combination with antibiotics.
Major Hurdles include the potential for immunogenicity of enzyme therapies, the rapid degradation of therapeutic agents in vivo, and the complexity of the regulatory pathway for combination anti-biofilm/antibiotic treatments. Furthermore, the heterogeneity of EPS composition across different pathogens and infection sites necessitates the development of tailored or broad-spectrum approaches.
The EPS matrix is a fundamental determinant of biofilm-associated resistance in ESKAPE pathogens, representing both a formidable challenge and a promising therapeutic target. The integration of advanced analytical techniques—such as FT-IR spectroscopy for compositional analysis, CLSM for 3D structural quantification, and OCT for non-destructive monitoring—provides a robust framework for dissecting the EPS and evaluating novel interventions. Enzyme-based strategies and quorum sensing inhibitors, particularly when used in combination with conventional antibiotics, hold significant potential to disrupt the protective biofilm barrier and resensitize pathogens to treatment. Translating these strategies from the laboratory to the clinic will require focused efforts on overcoming delivery challenges, validating efficacy in complex in vivo environments, and navigating the regulatory landscape. Success in this endeavor will mark a critical advancement in the ongoing battle against multidrug-resistant infections.
Conclusion
The extracellular polymeric substance is unequivocally the master architect of biofilm structure and the primary mediator of its recalcitrance. Research unequivocally demonstrates that targeting the EPS matrix—through enzymatic degradation, quorum sensing inhibition, or novel small molecules like 2-AI—represents a paradigm shift from traditional biocidal approaches. The low propensity for resistance against these anti-virulence strategies, as they target public goods, offers a significant clinical advantage. Future directions must focus on developing highly specific, multi-targeted therapies and advanced drug delivery systems that can effectively penetrate and disrupt the EPS barrier. Integrating a deep mechanistic understanding of EPS composition and function with innovative materials science and pharmacology is the key to unlocking new, effective treatments for biofilm-associated infections, directly addressing the global crisis of antimicrobial resistance and improving patient outcomes.
References
- 1. Biofilms: structure, resistance mechanism, emerging control ... [https://pubs.rsc.org/en/content/articlehtml/2025/pm/d5pm00094g]
- 2. Extracellular polymeric substance [https://en.wikipedia.org/wiki/Extracellular_polymeric_substance]
- 3. Extracellular polymeric substances, a key element in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6604936/]
- 4. Constituent of extracellular polymeric substances (EPS ... [https://bmcmicrobiol.biomedcentral.com/articles/10.1186/s12866-025-04034-z]
- 5. Microbial Extracellular Polymeric Substances: Ecological ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2018.01636/full]
- 6. Biofilm formation and analysis of EPS architecture ... [https://www.sciencedirect.com/science/article/abs/pii/S0963996925006118]
- 7. Bacterial Extracellular Polysaccharides Involved in Biofilm ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6254922/]
- 8. The EPS Matrix: The “House of Biofilm Cells” - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC2168682/]
- 9. Polysaccharides' Structures and Functions in Biofilm ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9965654/]
- 10. Regulation of Biofilm Exopolysaccharide Production by ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2021.730980/full]
- 11. Bacillus subtilis EpsA-O: A novel exopolysaccharide ... [https://www.nature.com/articles/s41522-024-00555-z]
- 12. Functional amyloids from bacterial biofilms – structural ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9172111/]
- 13. The role of extracellular DNA in the formation, architecture ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8667714/]
- 14. Z-form extracellular DNA is a structural component of the ... [https://www.sciencedirect.com/science/article/pii/S0092867421012204]
- 15. Biofilms: Microbial Life on Surfaces - Volume 8, Number 9 ... [https://wwwnc.cdc.gov/eid/article/8/9/02-0063_article]
- 16. Strategy to combat biofilms: a focus on biofilm dispersal ... [https://www.nature.com/articles/s41522-023-00427-y]
- 17. What's on the Outside Matters: The Role of the Extracellular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4933457/]
- 18. Constituent of extracellular polymeric substances (EPS ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12079940/]
- 19. Bioactivity of microbial biofilms in extreme environments - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12446376/]
- 20. Biofilm dispersion: The key to biofilm eradication or ... [https://www.sciencedirect.com/science/article/pii/S2590207520300095]
- 21. A novel dual-staining method for cost-effective visualization ... [https://www.nature.com/articles/s41598-024-80644-3]
- 22. Quantitative and Qualitative Assessment Methods for Biofilm ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6133255/]
- 23. Stress-hardening behaviour of biofilm streamers - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12569198/]
- 24. A Review on the Roles of Extracellular Polymeric ... [https://www.mdpi.com/2073-4441/16/19/2812]
- 25. The interplay of adhesion, friction, and nutrient availability ... [https://www.nature.com/articles/s44431-025-00007-4]
- 26. The driving role of extracellular polymeric substances in ... [https://bioresourcesbioprocessing.springeropen.com/articles/10.1186/s40643-025-00922-4]
- 27. Environmental Stress Promotes Biofilm Formation and ... [https://www.mdpi.com/2076-2607/13/8/1812]
- 28. Enhanced Biofilm Inhibition via Extracellular Polymeric ... [https://link.springer.com/article/10.1007/s11814-025-00583-1]
- 29. Potential functions and applications of diverse microbial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9626724/]
- 30. Use sufficient color contrast | Digital Accessibility [https://accessibility.huit.harvard.edu/use-sufficient-color-contrast]
- 31. The non-attached biofilm aggregate [https://www.nature.com/articles/s42003-023-05281-4]
- 32. Visualisation and biovolume quantification in the ... [https://www.nature.com/articles/s41598-021-90455-5]
- 33. Quantitative image analysis of microbial communities with ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7840502/]
- 34. Detection techniques for extracellular polymeric substances in ... [https://bioresources.cnr.ncsu.edu/resources/detection-techniques-for-extracellular-polymeric-substances-in-biofilms-a-review/]
- 35. Extracellular polymeric substances, a key element in understanding... [https://www.aimspress.com/article/10.3934/microbiol.2018.2.274]
- 36. (PDF) Insight Into Extracellular Polymeric Substances Eps ... Extraction [https://www.academia.edu/82234094/Insight_Into_Extracellular_Polymeric_Substances_Eps_Extraction_and_Quantification_Techniques_Causes_Mechanisms_and_Applications_Mini_Review]
- 37. Production and characterization of exopolysaccharides ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12077034/]
- 38. Microscale assay for the quantification of total ... [https://pubmed.ncbi.nlm.nih.gov/41224112/]
- 39. Rapid quantification of pullulan in fermentation broth using UV-visible... [https://pubs.rsc.org/en/content/articlelanding/2025/ay/d5ay00034c]
- 40. Analysis of biofilm assembly by large area automated AFM [https://www.nature.com/articles/s41522-025-00704-y]
- 41. Biofilm research advances from an innovative bibliometric ... [https://www.sciencedirect.com/science/article/pii/S2949864325000141]
- 42. Extracellular Polymeric Substance - an overview [https://www.sciencedirect.com/topics/immunology-and-microbiology/extracellular-polymeric-substance]
- 43. Matrix matters: How extracellular substances shape biofilm ... [https://www.sciencedirect.com/science/article/abs/pii/S0927776524006003]
- 44. Structural and mechanical characterization of biofilm ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8071786/]
- 45. The FT-IR and Raman Spectroscopies as Tools for Biofilm ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7313032/]
- 46. Confocal Laser Scanning Microscopy for Analysis of ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2019.00677/full]
- 47. Biofilm Cohesiveness Measurement Using a Novel Atomic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1892862/]
- 48. Combined use of confocal laser scanning microscopy ... [https://www.sciencedirect.com/science/article/abs/pii/S0043135408004569]
- 49. Microbial enzymes as powerful natural anti-biofilm candidates [https://pmc.ncbi.nlm.nih.gov/articles/PMC11664836/]
- 50. Enzymatic dispersion of biofilms: An emerging biocatalytic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9478923/]
- 51. Strategy to combat biofilms: a focus on biofilm dispersal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10485009/]
- 52. Microbial enzymes as powerful natural anti-biofilm candidates [https://microbialcellfactories.biomedcentral.com/articles/10.1186/s12934-024-02610-y]
- 53. Enzymatic Disruption of Biofilms During Cheese ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2021.791061/full]
- 54. Formation, architecture, and persistence of oral biofilms [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2025.1602962/full]
- 55. EDTA: An Antimicrobial and Antibiofilm Agent for Use in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4486448/]
- 56. Novel colistin-EDTA combination for successful eradication ... [https://www.nature.com/articles/s41598-021-01052-5]
- 57. Biofilm Formation by Otopathogenic Strains of P. aeruginosa ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3399970/]
- 58. Tailoring Nanoparticle-Biofilm Interactions to Increase the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7354952/]
- 59. Nanomaterial-enabled anti-biofilm strategies - RSC Publishing [https://pubs.rsc.org/en/content/articlehtml/2025/nr/d4nr04774e]
- 60. Microbial extracellular polymeric substances in the ... [https://pubmed.ncbi.nlm.nih.gov/39333414/]
- 61. Nanoparticles targeting biofilms: A new era in combating ... [https://www.sciencedirect.com/science/article/pii/S2590097825000370]
- 62. New ultrasound drug delivery system found to be highly ... [https://www.ox.ac.uk/news/2025-04-22-new-ultrasound-drug-delivery-system-found-be-highly-effective-against-bacterial]
- 63. Janus nanoparticles targeting extracellular polymeric ... [https://www.nature.com/articles/s41467-023-40830-9]
- 64. Nanotechnology Meets superbugs [https://www.nature.com/articles/s41598-025-21606-1]
- 65. A comprehensive comparison of biofilm formation and ... [https://www.sciencedirect.com/science/article/pii/S2590207523000023]
- 66. Characterizing the Shearing Stresses within the CDC Biofilm ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8399442/]
- 67. A Bead Biofilm Reactor for High-Throughput Growth and ... [https://www.mdpi.com/2076-2607/12/8/1588]
- 68. Experimental models and model organisms in cardiac ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10512071/]
- 69. Investigation of shockwave treatment for disruption ... [https://www.nature.com/articles/s41598-025-19955-y]
- 70. Strategies for biofilm inhibition: the role of synthetic drug [https://pubmed.ncbi.nlm.nih.gov/41269277/]
- 71. Natural product-based inhibitors of quorum sensing [https://pubmed.ncbi.nlm.nih.gov/40641742/]
- 72. The role of cyano-phycocyanin as a quorum sensing ... [https://www.frontiersin.org/journals/cellular-and-infection-microbiology/articles/10.3389/fcimb.2025.1624927/full]
- 73. Marine-Derived Furanones Targeting Quorum-Sensing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11378241/]
- 74. Balancing activity and toxicity in 2N-aminated 5-aryl-2- ... [https://www.sciencedirect.com/science/article/abs/pii/S0968089625000562]
- 75. Characterization, structure, and function of extracellular ... [https://www.sciencedirect.com/science/article/pii/S1944398624192429]
- 76. Inhibiting bacterial cooperation is an evolutionarily robust ... [https://www.nature.com/articles/s41467-019-13660-x]
- 77. An Improved 2-Aminoimidazole Based Anti-Biofilm Coating ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2021.658521/full]
- 78. EPS inhibitor treatment of Salmonella impacts evolution ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12056028/]
- 79. An Improved 2-Aminoimidazole Based Anti-Biofilm Coating for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8097006/]
- 80. Innovative Strategies Toward the Disassembly of the EPS ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2020.00952/full]
- 81. Mechanisms of antimicrobial resistance in biofilms [https://www.nature.com/articles/s44259-024-00046-3]
- 82. Frontiers | Bioactivity of microbial biofilms in extreme environments [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2025.1602583/full]
- 83. Biofilms: Formation, drug resistance and alternatives to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9576500/]
- 84. Synergistic Effect of Plant Compounds in Combination with ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10675371/]
- 85. Dissecting Targets in the Biofilm Life Cycle [https://www.mdpi.com/1424-8247/15/10/1253]
- 86. Contemporary strategies and approaches for ... [https://www.sciencedirect.com/science/article/pii/S2095177923002794]
- 87. Linking composition of extracellular polymeric substances ... [https://www.sciencedirect.com/science/article/abs/pii/S0043135417310539]
- 88. Optimization of the production of Exopolysaccharide (EPS) ... [https://www.sciencedirect.com/science/article/pii/S2950194624000840]
- 89. Biofilm Formation, Modulation, and Transcriptomic Regulation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11765580/]
- 90. Production and characterization of exopolysaccharides from ... [https://microbialcellfactories.biomedcentral.com/articles/10.1186/s12934-025-02730-z]
- 91. Therapeutic Strategies against Biofilm Infections - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9866932/]
- 92. Combination drug strategies for biofilm eradication using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10149696/]
- 93. Combatting biofilm-mediated infections in clinical settings ... [https://www.sciencedirect.com/science/article/pii/S246823302400015X]
- 94. Salmonella Biofilm Formation, Chronic Infection, and ... [https://www.frontiersin.org/journals/cellular-and-infection-microbiology/articles/10.3389/fcimb.2020.624622/full]
- 95. Salmonella Extracellular Polymeric Substances Modulate ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7074811/]
- 96. EPS inhibitor treatment of Salmonella impacts evolution ... [https://www.nature.com/articles/s41522-025-00693-y]
- 97. Exposure of Salmonella biofilms to antibiotic ... [https://www.nature.com/articles/s41522-020-00178-0]
- 98. Stimuli-responsive nanocarriers for bacterial biofilm treatment [https://pmc.ncbi.nlm.nih.gov/articles/PMC8333162/]
- 99. Epitope alteration by small molecules and applications in drug ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9278120/]
- 100. Common Enzyme Inhibition Mechanisms Explained with ... [https://synapse.patsnap.com/article/common-enzyme-inhibition-mechanisms-explained-with-examples]
- 101. Enzymes: Inhibitors (A-level Biology) [https://studymind.co.uk/notes/enzymes-inhibitors/]
- 102. Mechanism of Action Assays for Enzymes - NCBI [https://www.ncbi.nlm.nih.gov/books/NBK92001/]
- 103. Small-molecule delivery by nanoparticles for anticancer therapy [https://pmc.ncbi.nlm.nih.gov/articles/PMC3729441/]
- 104. Public goods-mediated bacterial interplay in aquatic ... [https://www.sciencedirect.com/science/article/abs/pii/S0043135425012163]
- 105. Evolution-proof inhibitors of public good cooperation [https://pmc.ncbi.nlm.nih.gov/articles/PMC9616471/]
- 106. Biofilms Exposed: Innovative Imaging and Therapeutic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12466655/]
- 107. Genomic and metabolic adaptations of biofilms to ... [https://www.nature.com/articles/s41467-022-29914-0]
- 108. Mathematical modelling of nutrient-dependent biofilm ... [https://arxiv.org/html/2509.18065v1]
- 109. Mathematical Modeling of Biofilm Structures Using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5751404/]
- 110. Salmonella stress response systems as targets for anti ... [https://bmcmicrobiol.biomedcentral.com/articles/10.1186/s12866-025-04003-6]
- 111. The AcrAB efflux pump contributes to the virulence of ... [https://www.frontiersin.org/journals/cellular-and-infection-microbiology/articles/10.3389/fcimb.2025.1633585/full]
- 112. Clinical relevance of the ESKAPE pathogens [https://pubmed.ncbi.nlm.nih.gov/23458769/]
- 113. Revisiting ESKAPE Pathogens: virulence, resistance, and ... [https://www.frontiersin.org/journals/cellular-and-infection-microbiology/articles/10.3389/fcimb.2023.1159798/full]
- 114. Visualisation and biovolume quantification in the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8160185/]
- 115. Bacterial Density and Biofilm Structure Determined by ... [https://www.nature.com/articles/s41598-019-46196-7]