Overcoming the Challenges: A Guide to AFM Imaging of Hydrated Biofilm Structures for Biomedical Research
Atomic Force Microscopy (AFM) offers unparalleled potential for characterizing the structural and mechanical properties of hydrated biofilms under near-physiological conditions, which is crucial for developing effective anti-biofilm strategies.
Overcoming the Challenges: A Guide to AFM Imaging of Hydrated Biofilm Structures for Biomedical Research
Abstract
Atomic Force Microscopy (AFM) offers unparalleled potential for characterizing the structural and mechanical properties of hydrated biofilms under near-physiological conditions, which is crucial for developing effective anti-biofilm strategies. However, researchers face significant challenges, including the soft and dynamic nature of biofilms, limited imaging areas, and difficulties in maintaining sample integrity. This article explores the foundational principles behind these hurdles, details advanced methodological approaches for high-resolution imaging and nanomechanical mapping, and provides troubleshooting strategies for sample immobilization and data interpretation. Furthermore, it validates AFM data through comparative analysis with other techniques and discusses the transformative impact of emerging technologies like automated large-area AFM and artificial intelligence. This comprehensive guide is tailored for scientists and drug development professionals seeking to leverage AFM for robust, reproducible biofilm analysis in clinical and biomedical contexts.
Why Hydrated Biofilms Are a Formidable Challenge for AFM
The Critical Importance of Hydrated State Analysis for Clinical Relevance
Frequently Asked Questions (FAQs)
Q1: Why is it so challenging to image hydrated biofilms with AFM? Hydrated biofilms are soft, diffuse, and easily disrupted by the AFM tip. Cells are often only weakly attached to the surface and can be moved or swept away during scanning. Furthermore, microbial motility in liquid environments makes sustained, high-resolution imaging exceptionally difficult without proper immobilization [1].
Q2: What are the most common artifacts seen in AFM images of biofilms, and how can I fix them? Common artifacts include repetitive patterns from a contaminated or broken tip, streaks from environmental vibration or loose surface contamination, and difficulty imaging vertical structures due to an inappropriate tip geometry. Solutions involve using a new, sharp probe, ensuring proper sample preparation to remove loose debris, working on a vibration-isolation table, and selecting high-aspect-ratio tips for complex topographies [2].
Q3: My biofilm samples keep getting damaged during scanning. What imaging mode should I use? For soft, hydrated biological samples like biofilms, Tapping Mode (or intermittent contact mode) is strongly recommended. This mode minimizes lateral forces and friction compared to Contact Mode, thereby reducing sample damage and deformation. It allows for reliable imaging of delicate biofilm structures under physiological conditions [1] [3].
Q4: How can I obtain quantitative mechanical data from my hydrated biofilm sample? Force Spectroscopy or Peak Force Quantitative Nanomechanical Mapping (PF-QNM) modes are used. By collecting force-distance curves across the sample surface, you can map properties like elastic modulus (stiffness), adhesion, and deformation. This provides crucial data on biofilm mechanical properties and their heterogeneity [1] [3] [4].
Troubleshooting Guides
Problem 1: Poor Image Quality with Streaks and Blurring in Liquid
| Symptoms | Possible Causes | Recommended Solutions |
|---|---|---|
| Horizontal streaks across image [2] | Environmental noise/vibration [2] | Use an active anti-vibration table; relocate AFM to a quieter location (e.g., basement); perform imaging during quieter hours [2]. |
| Blurred images, tip seems to drag sample | Loose surface contamination [2] | Improve sample preparation protocols to minimize loosely adhered material; rinse sample gently before imaging [2]. |
| Unstable laser signal, noisy baseline | Laser interference from reflective substrate [2] | Use a probe with a reflective back-coating (e.g., gold, aluminum) to minimize interference from the sample [2]. |
Problem 2: Sample Detachment or Disruption During Scanning
| Symptoms | Possible Causes | Recommended Solutions |
|---|---|---|
| Cells are swept away by the tip | Inadequate immobilization of cells/biofilm [1] | Use mechanical entrapment in porous membranes or chemical immobilization on poly-L-lysine or other treated surfaces [1]. |
| Soft, diffuse biofilm is deformed | Use of inappropriate Contact Mode [1] [3] | Switch to Tapping Mode to reduce lateral forces and sample damage [1] [3]. |
| Loss of resolution over time | Biofilm growth or motility during experiment [1] | Consider gentle chemical fixation (e.g., with glutaraldehyde) if viability is not required, or use real-time imaging to capture dynamics [1]. |
Problem 3: Inaccurate Topography and Tip Artifacts
| Symptoms | Possible Causes | Recommended Solutions |
|---|---|---|
| Features appear duplicated or wider than expected [2] | Contaminated or broken AFM tip [2] | Replace the probe with a new, guaranteed-sharp one [2]. |
| Inability to resolve deep trenches or vertical structures [2] | Low aspect-ratio pyramidal tip geometry [2] | Switch to a conical or High-Aspect-Ratio (HAR) probe to better access complex features [2]. |
| Phase images show artifacts not visible in topography | Operating in the wrong interaction regime [3] | Optimize the setpoint and amplitude to ensure the tip is operating primarily in the repulsive regime for accurate phase data [3]. |
Table 1: Summary of AFM Techniques for Hydrated Biofilm Analysis
| AFM Mode | Key Measurable Parameters | Typical Values/Units | Clinical & Research Relevance |
|---|---|---|---|
| Tapping Mode | Topography, Roughness, 3D Architecture | nm-µm scale height; Roughness (Ra, Rq) in nm | Visualizes biofilm heterogeneity, microcolony formation, and water channels in near-native state [1] [3]. |
| Force Spectroscopy / Nanoindentation | Elastic (Young's) Modulus, Adhesion Force, Stiffness | Elastic Modulus: kPa to MPa range [1] [4] | Quantifies biofilm mechanical robustness, linked to antibiotic resistance and physical stability [1] [4]. |
| Phase Imaging | Qualitative Material Properties (adhesion, viscoelasticity) | Phase Lag (degrees) | Distinguishes between EPS components, cells, and abiotic surfaces based on mechanical differences [1] [3]. |
| Large-Area Automated AFM | Cell Count, Orientation, Confluency, Spatial Distribution | 10,000+ cells over mm² areas [5] [6] | Links single-cell details to community-scale organization, revealing patterns like honeycomb structures [5] [6]. |
Table 2: Common Functionalized Tips for Biofilm Interaction Studies
| Tip Functionalization | Measured Interaction | Application in Biofilm Research |
|---|---|---|
| Hydrophobic Groups | Hydrophobic Interactions | Probes adhesion forces related to hydrophobic cell surfaces and EPS [3]. |
| Specific Antibodies | Ligand-Receptor Binding | Maps the distribution of specific surface antigens or adhesins on biofilm cells [1]. |
| Lectins | Carbohydrate-Binding | Characterizes polysaccharide components within the EPS matrix [1]. |
Experimental Protocols
Protocol 1: Reliable Immobilization of Hydrated Biofilms for AFM
Principle: Securely attach biofilm or planktonic cells to a substrate to withstand lateral scanning forces without altering their native physiological state [1].
Materials:
- Freshly cleaved mica or glass coverslips.
- Poly-L-lysine solution (0.1% w/v) or other chemical adhesives (e.g., trimethoxysilyl-propyl-diethylenetriamine).
- Cell suspension or pre-grown biofilm.
- AFM liquid cell and appropriate buffer.
Method:
- Substrate Preparation: Treat the mica or glass surface with 50-100 µL of poly-L-lysine solution for 30 minutes. Rinse thoroughly with ultrapure water to remove excess, unbound poly-L-lysine and air dry.
- Sample Deposition: Apply 20-50 µL of the cell suspension onto the treated substrate and allow to adhere for 15-60 minutes. For pre-grown biofilms, place the substrate in the growth medium during cultivation.
- Gentle Rinsing: Carefully rinse the substrate with a compatible buffer (e.g., PBS or a low-salt buffer) to remove non-adherent cells. Avoid high shear forces that could disrupt the biofilm.
- AFM Mounting: Immediately transfer the substrate to the AFM liquid cell, ensuring the sample remains hydrated at all times. Add a sufficient volume of buffer to submerge the sample.
Technical Notes: The addition of divalent cations (e.g., Mg²⁺, Ca²⁺) to the immobilization buffer or growth medium can improve attachment for some bacterial strains without significantly affecting viability [1].
Protocol 2: In-Liquid Mechanical Property Mapping via Force Spectroscopy
Principle: Acquire force-distance curves at multiple points on the biofilm surface to generate quantitative maps of nanomechanical properties [1] [3].
Materials:
- Properly immobilized hydrated biofilm sample.
- AFM with force spectroscopy capability.
- Sharp, non-functionalized tip (e.g., silicon nitride) for stiffness measurements, or a functionalized tip for specific adhesion studies.
- Calibrated cantilever spring constant.
Method:
- Cantilever Calibration: Precisely calibrate the spring constant of the cantilever using the thermal tune method or a reference sample.
- Engage and Locate: Engage the tip with the sample in Tapping Mode to locate a region of interest.
- Force Volume Setup: Switch to Force Spectroscopy mode. Define a grid (e.g., 32x32 or 64x64 points) over the area to be mapped. Set the maximum applied force to a low value (typically 0.5-5 nN) to avoid damaging the soft sample.
- Data Acquisition: Automatically collect a force-distance curve at each point in the grid.
- Data Analysis: Use appropriate software (often provided by the AFM manufacturer) to fit the retraction part of the curve to calculate adhesion force and the approach part to a contact mechanics model (e.g., Hertz, Sneddon, or DMT models) to extract the Elastic Modulus.
Technical Notes: The Hertz model is commonly used for fitting, assuming a parabolic tip indenting an elastic, homogeneous sample. Ensure the indentation depth is not too large compared to the sample thickness to avoid substrate effects [1].
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Hydrated Biofilm AFM Studies
| Item Name | Function/Application | Key Considerations |
|---|---|---|
| Poly-L-lysine | Chemical immobilization agent for securing cells to substrates. | Provides a positively charged surface for cell attachment. Ensure biocompatibility for live-cell studies [1]. |
| Silicon Nitride Tips | Standard probes for imaging and force measurement in liquid. | Low spring constants are critical for soft sample imaging. Sharp tips (high resolution) vs. spherical tips (better for mechanics) [1] [3]. |
| Functionalized Tips | Probes coated with specific molecules (e.g., lectins, antibodies). | Enables measurement of specific molecular interactions (e.g., ligand binding) within the biofilm [1] [3]. |
| High-Aspect-Ratio (HAR) Tips | Probes with elongated, sharp tips. | Essential for accurately resolving deep, narrow pores and channels in the complex 3D structure of biofilms [2]. |
| Liquid Cell | AFM accessory for housing the sample and maintaining hydration. | Must be chemically compatible with buffers and biological samples. Allows for in-situ experimentation [1]. |
| Machine Learning Software | For automated image stitching and data analysis. | Crucial for analyzing large-area AFM scans, enabling cell detection, classification, and extraction of quantitative data from thousands of cells [5] [7]. |
Troubleshooting Guide: Common AFM Artifacts in Biofilm Imaging
This guide helps diagnose and resolve frequent artifacts encountered during Atomic Force Microscopy (AFM) of hydrated biofilms.
Table 1: Troubleshooting Common AFM Imaging Problems with Biofilms
| Problem & Symptom | Potential Cause | Recommended Solution | Preventive Measures |
|---|---|---|---|
| Unexpected/Repetitive Patterns [8] [2]: Duplicated structures, irregular shapes repeating across image. | Tip Artefacts: Broken or contaminated tip, resulting in a blunt tip. | Replace the AFM probe with a new, sharp one [2]. | Use conical tips over pyramidal/tetrahedral shapes; ensure proper probe storage and handling [2]. |
| Difficulty Imaging Vertical Structures/Deep Trenches [2]: Inability to resolve steep-edged features or trench bottoms. | Low Aspect Ratio Probe: Tip geometry prevents access to deep or narrow features [2]. | Switch to a High Aspect Ratio (HAR) probe [2]. | Select probe shape (conical) and aspect ratio appropriate for expected sample topography [2]. |
| Repetitive Lines Across Image [2]: Regular, repeating lines in the trace and retrace directions. | Electrical Noise (50/60 Hz) or Laser Interference from reflections off a reflective sample surface [2]. | Image during quieter electrical periods (e.g., evenings); use a probe with a reflective coating to minimize laser interference [2]. | Ensure proper grounding; use reflective-coated probes for highly reflective samples [2]. |
| Streaks on Images [2]: Lines or smearing in the scan direction. | Environmental Vibration or Surface Contamination where loose particles interact with the tip [2]. | Relocate AFM to a quieter location (e.g., basement); use anti-vibration tables; ensure sample preparation minimizes loose material [2]. | Image during quiet hours; use "STOP AFM in progress" signs; optimize sample rinse protocols to remove unattached cells [5]. |
| Thermal Drift [8]: Gradual displacement between tip and sample, causing image distortion. | Inherent Thermal Effects in the system, causing scanner drift over time [8]. | Use AFM systems with closed-loop scanners and real-time drift correction algorithms [8]. | Allow the system sufficient time to thermally equilibrate before starting high-resolution scans [8]. |
Frequently Asked Questions (FAQs)
Q1: How does the softness of hydrated biofilms challenge AFM imaging, and what modes are best to use?
The inherent softness and high hydration of biofilms make them easily damaged by the AFM tip and difficult to image without distortion. Tapping (intermittent contact) mode is highly recommended because it minimizes lateral (dragging) forces on the sample, reducing damage and friction compared to contact mode [1]. For nanomechanical mapping, advanced modes like PeakForce Tapping can provide superior force control, significantly reducing sample damage and image artifacts by managing the maximum force applied to the sample at each pixel [8].
Q2: What are the best practices for immobilizing soft biofilm cells without altering their native properties?
Secure yet benign immobilization is critical. Methods can be broadly categorized as mechanical or chemical [1].
- Mechanical Entrapment: Using porous membranes or polydimethylsiloxane (PDMS) stamps with micro-wells to physically trap cells. This method is generally benign but can be sporadic [1].
- Chemical Immobilization: Coating substrates (e.g., glass, mica) with agents like poly-L-lysine or aminopropyltriethoxy silane (APTES) to promote electrostatic adhesion [1] [9]. While offering strong attachment, some chemical agents can affect cell viability and nanomechanical properties. A promising benign method is using substrates treated with divalent cations (e.g., Mg²⁺, Ca²⁺), which can promote optimal attachment without a significant reduction in viability [1].
Q3: How can I address the heterogeneity of biofilms to ensure my AFM data is representative?
Traditional AFM's small scan area (<100 µm) makes it difficult to capture the full spatial complexity of millimeter-scale biofilms [5]. To overcome this:
- Automated Large-Area AFM: Implement systems that automate the collection and stitching of multiple high-resolution images over millimeter-scale areas. This bridges the gap between cellular and macro-scale organization [5].
- Machine Learning (ML): Utilize ML-based image segmentation and analysis to automatically and quantitatively analyze large datasets, extracting parameters like cell count, confluency, and orientation across large, heterogeneous areas [5].
Q4: What are the main sources of noise and artifacts, and how can they be minimized?
Common sources and their mitigations are summarized in Table 1. Key strategies include:
- Tip Selection: Using sharp, clean, and appropriately shaped probes to avoid tip artifacts [2].
- Environmental Control: Placing the AFM on an active anti-vibration table in a quiet location to minimize environmental noise [2].
- System Calibration: Regularly calibrating the system using standardized protocols and calibration samples (e.g., silicon gratings) to reduce scanner nonlinearities and hysteresis effects [8].
Experimental Protocols for Key Analyses
Protocol 1: Cell Immobilization for Single-Cell Topography
This protocol outlines steps to securely immobilize microbial cells for high-resolution AFM imaging under aqueous conditions [1].
1. Substrate Preparation:
- Clean a glass coverslip or mica disk with appropriate solvents and plasma cleaning to remove organic contaminants.
- Option A (Chemical): Functionalize the substrate by coating it with a 0.1% w/v solution of poly-L-lysine for 15 minutes, then rinse gently with deionized water and air dry [1] [9].
- Option B (Mechanical): Use a pre-fabricated PDMS stamp with microwells designed to fit the target cell size [1].
2. Cell Deposition:
- Deposit a small volume (e.g., 20 µL) of a concentrated cell suspension onto the prepared substrate.
- Allow cells to settle and adhere for 15-30 minutes in a humidified chamber to prevent evaporation.
3. Rinsing:
- Gently rinse the substrate with a mild buffer (e.g., PBS or a low-concentration HEPES buffer) to remove any non-adherent, planktonic cells. The goal is to leave only firmly attached cells for imaging [5].
4. Hydration:
- Immediately place the substrate into the AFM liquid cell and add the appropriate imaging buffer to keep the cells hydrated throughout the experiment.
Protocol 2: Nanomechanical Mapping via Force Spectroscopy
This protocol details the use of force-distance curves to measure the mechanical properties of biofilm components [1] [9].
1. Probe and Mode Selection:
- Select a cantilever with a well-defined tip geometry and a spring constant appropriate for soft samples (typically 0.01 - 0.5 N/m). Calibrate the cantilever's spring constant and sensitivity beforehand [9].
- Choose the force spectroscopy or nanomechanical imaging mode on your AFM system.
2. Reference Measurement:
- Acquire force-distance curves on a clean, rigid reference surface (e.g., clean silicon wafer) to define the "zero" indentation point and characterize the tip shape.
3. Sample Measurement:
- Systematically acquire an array of force-distance curves across the area of interest on the biofilm sample. A sufficient number of curves should be collected for statistical relevance.
4. Data Analysis:
- Indentation Depth: For each curve, calculate the indentation (δ) by comparing the slope of the curve on the sample to the reference curve on the rigid surface [1].
- Model Fitting: Fit the approach segment of the force curve to a mechanical model, such as the Hertz model, to extract the Young's modulus (E). The Hertz model for a parabolic tip is given by: ( F = \frac{4}{3} \frac{E}{1-\nu^2} \sqrt{R} \delta^{3/2} ) where F is force, E is Young's modulus, ν is the Poisson's ratio (often assumed to be 0.5 for soft, incompressible materials), R is the tip radius, and δ is the indentation depth [1].
Diagram 1: Workflow for nanomechanical mapping of biofilms.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagents and Materials for AFM Biofilm Studies
| Item Name | Function/Application | Key Considerations |
|---|---|---|
| Poly-L-Lysine | Adhesion-promoting coating for substrates to immobilize cells chemically [1] [9]. | Effective for attachment but may affect cell viability and native mechanical properties; use lower concentrations [1]. |
| Polydimethylsiloxane (PDMS) Stamps | Micro-structured stamps for mechanical entrapment and immobilization of individual cells [1]. | Provides benign immobilization without chemicals; microstructure dimensions must be tailored to the specific cell size [1]. |
| Silicon Nitride AFM Probes | Standard probes for contact and tapping mode imaging in liquid [1]. | Choose a sharp tip radius for high resolution and a low spring constant (e.g., 0.01 - 0.5 N/m) for soft samples to minimize damage [1] [9]. |
| Conical/Tapped-Up Tips | High-aspect-ratio tips for imaging complex, heterogeneous biofilm structures with deep features [2]. | Superior to pyramidal tips for resolving steep-edged features and trenches common in 3D biofilm architectures [2]. |
| Calibration Gratings | Reference standards (e.g., silicon gratings with precise step heights) for calibrating AFM scanner accuracy [8]. | Essential for ensuring dimensional accuracy and correcting for scanner nonlinearities and thermal drift [8]. |
| PFOTS (Perfluorooctyltrichlorosilane) | A chemical used to create hydrophobic surfaces on substrates like glass to study its effect on bacterial attachment and biofilm assembly [5]. | Useful for investigating how surface chemistry and hydrophobicity influence initial cell attachment and subsequent biofilm formation [5]. |
Diagram 2: Relating core obstacles to AFM solutions and essential tools.
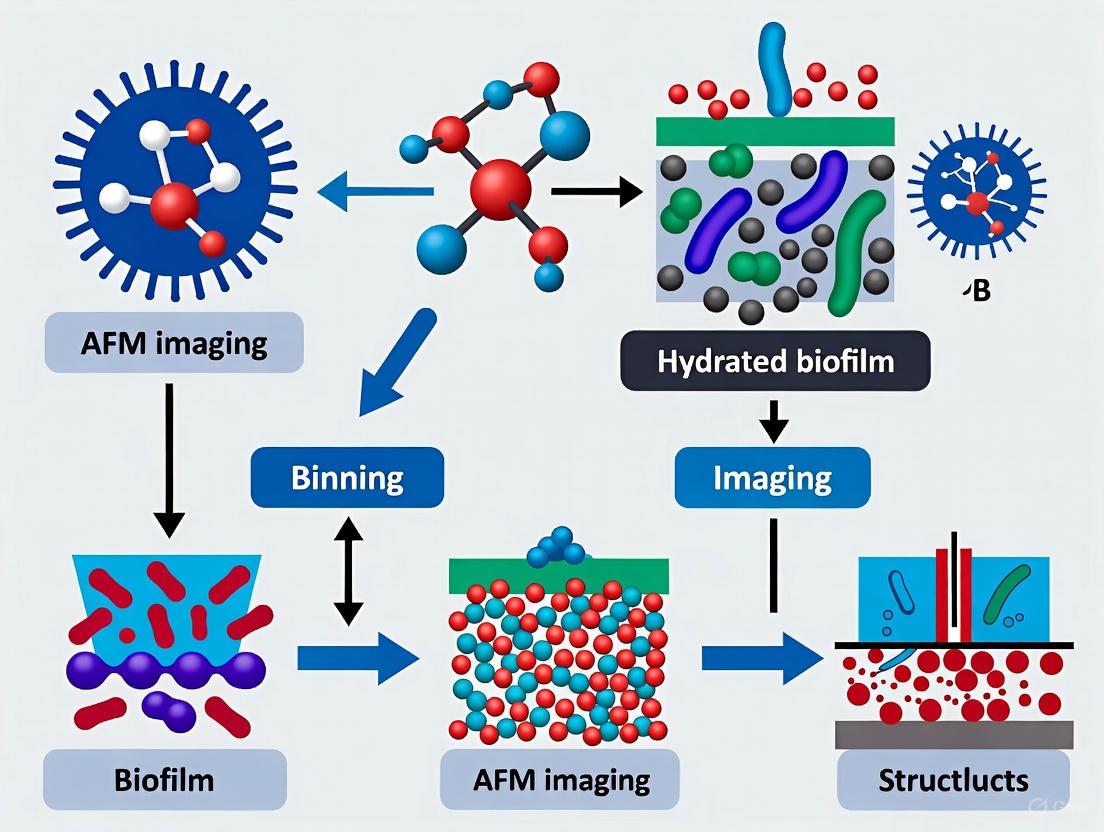
Atomic Force Microscopy (AFM) has established itself as a powerful tool for investigating microbial biofilms, providing unprecedented nanoscale resolution of structural and functional properties at the cellular and even sub-cellular level. However, a fundamental limitation has persistently hampered its effectiveness for comprehensive biofilm studies: the significant scale mismatch between AFM's traditional imaging areas (typically less than 100×100 μm) and the millimeter-scale spatial heterogeneity that defines functional biofilm architectures. This technical constraint has previously restricted the ability to link critical nanoscale features, such as individual cell appendages and matrix components, to the emergent macroscale organization and behavior of biofilms. This article establishes a technical support framework to address this challenge, providing researchers with methodologies and troubleshooting guides to bridge this scale gap effectively.
Table 1: The Scale Mismatch Problem in Conventional AFM Biofilm Imaging
| Aspect | Conventional AFM Capability | Biofilm Requirement | Consequence of Mismatch |
|---|---|---|---|
| Imaging Area | < 100 μm × 100 μm [5] | Millimeter-scale areas [5] | Inability to capture spatial complexity and representativeness |
| Resolution | Nanoscale (can visualize flagella ~20-50 nm) [5] | Nanoscale to mesoscale | Detailed features not linked to larger community structure |
| Throughput | Slow, labor-intensive [5] | High-throughput for statistics | Limited data on dynamic changes and heterogeneity |
| Data Integration | Single, small images | Stitched, large-area maps | Fragmented understanding of biofilm architecture |
Technical Solutions: Methodologies for Large-Area Analysis
Automated Large-Area AFM Imaging
Recent advances have begun to address this scale limitation through the development of automated large-area AFM approaches. This methodology involves automating the scanning process to capture multiple contiguous high-resolution images over millimeter-scale areas, effectively creating a detailed map of the biofilm surface [5]. The process requires specific instrumentation and software capable of precise stage movement and automated image capture sequences, overcoming the traditional restrictions imposed by piezoelectric actuator constraints.
Key Experimental Protocol: Large-Area AFM for Early Biofilm Formation
- Sample Preparation: Grow Pantoea sp. YR343 (or your target organism) on PFOTS-treated glass coverslips. At selected time points (e.g., 30 minutes for initial attachment), remove the coverslip, gently rinse to remove unattached cells, and air-dry prior to imaging [5].
- Instrument Setup: Configure the AFM for large-area scanning. This typically involves defining a grid pattern over the desired millimeter-scale area.
- Automated Imaging: Initiate the automated sequence to capture high-resolution images (e.g., 10×10 μm or larger) at each grid point with minimal user intervention.
- Image Stitching: Use integrated software or algorithms to seamlessly stitch the individual images into a single, large-area map. Machine learning (ML) approaches can significantly aid this process, especially with minimal feature overlap between individual scans [5].
- Data Analysis: Apply ML-based image segmentation and analysis to automatically extract quantitative parameters from the large-area map, such as cell count, confluency, cell shape, and orientation [5].
Figure 1: Workflow for automated large-area AFM imaging of biofilms.
Machine Learning and Data Analysis Integration
The high-volume, information-rich data generated by large-area AFM necessitates robust computational tools. Machine learning (ML) and artificial intelligence (AI) are transforming this aspect by enabling automated data processing and analysis [5]. ML applications crucial for biofilm research include:
- Automated Image Stitching: Creating seamless large-area maps from individual scans with limited overlap [5].
- Cell Detection and Classification: Automatically identifying and categorizing cells within complex images [5].
- Segmentation and Defect Detection: Isolating specific features of interest, such as individual cells, flagella, or regions of EPS [5].
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Research Reagent Solutions for Biofilm AFM
| Item | Function/Description | Application Example |
|---|---|---|
| PFOTS-Treated Glass | Creates a hydrophobic surface to promote specific bacterial attachment and study surface modification effects. | Investigating organization of Pantoea sp. YR343 during early biofilm assembly [5]. |
| Silicon or Silicon Nitride AFM Probes | Sharp tips mounted on cantilevers that physically probe the sample surface. | Standard topographical and mechanical property imaging [10]. |
| High-Aspect Ratio (HAR) Probes | Probes with a high height-to-width ratio, allowing them to accurately resolve deep, narrow trenches. | Imaging highly non-planar features or complex EPS structures [2]. |
| Conical-Tipped Probes | Superior to pyramidal tips for accurately tracing steep-edged features. | Profiling complex biofilm topography with vertical heterogeneity [2]. |
| Polydimethylsiloxane (PDMS) Stamps | Micro-structured stamps used for mechanical immobilization of microbial cells. | Spatially controlled trapping of spherical cells for live imaging [1]. |
| Polycarbonate Membranes | Porous membranes with pore size comparable to cell size for gentle physical entrapment. | Immobilizing single bacterial, yeast, or fungal cells under aqueous conditions [10]. |
| Aminosilane-Modified Substrates | Chemically functionalized surfaces (e.g., glass) for covalent bonding of cells. | Strong immobilization of cells for force spectroscopy measurements [10]. |
Troubleshooting Guide: FAQs for Biofilm AFM Experiments
Q1: Our large-area scans show unexpected, repeating patterns or widened features. What is the likely cause and solution?
- Cause: This is typically a tip artifact, often caused by a contaminated or broken AFM tip. A blunt tip will make structures appear larger and trenches smaller than they are [2].
- Solution: Replace the AFM probe with a new, clean one. To prevent contamination, ensure your sample preparation protocols minimize loosely adhered material [2]. For high-resolution imaging of biofilms, consider using conical-shaped tips for more accurate profiling [2].
Q2: We are having difficulty imaging the bottom of deep trenches or valleys in our heterogeneous biofilm. How can we improve this?
- Cause: This is often due to using a probe with an inappropriate shape or low aspect ratio. The tip apex cannot reach the bottom of deep features, leading to inaccurate topography [2].
- Solution: Switch to a High-Aspect Ratio (HAR) AFM probe. HAR probes are specifically designed to fit inside and accurately resolve deep and narrow features, providing a true representation of the biofilm's 3D structure [2].
Q3: Our images have repetitive horizontal lines across them. What sources of noise should we investigate?
- Cause A: Electrical Noise. This often appears at 50/60 Hz and its harmonics. You can identify it by comparing the noise frequency to your scan rate [2].
- Solution: Imaging during quieter periods (e.g., early mornings/late evenings) when electrical noise is lower can sometimes help. This is often governed by building circuitry [2].
- Cause B: Laser Interference. On highly reflective samples, laser light reflecting off the sample surface can interfere with the signal from the cantilever [2].
- Solution: Use a probe with a reflective coating (e.g., gold or aluminum), which helps to prevent this interference [2].
- Cause C: Environmental Vibration. Vibrations from building doors, people, or traffic can introduce streaks [2].
- Solution: Ensure the anti-vibration table is functioning. Relocate the instrument to a quieter location (e.g., a basement lab) or place a "STOP AFM in Progress" sign to alert colleagues [2].
Q4: How can we effectively immobilize hydrated, live bacterial cells for AFM without affecting their viability or nanomechanical properties?
- Solution: This is a critical step for successful imaging. A comparison of common methods is provided below.
Table 3: Cell Immobilization Strategies for Hydrated Biofilm AFM
| Method | Procedure | Advantages | Disadvantages |
|---|---|---|---|
| Mechanical Entrapment (Porous Membrane) [10] | Concentrated cell suspension is gently sucked through a polycarbonate membrane with pore size matching cell dimensions. | Minimizes denaturation of surface molecules; suitable for aqueous imaging. | Works best for spherical cells; can be sporadic for rod-shaped cells. |
| Mechanical Entrapment (PDMS Microstamps) [1] | Use lithographically patterned PDMS stamps to physically trap cells of specific sizes. | High level of immobilization; allows for controlled cell orientation. | Requires fabrication of specific masters; best for spherical cells. |
| Chemical Fixation (Aminosilane + EDC/NHS) [10] | Covalently bond cells to aminosilane-modified glass slides using cross-linkers (EDC/NHS). | Very strong attachment, withstands lateral scanning forces. | Chemical treatment may alter surface properties and viability. |
| Physico-Chemical Adhesion [1] | Use of divalent cations (Mg²⁺, Ca²⁺) and glucose to promote attachment to substrates. | Benign, does not force physiological changes; maintains viability. | Attachment strength may be variable and less robust. |
Figure 2: A logical troubleshooting guide for common AFM imaging artifacts.
The scale mismatch between AFM's nanoscale resolution and biofilm's millimeter-scale architecture is no longer an insurmountable obstacle. By adopting automated large-area scanning techniques, integrating machine learning for data analysis and stitching, and applying rigorous troubleshooting and sample preparation protocols, researchers can now bridge these scales. This empowers the scientific community to unravel the complex spatial heterogeneity of biofilms with unprecedented detail, accelerating the development of effective strategies to control and manipulate these resilient microbial communities in medical, industrial, and environmental contexts.
Atomic Force Microscopy (AFM) is a powerful tool for studying hydrated biofilm structures, capable of providing high-resolution topographical and mechanical properties under physiological conditions. However, a significant challenge in this research is minimizing probe-sample interactions that can damage the delicate, native structure of biofilms. This technical support center article addresses common issues and provides solutions for researchers aiming to obtain accurate data while preserving biofilm integrity.
Frequently Asked Questions (FAQs)
1. My biofilm images appear blurry and lack fine detail. What could be causing this? This is often a symptom of "false feedback," where the AFM probe interacts with a surface contamination layer or electrostatic forces instead of the sample's hard surface forces. This is common in humid environments or with samples exposed to air for long periods. To resolve this, increase the probe-surface interaction force by decreasing the setpoint value in vibrating (tapping) mode or increasing it in non-vibrating (contact) mode to push the probe through the contamination layer [11].
2. I see repetitive patterns or duplicated features in my images that don't match my sample. What is happening? This is typically a tip artifact, indicating a blunt, broken, or contaminated AFM probe. A damaged tip can cause irregular shapes to repeat across the image, make structures appear larger, and make trenches appear smaller. The solution is to replace the probe with a new, sharp one. To prevent this, ensure proper handling of probes and use an ESD bracelet to avoid electrostatic discharge that can damage the tip [2] [12].
3. Why can't I accurately image deep, narrow trenches or vertical structures in my biofilm? This problem arises from using a probe with an inappropriate shape or low aspect ratio. Pyramidal or tetrahedral tips have sidewalls that can prevent the tip apex from reaching the bottom of fine features. Switch to a conical tip or a High Aspect Ratio (HAR) probe, which are designed to accurately resolve steep-edged features and deep trenches common in biofilm architectures [2].
4. How can I minimize damage to soft, hydrated biofilm samples during scanning? For delicate samples, consider using True Non-Contact Mode. This mode operates by detecting attractive van der Waals forces without making physical contact, preventing tip wear and sample damage. It is particularly suited for imaging soft, sticky, or brittle samples that could be damaged by tapping mode [13]. Additionally, using softer cantilevers (with lower force constants) can reduce interaction forces on delicate samples [12].
Troubleshooting Guides
Problem: Excessive Sample Damage and Deformation
Potential Causes and Solutions:
- Cause: Excessive imaging force due to an overly stiff cantilever.
- Solution: Select a cantilever with a lower force constant (stiffness). For soft biological samples like biofilms, choose a stiffness comparable to or slightly stiffer than the sample itself [12].
- Cause: Operating in a repulsive force regime on a delicate sample.
- Solution: Switch to a non-destructive imaging mode. True Non-Contact Mode maintains a small, stable distance from the sample by sensing attractive forces, virtually eliminating sample damage [13].
- Cause: High adhesion forces in a contaminated or sticky sample.
Problem: Persistent Image Artifacts
Potential Causes and Solutions:
- Cause: Tip degradation or contamination.
- Cause: Electrical or environmental noise.
- Solution: Identify noise sources. Electrical noise (50/60 Hz) can sometimes be mitigated by imaging during quieter times (e.g., early mornings). Environmental vibrations can be reduced by ensuring anti-vibration tables are functional and using acoustic enclosures. A "STOP AFM in progress" sign can alert others to minimize activity [2].
- Cause: Laser interference on reflective samples.
- Solution: Use a probe with a reflective coating (e.g., gold or aluminium) on the cantilever. This coating prevents spurious laser light from the sample surface from interfering with the primary signal at the photodetector [2].
Quantitative Data for AFM of Biofilms
Table 1: AFM Cantilever Selection Guide for Biofilm Imaging
| Parameter | Recommended Range for Soft Biofilms | Functional Impact |
|---|---|---|
| Force Constant | < 1 N/m to 5 N/m | Softer cantilevers reduce deformation; slightly stiffer ones help with sticky surfaces [12]. |
| Resonant Frequency | > 300 kHz | Higher frequencies allow faster scanning and reduce sample damage [12]. |
| Tip Radius | < 10 nm (sharp) | A sharper tip provides higher resolution, allowing visualization of fine features like flagella [5] [12]. |
| Tip Aspect Ratio | High (conical preferred) | Enables accurate imaging of deep trenches and vertical structures in biofilm clusters [2]. |
| Q Factor | High | Indicates low damping, leading to greater sensitivity to sample profile [12]. |
Table 2: Comparison of AFM Operational Modes for Biofilm Integrity
| Imaging Mode | Probe-Sample Interaction | Risk of Sample Damage | Best for Biofilm Applications |
|---|---|---|---|
| True Non-Contact Mode | Attractive van der Waals forces only | Very Low | Ideal for pristine imaging of delicate, hydrated structures and unbaked polymers [13]. |
| Tapping Mode | Intermittent contact (repulsive forces) | Moderate | A versatile balance between resolution and sample protection for most biofilm samples [11]. |
| Contact Mode | Constant physical contact | High | Generally not recommended for delicate, hydrated biofilms due to high shear forces. |
Experimental Protocols
Protocol: Large-Area, High-Resolution AFM of Native Biofilm Structures
This protocol, adapted from recent research, details a method for imaging the native structure of biofilms over millimeter-scale areas with minimal damage [5].
1. Sample Preparation (Pantoea sp. YR343 Biofilm)
- Surface Treatment: Use glass coverslips treated with PFOTS to create a hydrophobic surface for controlled biofilm attachment [5].
- Inoculation: Inoculate a petri dish containing the treated coverslips with bacteria in a liquid growth medium.
- Incubation & Harvesting: Incubate for a desired time (e.g., 30 minutes for initial attachment studies). Gently remove and rinse the coverslip to eliminate non-adherent cells.
- Drying: Air-dry the sample before imaging. For fully hydrated imaging, specialized liquid cells would be used [5].
2. AFM Setup and Imaging
- Probe Selection: Choose a sharp, high-resolution probe with parameters from Table 1 (e.g., force constant ~1-5 N/m, resonant frequency >300 kHz).
- Mounting: Secure the prepared sample onto the AFM sample stage.
- Image Acquisition:
- Engage the probe using the automated approach, carefully monitoring for false feedback [11].
- Use True Non-Contact or Tapping Mode to scan the sample. For large-area analysis, employ an automated large-area AFM system that captures multiple high-resolution images over millimeter-scale areas.
- Use machine learning-assisted algorithms for seamless stitching of individual image tiles [5].
3. Data Analysis
- Image Processing: Use software to stitch large-area images and correct for drift and tilt.
- Feature Analysis: Implement machine learning-based segmentation to automatically extract quantitative parameters such as cell count, confluency, cellular orientation, and flagellar distribution [5].
The workflow for this protocol is summarized in the following diagram:
Logical Pathway for Minimizing Probe-Sample Damage
The following decision tree guides the selection of the optimal strategy to preserve sample integrity:
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for AFM Biofilm Imaging
| Item | Function/Application |
|---|---|
| PFOTS-treated Glass Coverslips | Creates a controlled hydrophobic surface for studying initial bacterial attachment and biofilm assembly dynamics [5]. |
| Sharp Etched Silicon Probes | High-resolution tips (radius < 10 nm) are essential for visualizing subcellular features like flagella and pili without distortion [5] [12]. |
| Soft Cantilevers (Force Constant: ~1-5 N/m) | Minimizes loading force on delicate biofilm structures, preserving native morphology and preventing deformation during scanning [12]. |
| High Aspect Ratio (HAR) Conical Tips | Enables accurate topography measurement of deep, narrow valleys and high vertical features within heterogeneous biofilm clusters [2]. |
| Liquid Cell Setup | Allows AFM imaging to be performed under physiological buffer conditions, maintaining biofilm hydration and native state [14]. |
| Optimal Cutting Temperature (OCT) Compound | An aqueous embedding medium for cryo-preservation of tissue or biofilm samples prior to cryo-sectioning for AFM analysis [15]. |
Advanced AFM Techniques for High-Resolution Biofilm Characterization
Atomic Force Microscopy (AFM) provides unparalleled nanoscale resolution for studying hydrated biofilm structures, critical for understanding their development and resistance mechanisms. The central challenge for researchers lies in choosing an operating mode that minimizes disturbance to these soft, dynamic biological systems while still generating high-fidelity data. In liquids, where many biofilm experiments are conducted, this choice is paramount. The decision between Tapping Mode and Contact Mode fundamentally influences image quality, sample integrity, and the biological relevance of your results. This guide provides a direct, troubleshooting-focused comparison to help you select and optimize the right mode for your specific experimental needs in liquid environments.
Head-to-Head Comparison: Tapping Mode vs. Contact Mode
The following table summarizes the key operational differences and performance characteristics of each AFM mode in liquid environments, based on quantitative data and typical use cases.
Table 1: AFM Mode Comparison for Liquid Imaging
| Feature | Contact Mode | Tapping Mode in Liquid |
|---|---|---|
| Basic Principle | AFM tip is in constant contact with the sample surface [16] [17]. | Cantilever oscillates at or near its resonance frequency; tip intermittently contacts the surface [16] [17]. |
| Tip-Sample Interaction | Constant deflection maintained, equivalent to constant interaction force [16]. | Constant oscillation damping maintained, equivalent to constant interaction force [16]. |
| Typical Forces | Higher (x1 nN - x100 nN) [16]. | Lower (forces significantly reduced) [16]. |
| Lateral Forces | Significant, can distort features and cause sample damage [17]. | Negligible, as the tip only touches at the bottom of its swing [16]. |
| Ideal Sample Type | Hard, flat surfaces without sharp edges or loose debris [16]. | Soft, fragile, and hydrated samples like biofilms [16]. |
| Handling Contamination | Prone to false feedback from fluid layers; requires increased force to penetrate [18]. | Superior; stiff cantilevers have enough energy to overcome adhesive forces in the fluid layer [16] [18]. |
| Scan Speed | High scan speeds possible [17]. | Slower than Contact Mode [17]. |
| Ease of Use | Fewer parameters to control; more suitable for beginners [16]. | Additional parameters to control related to oscillatory motion [16]. |
| Common Cantilevers | Softer cantilevers (C ≤ 1 N/m, f₀ ≤ 15 kHz) [16]. | Stiffer cantilevers (C ~ 40 N/m, f₀ ~ x100 kHz) to avoid sticking in liquid [16]. |
| Unique Capabilities | Lateral force measurement; essential for C-AFM, TUNA, SSRM [16]. | Phase imaging; essential for EFM, MFM, SCM [16]. |
Troubleshooting FAQs and Guides
FAQ 1: Why are my images of biofilms in liquid blurry and lacking detail?
Probable Cause: "False feedback," where the AFM's automated tip approach is tricked into stopping before the probe interacts with the sample's hard forces. In liquid, this is often caused by a thick contamination layer or electrostatic forces [18].
Solutions:
- For Tapping Mode: Decrease the setpoint value to increase the tip-sample interaction force, pushing the probe through the contamination layer [18].
- For Contact Mode: Increase the setpoint value to achieve the same effect of increasing interaction force [18].
- General Practice: Ensure thorough cleaning of your substrate and sample preparation to minimize loosely adhered material or contaminants [2].
FAQ 2: I see repetitive lines or streaks across my image. What is happening?
Probable Cause: Environmental noise or vibration, or loose particles on the sample surface interacting with the tip [2].
Solutions:
- Check your environment: Ensure the anti-vibration table is functional. Image during quieter times (e.g., early morning, late evening) to minimize noise from building vibrations [2].
- Use an acoustic enclosure if available.
- Improve sample preparation: Protocols should minimize loosely adhered material. If a loose particle adheres to the tip, it can cause persistent streaking and requires changing the probe [2].
FAQ 3: My biofilm appears damaged or features are being moved by the tip. How can I prevent this?
Probable Cause: Excessive lateral (shear) forces and normal forces applied to the soft sample, which is a hallmark risk of Contact Mode [16] [17].
Solutions:
- Switch to Tapping Mode: This is the primary solution. Tapping Mode virtually eliminates lateral forces, drastically reducing sample damage and deformation [16] [17].
- Softer Cantilevers (Contact Mode): If you must use Contact Mode, switch to a cantilever with a lower spring constant to reduce the applied force.
- Reduce Setpoint: In either mode, further reduce the force applied to the sample by adjusting the setpoint.
FAQ 4: Why is my tip getting stuck to the sample surface in liquid?
Probable Cause: Strong adhesive forces, such as capillary forces from fluid layers or electrostatic attraction [18].
Solutions:
- Use Stiffer Cantilevers: This is particularly effective in Tapping Mode. The higher energy of the oscillation helps the tip overcome adhesive forces [16] [18].
- Adjust Oscillation Parameters: In Tapping Mode, increasing the drive amplitude can provide more energy to pull the tip free from adhesions.
- Create a Conductive Path: If electrostatic forces are the issue, creating a conductive path between the cantilever and sample can help dissipate charge [18].
Experimental Protocol: Imaging Early Biofilm Formation in Liquid
This protocol outlines a methodology for high-resolution imaging of bacterial surface attachment and early biofilm formation, adapted from recent research [5].
Aim: To visualize the topographical and structural details of surface-attached bacterial cells and their appendages (e.g., flagella) under physiological liquid conditions.
Materials & Reagents:
- Bacterial Strain: e.g., Pantoea sp. YR343 or other relevant gram-negative bacterium [5].
- Growth Medium: Appropriate liquid growth medium (e.g., Lysogeny Broth).
- Substrate: PFOTS-treated glass coverslips or other suitably modified surfaces to promote/prohibit adhesion [5].
- AFM: System capable of operation in liquid cell.
- AFM Probes: Sharp, stiff cantilevers recommended for Tapping Mode in liquid (e.g., nominal spring constant ~40 N/m, resonance frequency ~x100 kHz) [16].
Procedure:
- Sample Preparation:
- Inoculate a Petri dish containing your prepared substrate (e.g., PFOTS-treated coverslip) with the bacterial culture in liquid growth medium.
- Incubate at the appropriate temperature for a selected time (e.g., ~30 minutes for initial attachment studies) [5].
- At the time point, carefully remove the coverslip from the Petri dish.
- Gently rinse with a buffer solution (e.g., PBS or purified water) to remove non-adherent planktonic cells. Avoid harsh rinsing that could disrupt attached cells.
- For imaging in liquid, place the coverslip in the AFM liquid cell and immerse in the appropriate buffer. For high-resolution imaging of fine structures like flagella, the sample may be air-dried, though this sacrifices physiological conditions [5].
- AFM Setup and Imaging:
- Mount a Tapping Mode-optimized cantilever into the holder.
- Assemble the liquid cell and engage the fluid.
- Perform the laser alignment and photodetector adjustment on the cantilever in liquid.
- Find a resonant peak of the cantilever in liquid (its frequency will be significantly lower than in air).
- Engage the tip approach. Monitor for a stable engagement.
- Begin scanning at a slow scan rate (e.g., 0.5-1 Hz) to maximize resolution of delicate biological features. Optimize the setpoint and drive amplitude to achieve a clear, stable image with minimal force.
The workflow for this experimental protocol is summarized in the following diagram:
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for AFM Biofilm Imaging
| Item | Function/Benefit | Example/Specification |
|---|---|---|
| PFOTS-treated Glass | Creates a hydrophobic surface to study controlled bacterial adhesion and early biofilm assembly [5]. | (Tridecafluoro-1,1,2,2-tetrahydrooctyl)trichlorosilane treated coverslips. |
| Tapping Mode Cantilevers | Stiff, sharp probes for stable oscillation in liquid, minimizing adhesion and sample damage [16]. | Nominal spring constant ~40 N/m, resonance frequency ~300 kHz in air (lower in liquid) [16]. |
| Contact Mode Cantilevers | Softer levers for maintaining constant force on hard samples; not ideal for soft biofilms. | Nominal spring constant ≤ 1 N/m, low resonance frequency [16]. |
| Liquid Cell | Enables AFM imaging under physiological buffer conditions, preserving native biofilm state. | Sealed cell to prevent evaporation; compatible with various buffers (e.g., PBS). |
| Flagella-Deficient Mutant | A critical control strain to confirm the identity of nanoscale appendages imaged by AFM [5]. | e.g., ΔfliC mutant of the studied bacterial strain. |
Decision Framework for AFM Mode Selection
Use the following logic diagram to guide your choice between Contact and Tapping Mode for imaging hydrated biofilms.
Strategies for Effective Cell and Biofilm Immobilization
This guide addresses a central challenge in biofilm research: securing fragile, hydrated biofilm structures for reliable Atomic Force Microscopy (AFM) analysis. Effective immobilization is the critical first step for obtaining high-resolution nanoscale data on biofilm morphology, mechanics, and interactions. The following troubleshooting guides, FAQs, and protocols are designed to help researchers overcome common experimental hurdles.
Troubleshooting Guide: Common Immobilization Challenges
Table 1: Troubleshooting Common AFM Biofilm Immobilization Issues
| Problem | Possible Causes | Recommended Solutions |
|---|---|---|
| Cells detach during AFM scanning [1] | Weak adhesion forces; excessive lateral scanning forces from the AFM tip [1]. | Use mechanical entrapment in porous membranes or PDMS micro-wells [1]. Chemically functionalize substrate with poly-L-lysine or enhance adhesion with divalent cations (Mg²⁺, Ca²⁺) [1]. |
| Poor image quality on hydrated samples [19] [1] | Sample is too soft and diffuse; tip-sample interactions distort native structure [1]. | For delicate structures, use tapping mode AFM in liquid to minimize shear forces [1]. For cohesive strength measurements, maintain high humidity (~90%) to preserve biofilm-water content without full submersion [19]. |
| Inability to identify structures in AFM topographs | AFM lacks inherent chemical specificity; unknown topographic features are hard to distinguish [20]. | Combine AFM with epifluorescence microscopy (EFM). Stain specific components (e.g., DNA with DAPI) for correlation between fluorescence and topography [20]. |
| Low throughput and irreproducible data | Manual AFM operation limits scan area and consistency; small scans may not represent the whole biofilm [5]. | Implement automated large-area AFM scanning. Use machine learning algorithms to stitch images and analyze data over millimeter-scale areas [5]. |
Frequently Asked Questions (FAQs)
Q1: Why is chemical fixation sometimes avoided for cell immobilization? While chemical treatments like cross-linkers provide strong adhesion, they can alter the native nanomechanical properties and viability of the cells, which is undesirable for live-cell experiments [1].
Q2: What is the key advantage of using a polyester nonwoven carrier for immobilization? Polyester nonwovens provide a high surface area with pore spaces that trap moisture and cells, facilitating high cell density immobilization and robust biofilm formation suitable for continuous fermentation processes [21].
Q3: How can I measure the cohesive strength of a hydrated biofilm without drying it? A specialized AFM method can measure cohesive energy in moist biofilms. It involves calculating the frictional energy dissipated to abrade a defined biofilm volume under controlled humidity (e.g., 90%), providing values in nJ/μm³ [19].
Q4: My biofilm is heterogeneous. How can I ensure my AFM data is representative? Traditional AFM with small scan areas (<100 µm) struggles with this. Employ large-area automated AFM, which can perform high-resolution scans over millimeter-scale areas, capturing the true spatial complexity and heterogeneity of the biofilm [5].
Standard Experimental Protocols
Protocol 1: Mechanical Immobilization of Single Cells using PDMS Micro-Wells
This protocol is ideal for immobilizing spherical microbial cells for single-cell analysis without chemical modification [1].
- Fabricate a silicon master with microwells of specific dimensions (e.g., 1.5–6 µm wide, 1–4 µm deep) via deep reactive ion etching.
- Cast a Polydimethylsiloxane (PDMS) stamp from the silicon master and cure.
- Deposit the cell suspension onto the PDMS stamp. Use convective and capillary forces to guide cells into the microwells.
- Place the cell-loaded PDMS stamp onto a clean glass slide for AFM analysis. The wells physically restrain the cells during scanning.
Protocol 2: Correlative AFM and Epifluorescence Microscopy (EFM) on Opaque Substrates
This protocol allows for the precise correlation of topographic features with biological identity on opaque surfaces like minerals or medical implants [20].
- Grow biofilm on the substrate of interest (e.g., a pyrite coupon).
- Stain the sample with a fluorescent dye (e.g., DAPI for DNA) for 10 minutes.
- Mount the sample on a specialized shuttle stage glass slide.
- Image with EFM first to locate fluorescently labeled cells of interest and record the coordinates.
- Transfer the shuttle stage to the AFM. The system allows for relocating the same sample area with a precision of 3-5 µm.
- Perform AFM imaging (in air or liquid) on the pre-identified area to obtain high-resolution topographical data of the features seen in the fluorescence image.
Experimental Workflow Visualization
The following diagram illustrates the logical pathway for selecting an appropriate immobilization strategy based on experimental goals.
Research Reagent Solutions
Table 2: Essential Materials for Biofilm Immobilization and AFM Analysis
| Reagent / Material | Function / Application | Key Considerations |
|---|---|---|
| Poly-L-lysine [1] | Coats substrates to improve cell adhesion via electrostatic interactions. | A common chemical immobilization agent; may affect cell viability or mechanics [1]. |
| PDMS Micro-well Stamps [1] | Physically traps individual cells for single-cell AFM analysis. | Provides secure, orientation-controlled immobilization ideal for spherical cells [1]. |
| Divalent Cations (Mg²⁺, Ca²⁺) [1] | Added to suspension to strengthen binding of cells to substrates. | A gentler alternative to cross-linkers; helps maintain cell viability [1]. |
| Propidium Monoazide (PMA) [21] | Distinguishes viable from non-viable cells in conjunction with qPCR. | Useful for quantifying the viability of immobilized cells within a biofilm consortium [21]. |
| Polyester Nonwoven [21] | A fibrous, porous carrier for high-density cell immobilization in bioreactors. | Excellent for forming biofilms in flow-through systems for industrial biocatalysis [21]. |
| DAPI (4',6-diamidino-2-phenylindole) [20] | Fluorescent DNA stain for identifying bacterial cells in correlative AFM-EFM. | Allows confirmation that topographic features are cells, not abiotic material [20]. |
Frequently Asked Questions (FAQs)
General AFM Principles for Biofilm Research
What nanomechanical properties can AFM measure on biofilms? Atomic Force Microscopy (AFM) can quantitatively map several key nanomechanical properties of biofilms. The most common is the elastic modulus (or Young's modulus), which measures the sample's stiffness or resistance to elastic deformation [22]. AFM is also used to characterize viscoelastic properties, including the storage modulus (E', energy elastically stored), loss modulus (E", energy dissipated), and loss tangent (tan d, the ratio of E"/E'), which describe how the material's stiffness depends on the loading frequency [22]. Furthermore, AFM directly measures adhesion force, the attractive force between the AFM tip and the biofilm surface upon retraction, and friction, the force resisting lateral motion of the tip [22].
Why is AFM particularly suitable for studying hydrated biofilms? A principal advantage of AFM for biofilm research is its ability to perform measurements under physiological conditions, including in liquid buffers [5] [1]. This allows researchers to interrogate biofilms in their native, hydrated state without the dehydration required by techniques like electron microscopy, thereby preserving their natural structure and mechanical properties [1]. This capability is crucial for obtaining biologically relevant data.
What is the difference between contact mode, tapping mode, and PeakForce Tapping for soft samples? Choosing the correct imaging mode is critical for successfully characterizing soft, delicate biofilms without causing damage.
- Contact Mode: The tip is in constant physical contact with the surface. This mode can exert high lateral forces during scanning, which may damage soft samples or displace loosely attached cells and polymers [23].
- Tapping Mode: The cantilever is oscillated at resonance, and the tip only intermittently "taps" the surface. This significantly reduces lateral forces and sample damage, making it a preferred method for imaging soft biological samples [23].
- PeakForce Tapping: A non-resonant mode where the tip performs a force-distance curve at every pixel. It provides direct, quantitative control of the peak force applied to the sample, enabling high-resolution imaging and simultaneous mapping of topography, adhesion, and stiffness at forces as low as 10 pN. This makes it ideal for fragile biofilm structures [23].
Troubleshooting Common Experimental Challenges
My AFM images of a hydrated biofilm appear blurry and lack detail. What is wrong? This "blurry" image is a classic symptom of false feedback [24]. For biofilms in liquid, this often occurs because the tip is interacting with a soft, diffuse layer of extracellular polymeric substances (EPS) but has not reached the harder, underlying structures. The AFM's feedback loop is "tricked" into thinking it has found the surface, stopping the approach prematurely [24].
- Solution: Increase the tip-sample interaction force. In Tapping Mode, this is done by decreasing the setpoint amplitude. In PeakForce Tapping, increase the peak force setpoint. This will drive the tip through the soft EPS layer to achieve stable interaction with the firmer cellular structures [24].
I see repetitive patterns or my features look too wide. What is the cause? This is typically a tip artifact, indicating a contaminated or damaged AFM probe [2]. A blunt or contaminated tip will produce features that appear larger and broader than they are, and may create duplicated or "double" images of structures.
- Solution: Replace the AFM probe with a new, sharp one [2]. Always inspect your tips visually before use and be mindful of tip wear, especially when scanning large, rough areas of a biofilm.
How can I prevent bacterial cells from moving or detaching during scanning? Secure immobilization is one of the most critical steps for successful AFM of single cells within a biofilm [1]. Inadequate immobilization leads to cells being pushed around or swept away by the scanning tip.
- Solutions:
- Chemical Fixation: Use substrates functionalized with cell-adhesive coatings like poly-L-lysine or APTS (aminopropyltriethoxysilane) [1].
- Mechanical Trapping: Use porous membranes or specially fabricated PDMS (polydimethylsiloxane) micro-well stamps that physically trap cells of a specific size [1].
- Physiological Aids: For some bacteria, adding divalent cations (e.g., Mg²⁺, Ca²⁺) to the buffer can improve attachment to negatively charged surfaces like glass or mica without significantly affecting viability [1].
Troubleshooting Guide: Common Issues and Solutions
The table below summarizes frequent problems encountered during nanomechanical characterization of biofilms and their solutions.
| Problem | Possible Cause | Recommended Solution |
|---|---|---|
| Blurry, out-of-focus images in liquid | False feedback; tip trapped in soft EPS layer [24] | Increase tip-sample interaction (decrease amplitude setpoint in tapping mode; increase peak force setpoint in PeakForce Tapping) [24] |
| Cells are moved or swept by the tip | Inadequate cell immobilization [1] | Improve immobilization protocol (use poly-L-lysine coated surfaces, PDMS micro-wells, or add divalent cations to buffer) [1] |
| Repetitive patterns, broadened features | Contaminated or broken (blunt) AFM tip [2] | Replace the AFM probe with a new, sharp one [2] |
| Streaks or periodic noise in image | Environmental vibrations or electrical noise [2] | Ensure anti-vibration table is active; scan during quieter times; check for grounding issues and sources of electrical interference [2] |
| Inconsistent mechanical property maps | Poor force curve fit; inappropriate contact mechanics model | Ensure correct model selection (e.g., Hertz, DMT) for your sample and tip geometry; verify probe spring constant calibration [22] [1] |
| High adhesion obscuring other signals | Excessive capillary forces (in air); sticky EPS | Perform measurements fully submerged in liquid to eliminate meniscus forces; consider using a sharper, less adhesive probe [22] [24] |
Experimental Protocols & Methodologies
Core Protocol: Nanomechanical Mapping of a Hydrated Biofilm
This protocol details the steps for obtaining quantitative stiffness (DMTModulus) and adhesion maps of a hydrated biofilm using PeakForce Tapping mode [22].
Step-by-Step Methodology:
Sample Preparation:
- Grow your biofilm on a suitable substrate (e.g., glass coverslip, mica, or a treated surface).
- Crucially, immobilize the biofilm. Gently rinse with a compatible buffer (e.g., PBS) to remove non-adherent planktonic cells, leaving the adherent biofilm intact. For single-cell studies, use chemical (poly-L-lysine) or mechanical (PDMS stamps) immobilization methods [1].
- Mount the sample in the AFM liquid cell and carefully add buffer to fully submerge the biofilm.
AFM Probe Selection:
- Use a cantilever with a soft spring constant (typically 0.1 - 1 N/m) to avoid damaging the biofilm while maintaining sensitivity to force variations.
- Choose a tip with a sharp apex (nominal radius < 10 nm) for high spatial resolution.
- Pre-calibrated probes with a known spring constant are highly recommended for quantitative accuracy [23].
Instrument Setup and Engagement:
- Select PeakForce Tapping as the imaging mode.
- Set an initially low peak force setpoint (e.g., 0.1-0.5 nN) to minimize sample disturbance during engagement.
- Engage the probe with the surface. Once stable feedback is achieved, fine-tune the setpoint.
Parameter Optimization:
- Gradually increase the peak force setpoint until the topographic features are clear and stable. The goal is to use the minimum force necessary for clear imaging.
- Adjust the feedback gains to ensure the system is responsive but not oscillatory.
- Set a scan rate that balances data acquisition time with signal quality (typically 0.5-1.5 Hz).
Data Acquisition:
- Start scanning. The system will collect a force-distance curve at every pixel in the image.
- Channels to be collected simultaneously should include: Height, DMTModulus (elastic modulus), Adhesion, and Deformation [22].
Data Processing and Analysis:
- The AFM software will automatically fit each force curve using a contact mechanics model (e.g., the Derjaguin-Muller-Toporov (DMT) model for stiff samples with low adhesion) to calculate the DMTModulus and adhesion force [22].
- Apply a plane fit to the height image to level the data.
- Use histogram and cross-section analysis to quantify the distribution of mechanical properties across different regions of the biofilm.
The Scientist's Toolkit: Essential Research Reagents and Materials
The table below lists key materials and reagents essential for successful AFM-based nanomechanical characterization of biofilms.
| Item | Function/Application |
|---|---|
| Soft Cantilevers (0.1 - 1 N/m) | AFM probes with low spring constants are essential for sensitive force measurement on soft biological samples without causing damage [25]. |
| Sharp AFM Tips (<10 nm radius) | Tips with a sharp apex are required to achieve high spatial resolution, allowing the differentiation of individual cells and EPS structures [2]. |
| Poly-L-Lysine | A widely used adhesive coating for substrates (glass, mica) to chemically immobilize bacterial cells and prevent them from moving during scanning [1]. |
| PDMS Micro-well Stamps | Fabricated micro-structured stamps used for the mechanical immobilization of spherical microbial cells, providing organized and secure trapping [1]. |
| Mica or Glass Substrates | Atomically flat, pristine surfaces that are ideal for growing or depositing biofilms and cells for AFM analysis [1]. |
| Physiological Buffers (e.g., PBS) | Aqueous solutions used to maintain biofilm hydration and viability during liquid-mode AFM experiments [1]. |
| DMT / Hertz Contact Models | Analytical models used to fit the experimental force-distance curves and extract quantitative mechanical properties like elastic modulus [22] [1]. |
Advanced Applications: Large-Area AFM and Machine Learning
A major limitation of conventional AFM in biofilm research has been the small scan size (<100 µm), which makes it difficult to link nanoscale properties to the functional millimeter-scale architecture of biofilms [5]. Recent advances are overcoming this hurdle.
Large-Area Automated AFM: This approach automates the process of capturing and stitching together hundreds of high-resolution AFM images to create a seamless map over millimeter-scale areas [5]. This has revealed previously obscured spatial heterogeneities, such as honeycomb patterns of bacterial cells and the coordinated role of flagella in biofilm assembly beyond initial attachment [5].
Integration of Machine Learning (ML): The high-volume, information-rich data generated by these techniques is managed using ML. ML algorithms are used for tasks such as automated image stitching, cell detection, segmentation, and classification [5]. This enables efficient, quantitative analysis of parameters like cell count, confluency, shape, and orientation over very large areas, transforming AFM into a more high-throughput and objective tool for biofilm characterization [5].
FAQs: Core Technology and Applications
What is Large-Area Automated AFM and how does it address key challenges in biofilm research? Large-Area Automated AFM is an advanced imaging approach that combines hardware automation with machine learning to perform high-resolution atomic force microscopy over millimeter-scale areas [5]. Traditional AFM is limited by a small scan range (typically <100 µm), making it difficult to link high-resolution cellular features to the functional macroscale organization of biofilms [5]. This method overcomes the limitation by automating the scanning process, capturing multiple high-resolution images across a large surface, and using machine learning algorithms to seamlessly stitch them together, providing a comprehensive view of biofilm architecture from individual cells to entire communities [5] [26].
What level of quantitative data can this method provide for biofilm analysis? The integration of machine learning with large-area AFM enables the extraction of detailed quantitative data from massive datasets. In one demonstrated study, the system automatically analyzed more than 19,000 individual cells to generate detailed maps of cell properties across extensive surface areas [26]. This allows for the quantitative characterization of parameters such as cell count, confluency, cell shape, and orientation over biologically relevant scales [5].
Which AFM modes are most suitable for imaging delicate hydrated biofilm structures? For soft, fragile biological samples like hydrated biofilms, TappingMode and PeakForce Tapping are recommended over Contact Mode [23]. TappingMode oscillates the cantilever to minimize lateral forces that can damage samples [23]. PeakForce Tapping is particularly advanced as it performs a force curve at each pixel, enabling imaging at extremely low forces (down to ~10 pN) while simultaneously mapping mechanical properties, which is ideal for preserving sample integrity and achieving high resolution on delicate structures [23].
Can Large-Area AFM be used to test anti-biofilm surface strategies? Yes, this method is particularly powerful for screening and understanding surface modifications. Researchers have used it to characterize biofilm formation on engineered surfaces with nanoscale ridges, finding that specific patterns could disrupt normal biofilm organization [26]. This provides a valuable tool for identifying surface properties that resist bacterial adhesion and fouling [5] [26].
Troubleshooting Guides
Common Imaging Problems and Solutions
Table 1: Troubleshooting Common AFM Imaging Issues
| Problem | Possible Causes | Recommended Solutions |
|---|---|---|
| Unexpected/Repetitive Patterns [2] | - Contaminated or broken probe (tip artifact)- Electrical noise (50 Hz)- Laser interference | - Replace probe with a new, sharp one [2]- Image during quieter electrical periods (e.g., early morning) [2]- Use a probe with a reflective coating [2] |
| Blurry/Out-of-Focus Images (False Feedback) [27] | - Probe trapped in surface contamination layer- Electrostatic force between probe and sample | - Increase tip-sample interaction: Decrease setpoint in TappingMode; increase setpoint in Contact Mode [27]- Create conductive path between cantilever and sample; use a stiffer cantilever [27] |
| Difficulty with Vertical Structures/Trenches [2] | - Low aspect ratio or pyramidal probe shape | - Switch to a conical or High Aspect Ratio (HAR) probe [2] |
| Streaks in Images [2] | - Environmental noise/vibration- Loose particles on sample surface | - Ensure anti-vibration table is active; image in a quiet location [2]- Improve sample preparation to minimize loose material [2] |
Optimization for Large-Area Scans
Issue: Inefficient large-area data acquisition and analysis.
- Cause: Manual operation is time-consuming and prone to inconsistencies; large datasets are cumbersome to process.
- Solution: Leverage the integrated machine learning pipeline for autonomous operation. ML algorithms can optimize scanning site selection, automate the stitching of individual image tiles with minimal overlap, and perform high-throughput analysis of stitched images for tasks like cell detection and classification [5]. This automation allows for continuous, multi-day experiments without human supervision [5].
Experimental Protocol: Analyzing Biofilm Assembly on Modified Surfaces
The following protocol is adapted from the study "Analysis of biofilm assembly by large area automated AFM" which investigated Pantoea sp. YR343 biofilm formation on PFOTS-treated glass and silicon substrates [5].
1. Sample Preparation (Surface Treatment and Inoculation)
- Surface Functionalization: Treat glass coverslips with PFOTS (Perfluorooctyltrichlorosilane) to create a hydrophobic surface [5]. For combinatorial studies, silicon substrates with nanoscale ridge patterns can be used [26].
- Bacterial Inoculation: Inoculate a petri dish containing the treated coverslips with Pantoea sp. YR343 (or desired bacterial strain) suspended in a liquid growth medium [5].
- Incubation: Allow biofilm formation to proceed for desired time intervals (e.g., 30 minutes for initial attachment studies, 6-8 hours for cluster formation) under appropriate conditions [5].
2. AFM Sample Mounting and Preparation
- Harvesting: At selected time points, carefully remove a coverslip from the Petri dish.
- Rinsing: Gently rinse the coverslip with a buffer solution (e.g., deionized water or PBS) to remove non-adherent planktonic cells [5].
- Drying: Air-dry the sample before imaging. Note that this step may alter native hydrated structures, but is used for this specific protocol [5].
3. Large-Area Automated AFM Imaging
- Probe Selection: Choose a sharp, high-resolution probe appropriate for TappingMode or PeakForce Tapping.
- System Setup: Configure the automated large-area AFM system. Define the millimeter-sized area of interest.
- Automated Scanning: Initiate the automated routine. The system will capture multiple contiguous high-resolution images (tiles) across the defined area with minimal overlap [5].
- Image Stitching: Use the integrated machine learning algorithm to automatically align and stitch the individual image tiles into a single, seamless, high-resolution mosaic [5].
4. Data Analysis via Machine Learning
- Segmentation and Classification: Apply ML-based image analysis tools to the stitched large-area image.
- Quantitative Extraction: Automatically extract quantitative parameters such as:
Workflow and Problem-Solving Diagrams
Large-Area AFM Biofilm Analysis Workflow
Common AFM Problems and Solutions
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Materials and Reagents for Large-Area AFM Biofilm Studies
| Item | Function/Application in Research |
|---|---|
| PFOTS (Perfluorooctyltrichlorosilane) | Used to create a hydrophobic, self-assembled monolayer on glass substrates for studying biofilm assembly on modified surfaces [5]. |
| Pantoea sp. YR343 | A gram-negative, rod-shaped, motile bacterium with peritrichous flagella; used as a model organism for studying early biofilm formation and cellular patterning [5]. |
| High-Resolution AFM Probes | Sharp probes are critical for resolving nanoscale features like flagella (~20-50 nm in height) and individual cell structures [5] [2]. |
| Engineered Silicon Substrates | Surfaces with nanoscale ridges used to probe how physical topography influences bacterial adhesion and disrupts normal biofilm formation [26]. |
| Machine Learning Algorithms | Integrated software for autonomous image stitching, cell detection, and classification, enabling analysis of tens of thousands of cells from large-area scans [5] [26]. |
Solving Common Problems in Hydrated Biofilm AFM Imaging
Atomic Force Microscopy (AFM) offers unparalleled capability for investigating the nanoscale topography and mechanical properties of hydrated biofilm structures in near-physiological conditions [28] [1]. However, a significant challenge in these studies is the effective immobilization of soft, hydrated biological samples without altering their native physiological state or nanomechanical properties. Biofilms are particularly susceptible to disruption by the scanning AFM cantilever due to weak attachment forces and potential motility of constituent cells [1]. This technical guide examines the core methodologies for sample immobilization, comparing mechanical entrapment with chemical fixation approaches to help researchers select optimal strategies for their specific biofilm research applications.
Comparative Analysis: Mechanical Entrapment vs. Chemical Fixation
The table below summarizes the core characteristics, advantages, and limitations of the two primary immobilization approaches.
Table 1: Comparison of AFM Immobilization Techniques for Biofilm Research
| Feature | Mechanical Entrapment | Chemical Fixation |
|---|---|---|
| Basic Principle | Physical confinement of cells within porous media or microstructures [1]. | Chemical bonding of cells to substrate using adhesives or cross-linkers [1]. |
| Common Methods | Porous membranes (e.g., polycarbonate), agarose gels, PDMS microstamps [1]. | Poly-L-lysine, glutaraldehyde, silane-based adhesives, mica functionalization [1]. |
| Key Advantage | Generally considered more benign, minimizing physiochemical changes to cells [1]. | Provides strong, reliable adhesion capable of withstanding lateral scanning forces [1]. |
| Main Disadvantage | Immobilization can be sporadic and unpredictable, reducing reproducibility [1]. | Certain cross-linking agents can negatively impact nanomechanical properties and cell viability [1]. |
| Impact on Viability | Higher potential for maintaining cell viability [1]. | Risk of reduced viability depending on the chemical agent used [1]. |
| Best Use Cases | Imaging living cells where preserving native physiological state is critical [1]. | Applications requiring maximum immobilization strength, potentially with fixed cells [1]. |
Experimental Protocols for AFM Immobilization
Protocol for PDMS Microstamp Immobilization (Mechanical Entrapment)
This protocol describes a advanced mechanical method for immobilizing spherical microorganisms [1].
- Master Creation: Create a silicon master using deep reactive ion etching. The dimensions can be varied, with typical reports of features 1.5–6 µm wide, a pitch of 0.5 µm, and a depth of 1–4 µm to accommodate target cell sizes [1].
- PDMS Stamping: Cast a Polydimethylsiloxane (PDMS) stamp from the silicon wafer master [1].
- Cell Deposition: Deposit the bacterial cell suspension onto the PDMS stamp using convective and capillary forces to drive cells into the micro-wells [1].
- AFM Imaging: Transfer the stamp with immobilized cells to the AFM liquid cell for imaging in buffer solution under physiological conditions [1].
Protocol for Non-Perturbative Immobilization on ITO Substrates
A specialized protocol for imaging living bacteria in liquid without aggressive external immobilization [28].
- Substrate Selection: Use a glass substrate coated with a layer of Indium-Tin-Oxide (ITO). ITO's hydrophobic and smooth properties facilitate better bacterial cell adhesion [28].
- Sample Preparation: Pipette a volume (e.g., 500 µL) of bacterial culture in its exponential growth phase directly onto the ITO substrate housed in the AFM's electrochemical liquid cell [28].
- AFM Imaging: Perform AFM imaging in the culture medium using a high-speed approach/retract mode, such as Quantitative Imaging (QI) mode. This allows for stable imaging without chemical or mechanical entrapment [28].
Protocol for Chemical Fixation with Poly-L-Lysine
A common chemical method for securing cells to a substrate [1].
- Substrate Coating: Apply a solution of Poly-L-lysine to a clean glass or mica substrate for a set duration.
- Rinsing: Rinse the substrate to remove any excess, unbound Poly-L-lysine.
- Cell Deposition: Apply the bacterial suspension to the coated substrate, allowing cells to adhere via electrostatic interactions.
- Fixation (Optional): For more permanent immobilization, a mild chemical fixative like glutaraldehyde may be used, though this will compromise cell viability [1].
Troubleshooting Common Immobilization and Imaging Problems
FAQ 1: My bacterial cells are being displaced or swept away by the AFM tip during scanning. What can I do?
- Problem: Inadequate immobilization strength.
- Solutions:
- Chemical Route: Switch to or optimize a chemical fixation protocol. Ensure your substrate is properly functionalized (e.g., with APTES or NiCl₂ for enhanced electrostatic trapping) to increase adhesion strength [1] [29].
- Mechanical Route: If using mechanical entrapment, verify that the pore or well size of your membrane or PDMS stamp is appropriately matched to your cell size for a snug fit [1].
- Imaging Parameters: Softer tapping modes with lower setpoint forces can reduce lateral forces on poorly immobilized samples.
FAQ 2: After chemical fixation, my AFM force curves show a dramatic change in the mechanical properties of the cells. Is this expected?
- Problem: Chemical agents altering native nanomechanical properties.
- Solutions:
- This is a known drawback of many cross-linking fixatives like glutaraldehyde, which can harden cells [1].
- For nanomechanical studies, prioritize benign mechanical entrapment methods or use milder, non-crosslinking adhesives like Poly-L-lysine, acknowledging they may provide weaker adhesion.
- Explore the non-immobilization protocol using ITO substrates if your cells adhere sufficiently [28].
FAQ 3: I see repetitive streaks or blurred features in my AFM images of a biofilm. Is this an immobilization issue?
- Problem: This could be caused by poor immobilization, but also by other common AFM artifacts.
- Solutions:
- Check Immobilization: Ensure cells are firmly attached. Loose particles or cells can be moved by the tip, causing streaks [2].
- Tip Contamination: A contaminated or broken tip can cause repeating, irregular artifacts. Replace the AFM probe with a new, clean one [2].
- Laser Interference: If using a reflective sample and an uncoated cantilever, laser interference can cause fringes. Use a probe with a reflective coating to mitigate this [2].
- Environmental Noise: Vibrations from the environment can cause streaks. Ensure the anti-vibration table is functional and image during quieter periods if possible [2].
The following workflow diagram illustrates the decision-making process for selecting an appropriate immobilization method based on your experimental goals.
Research Reagent Solutions: Essential Materials
Table 2: Key Materials and Reagents for AFM Immobilization Protocols
| Item | Function/Benefit | Application Context |
|---|---|---|
| Indium-Tin-Oxide (ITO) Coated Substrate | Hydrophobic, smooth surface that facilitates bacterial adhesion without chemical treatment [28]. | Non-perturbative imaging of living bacteria in liquid [28]. |
| Polydimethylsiloxane (PDMS) Microstamps | Provides micro-wells for physical entrapment of cells, preserving viability and mechanics [1]. | Mechanical entrapment for high-resolution imaging of live cells [1]. |
| Poly-L-Lysine | Positively charged polymer that promotes cell adhesion to negatively charged surfaces like glass [1]. | Chemical fixation for moderate-strength immobilization. |
| Silicon Masters (for PDMS) | Used to create micro-structured stamps with defined well sizes for cell entrapment [1]. | Fabrication of custom PDMS microstamps [1]. |
| Size-Exclusion Chromatography (SEC) Media | For isolation and purification of biological nanoparticles like extracellular vesicles from biofluids [29]. | Sample preparation for analyzing biofilm-derived vesicles [29]. |
| Standard AFM Probes (PPP-CONTPt) | Conductive, cantilevers with a defined spring constant (e.g., 0.3 N/m) for imaging and force spectroscopy [28]. | General AFM imaging in liquid and mechanical mapping [28]. |
Frequently Asked Questions (FAQs)
Q1: Why is it so important to control hydration during AFM imaging of biofilms? Maintaining proper hydration is crucial because biofilms are biological systems whose complex architecture and mechanical properties are dependent on their aqueous environment. The extracellular polymeric substance (EPS) matrix, which provides structural integrity, can collapse upon dehydration, fundamentally altering the biofilm's topography and nanomechanical properties. Imaging under physiological liquid conditions or controlled humidity preserves the native state of the biofilm, allowing for accurate data on its true structure and function [19] [30].
Q2: What are the main techniques for maintaining hydration during AFM experiments? The two primary techniques are (1) Liquid Imaging and (2) Humidity Control. Liquid imaging involves submerging the tip and sample in a liquid cell containing an appropriate buffer, preserving the fully hydrated state. Humidity control involves placing the sample in a sealed chamber where the relative humidity is maintained at a high level (e.g., ~90%), preventing the moist biofilm from drying out while allowing for imaging in air [19].
Q3: I see distorted images and get inconsistent force measurements when probing my biofilm. Could hydration be the issue? Yes, inconsistent hydration is a common source of such artifacts. Drying of the biofilm sample causes shrinkage and hardening, which dramatically increases its measured stiffness and alters its topography. To troubleshoot, ensure your liquid cell is properly sealed and free of bubbles, or that your humidity chamber is correctly calibrated and stabilized before measurement. Always perform experiments as quickly as possible after sample extraction from its growth medium to minimize unintended dehydration [19] [31].
Q4: How can I verify that my hydration control methods are effective? A key indicator is the reproducibility of your nanomechanical measurements. If repeated force curves on the same biofilm sample yield consistent values for properties like adhesion force and elastic modulus, it suggests stable sample conditions. Furthermore, comparing your results with known values from literature for hydrated biofilms can serve as a benchmark. Advanced techniques like 3D AFM, which can map hydration layers, provide a direct verification method but require specialized equipment and analysis [32] [33].
Troubleshooting Guide
Table 1: Common Hydation-Related Issues and Solutions
| Problem | Potential Cause | Recommended Solution |
|---|---|---|
| Drift in Z-axis measurements | Evaporation of liquid in the cell, causing thermal drift or changing concentration. | Ensure the liquid cell is securely sealed. Use a closed-loop scanner if available. Allow the system to thermally equilibrate after adding liquid. |
| Unusually high stiffness (Young's modulus) measurements | Partial dehydration of the biofilm, making it more rigid. | Switch to a liquid environment or use a high-humidity chamber. Verify that the biofilm appears moist and has not shrunk under an optical microscope. |
| Poor image resolution or noisy data in liquid | Contamination or air bubbles on the tip or sample surface. | Use filtered buffers. Carefully clean the fluid cell and ensure proper degassing of liquids. Perform several approach-retract cycles to clean the tip in situ. |
| Inconsistent biofilm cohesion data | Variations in water content between different samples or measurement locations. | Standardize sample preparation and equilibration time in the humidity chamber. Use a humidity sensor to monitor and actively control the chamber environment [19]. |
| Tip convolution artifacts, overestimation of feature widths | The finite size of the AFM tip interacting with soft, hydrated features. | Use sharper, high-resolution tips. Apply tip-deconvolution algorithms during data processing. For nanofibers, account for the similar scale of the tip and sample [31]. |
Experimental Protocols for Hydrated Biofilm Analysis
Protocol 1: Measuring Biofilm Cohesiveness under Controlled Humidity
This protocol is adapted from a method designed to reproducibly measure the cohesive energy of moist biofilms [19].
Key Research Reagent Solutions:
- Saturated Salt Solution: Used to create a stable ~90% relative humidity environment within a sealed chamber, preventing sample dehydration during measurement.
- Biofilm Growth Medium: The specific nutrient medium required to cultivate the biofilm of interest, ensuring relevant physiological properties.
- PBS or Mild Buffer: For gently rinsing unattached cells without disrupting the fragile biofilm structure.
Methodology:
- Biofilm Growth: Grow your biofilm on a suitable substrate (e.g., a glass coupon or membrane) using a reactor system like a membrane-aerated biofilm reactor or a rotating disc reactor.
- Sample Equilibration: Gently rinse the biofilm sample to remove loosely attached cells. Place the sample in a sealed chamber containing a saturated NaCl solution (or another salt to achieve the desired relative humidity) for approximately one hour to allow the biofilm to equilibrate to a consistent water content.
- AFM Setup: Mount the equilibrated sample onto the AFM stage. Use a scanner and a closed-loop humidity chamber that can maintain the pre-set humidity level (e.g., 90%) throughout the experiment.
- Topographical Imaging: First, image a selected region of the biofilm (e.g., 5x5 µm) at a very low applied force ("non-perturbative" imaging) to obtain a reference topography.
- Abrasive Scanning and Cohesion Measurement: Zoom into a smaller sub-region (e.g., 2.5x2.5 µm). Abrade the biofilm by performing repeated raster scans with an elevated normal load (e.g., 40 nN). After a set number of scans, return to the low force and re-image the larger area. The volume of biofilm removed is calculated by subtracting the topographic images before and after abrasion. The cohesive energy is determined from this displaced volume and the corresponding frictional energy dissipated during abrasion [19] [34].
The following diagram illustrates the core workflow of this cohesiveness measurement protocol:
Protocol 2: High-Resolution Imaging in Liquid
This protocol outlines the steps for imaging biofilm structures in their fully hydrated state using a liquid cell [5] [32].
Key Research Reagent Solutions:
- Physiological Buffer (e.g., PBS or Growth Medium): Maintains the ionic strength and pH of the biofilm's native environment, crucial for preserving its structure and activity.
- Liquid Imaging Cell: A specialized, sealed chamber that holds the buffer and allows the AFM tip and laser to interact with the submerged sample.
Methodology:
- Sample Loading: Secure the biofilm-grown substrate firmly into the liquid cell of the AFM.
- Liquid Injection: Carefully inject an appropriate physiological buffer into the cell, ensuring all air bubbles are purged. The buffer should completely cover the tip and sample.
- System Equilibration: Allow the system to sit for 10-20 minutes after fluid injection to minimize thermal drift caused by temperature differences.
- Tip Engagement: Engage the AFM tip with the sample surface slowly and carefully within the liquid environment. The engagement parameters in liquid may differ from those in air.
- Imaging and Force Spectroscopy: Perform topographical scanning or collect force-volume maps using a suitable mode (e.g., tapping mode in fluid). Using advanced techniques like 3D-AFM allows for the mapping of solvation forces and hydration layers at the biofilm-solid interface with sub-nanometer resolution [32] [33].
Quantitative Data on Biofilm Properties
Table 2: Measured Properties of Biofilms under Different Hydration Conditions
| Biofilm Type / Source | Measurement Condition | Measured Property | Value | Citation |
|---|---|---|---|---|
| Activated Sludge (Mixed Culture) | Moist, ~90% Humidity | Cohesive Energy (surface layer) | 0.10 ± 0.07 nJ/µm³ | [19] |
| Activated Sludge (Mixed Culture) | Moist, ~90% Humidity | Cohesive Energy (deeper layer) | 2.05 ± 0.62 nJ/µm³ | [19] |
| Activated Sludge (+10mM Ca²⁺) | Moist, ~90% Humidity | Cohesive Energy (increased) | 1.98 ± 0.34 nJ/µm³ | [19] |
| Young Drinking Water Biofilm | Aqueous / Hydrated | Mechanical Removal Shear Stress | ~100 kPa | [34] |
| Pantoea sp. YR343 | Dried for Imaging | Cell Dimensions (Length x Diameter) | ~2 µm x ~1 µm | [5] |
Leveraging Machine Learning for Automated Image Stitching and Analysis
Frequently Asked Questions (FAQs)
Q1: What are the most common causes of failure in automated image analysis pipelines? Automated image analysis failures often stem from inconsistent measurements due to subjective manual segmentation, discrepancies across instruments, and variations in user input. Other major causes include invalid or corrupted image data (e.g., incorrect format, encoding, or excessive file size), insufficient user permissions or authentication errors, and network timeouts or service unavailability [35] [36].
Q2: How can I ensure my stitched AFM images are reproducible? To ensure reproducibility, minimize variability by adopting consistent calibration practices, meticulously document all protocols, and reduce subjective steps in image analysis. Use software tools that allow you to save settings, standardize workflows, and employ AI-powered segmentation to decrease user-to-user discrepancies. Implementing lockable analysis protocols is also recommended [35].
Q3: My image stitching algorithm fails to find overlapping features. What should I check? This failure often occurs with images that have varied features or complex transformations. The approach involves leveraging robust feature detection and matching algorithms like SIFT. You can then use a classifier, such as a Support Vector Machine (SVM), to accurately recognize which image pairs should be stitched together [37].
Q4: What are the hardware limitations of conventional AFM for large-area biofilm imaging, and how does your method overcome them? Conventional AFM has a limited imaging area (typically less than 100 µm), restricted by piezoelectric actuator constraints. This makes it difficult to study large, millimeter-scale biofilm structures and capture their full spatial complexity. Our automated large-area AFM method overcomes this by automating the scanning process over extended areas, using machine learning for seamless image stitching to create high-resolution, millimeter-scale maps [5].
Q5: I keep getting "API version deprecated" errors. How can I avoid service disruptions? Service providers periodically retire older API versions. To avoid disruptions, proactively monitor official release notes and deprecation schedules. It is strongly recommended to migrate to the latest generally available (GA) API versions instead of relying on preview versions, which may be disabled without consistent behavior. Also, subscribe to service health alerts for timely notifications [38].
Troubleshooting Guides
Guide 1: Addressing Image Analysis and Stitching Failures
| Problem | Cause | Solution |
|---|---|---|
| Failed Image Stitching | Lack of recognizable overlapping features between images [37]. | Use robust feature detection algorithms (e.g., SIFT) and ensure sufficient image overlap during acquisition. |
| Inconsistent Measurements | Subjective manual segmentation and user-to-user variability [35]. | Implement AI-powered segmentation tools and lockable, standardized analysis protocols. |
| "Invalid Image Data" Error | Malformed request, bad image data, or file size too large [36]. | Validate image format and size before processing; resize large images pre-upload. |
| "Permission Denied" Error | Incorrect credentials or lack of API access [36]. | Verify authentication keys and ensure the required API service is enabled in your project. |
| "API Version Deprecated" | Use of an outdated or retired API version [38]. | Switch to the latest stable API version and monitor official deprecation schedules. |
Guide 2: Resolving Common AFM Imaging Issues for Biofilms
| Problem | Cause | Solution |
|---|---|---|
| Limited Field of View | Conventional AFM's small imaging area (<100 µm) restricts visualization of macro-scale biofilm organization [5]. | Employ an automated large-area AFM approach that stitches multiple high-resolution scans. |
| Low Throughput & Labor Intensity | Slow, manual AFM operation prevents imaging of dynamic processes over large areas [5]. | Integrate machine learning to automate scanning, site selection, and probe conditioning [5]. |
| Data Overload | Large-area AFM generates high-volume, information-rich data that is difficult to process manually [5]. | Implement ML-based image segmentation and analysis for automated feature extraction (e.g., cell count, shape). |
| Sample Representativeness | Small scan areas may not capture the spatial heterogeneity inherent to biofilms [5]. | Use large-area scanning and ML-driven analysis to quantify parameters across millimeter-scale areas. |
Experimental Protocols
Protocol 1: Automated Large-Area AFM for Biofilm Analysis
This protocol details the methodology for imaging the early stages of Pantoea sp. YR343 biofilm formation using a large-area, automated AFM system aided by machine learning [5].
1. Sample Preparation
- Surface Treatment: Use glass coverslips treated with PFOTS to create a hydrophobic surface.
- Inoculation: Inoculate a petri dish containing the treated coverslips with Pantoea cells in a liquid growth medium.
- Incubation: Incubate for selected time points (e.g., ~30 minutes for initial attachment; 6-8 hours for cluster formation).
- Rinsing and Drying: At each time point, remove a coverslip, gently rinse with deionized water to remove unattached cells, and air-dry before AFM imaging [5].
2. Automated Large-Area AFM Imaging
- Instrument Setup: Configure the AFM for large-area scanning. This typically involves a specialized stage or scan strategy.
- Automated Scanning: Initiate the automated scanning protocol. The system should be programmed to capture multiple contiguous high-resolution images over a millimeter-scale area.
- Image Acquisition: Ensure minimal overlap between individual scans to maximize acquisition speed while still allowing for accurate stitching [5].
3. Image Stitching and Processing
- Stitching: Use a machine learning-powered algorithm to seamlessly stitch the individual AFM scans into a single, large-area, high-resolution image.
- Distortion Correction: The algorithm should correct for any spatial distortions or displacements between individual frames [5].
4. Machine Learning-Based Analysis
- Segmentation: Apply an ML-based image segmentation model to automatically identify and outline individual bacterial cells.
- Parameter Extraction: Use the model to extract quantitative data, including:
- Cell count
- Surface confluency (% coverage)
- Cellular morphology (e.g., length, width, surface area)
- Cellular orientation
- Flagella Mapping: Utilize high-resolution capabilities to map and analyze flagella interactions and distribution [5].
Protocol 2: SVM-Based Panorama Recognition for Image Stitching
This protocol describes a methodology for recognizing which images belong to the same panorama, a foundational step for fully automated stitching, using a Support Vector Machine (SVM) classifier [37].
1. Dataset Creation
- Synthetic Data: Create a synthetic dataset of image pairs. Some pairs should have overlapping regions (positive class), while others should not (negative class).
2. Feature Extraction
- Keypoint Detection: For each image in a pair, use the SIFT (Scale-Invariant Feature Transform) algorithm to detect keypoints.
- Descriptor Generation: For each keypoint, SIFT generates a descriptor (a feature vector).
- Feature Matching: Match the descriptors between the two images using a matching algorithm (e.g., k-nearest neighbors).
- Input Vector Formation: Use the results of the feature matching (e.g., the number of good matches, quality metrics) to form an input feature vector for the SVM classifier [37].
3. Model Training and Evaluation
- Hyperparameter Tuning: Systematically tune the SVM's hyperparameters (e.g., kernel type, regularization parameter) to optimize performance.
- Training: Train the SVM model on the labeled dataset of image pairs.
- Validation: Evaluate the model's performance using metrics such as precision, recall, and F1-score to ensure it can accurately recognize panoramas [37].
Research Reagent Solutions
The following materials are essential for conducting automated AFM imaging and analysis of hydrated biofilms.
| Item | Function |
|---|---|
| PFOTS-treated Substrate | Creates a uniform, hydrophobic surface to study bacterial attachment dynamics and early biofilm formation [5]. |
| Pantoea sp. YR343 | A gram-negative, rod-shaped model bacterium with peritrichous flagella, used to study biofilm assembly and structure [5]. |
| Automated Large-Area AFM | Enables high-resolution topographical and nanomechanical mapping over millimeter-scale areas, overcoming the limited field of view of conventional AFM [5]. |
| SIFT Algorithm | A robust feature detection and matching algorithm used to identify keypoints in overlapping images, which is crucial for the panorama recognition and stitching process [37]. |
| Support Vector Machine (SVM) | A machine learning classifier used to accurately determine whether image pairs contain sufficient overlapping features to be stitched into a panorama [37]. |
| AI Segmentation Models | Pre-trained or trainable deep learning models that automatically identify and outline biological structures (e.g., cells, flagella) in complex AFM images, enabling high-throughput quantitative analysis [35] [5]. |
Workflow and Signaling Diagrams
Automated AFM Workflow for Biofilm Analysis
SVM for Panorama Recognition
Best Practices for Cantilever Selection and Force Calibration
FAQs: Cantilever Selection and Calibration for Biofilm Research
1. Why is cantilever calibration so critical for quantitative AFM force measurements on biofilms? Accurate force calibration is the foundation for converting the AFM's raw photodetector signal (volts) into a quantitative force (newtons). Uncalibrated or poorly calibrated cantilevers can lead to force errors of 100% or more [39]. For biofilm research, this is particularly important when measuring nanomechanical properties like stiffness and adhesion or interaction forces between functionalized tips and biofilm components, as the results must be reliable and reproducible [40] [1].
2. What type of cantilever should I use for imaging hydrated biofilm structures? For topographical imaging of soft, hydrated biofilms, soft cantilevers with spring constants in the range of 0.01 N/m to 0.1 N/m are generally recommended to minimize sample damage [1]. Tapping mode (or intermittent contact mode) is the preferred imaging technique as it reduces lateral (drag) forces on the delicate biofilm structure compared to contact mode [1].
3. What type of cantilever is best for force spectroscopy measurements on biofilms? The choice depends on the expected interaction forces. For measuring weak adhesion forces or the mechanical properties of the extracellular polymeric substance (EPS), a soft cantilever (~0.01 N/m to 0.1 N/m) is suitable. For studies involving stiff cellular components or stronger molecular interactions, a stiffer cantilever (~0.1 N/m to 1 N/m) may be necessary to avoid excessive deformation [1]. The thermal noise method is often used to calibrate these cantilevers [39].
4. My force curves on a biofilm seem noisier than on a hard surface. Is this normal? Yes, this is expected. Biofilms are soft, viscoelastic materials that can be easily indented. The "contact portion" of a force curve on a rigid surface like glass or mica has a steep, linear slope, which is used to calibrate the detector's sensitivity. On a soft biofilm, this slope is more gradual, which can appear noisier. Always perform your sensitivity calibration on a rigid, dry part of your substrate before engaging with the biofilm [39].
Troubleshooting Guides
Issue: Inconsistent Spring Constant Values Between Calibration Methods
| Observation | Possible Cause | Solution |
|---|---|---|
| Theoretical calculation disagrees with thermal or experimental method. | Nominal dimensions from manufacturer differ from actual cantilever geometry; uncertainty in material properties (Young's modulus). | Avoid relying solely on theoretical values. Use an experimental method like the thermal noise method or calibrate against a standard reference material (SRM) [39] [41]. |
| Thermal method gives a different value than the added-mass method. | Inaccurate measurement of the added mass or its position on the cantilever in the added-mass method. | The thermal method is generally considered more reliable and less destructive. Use the thermal method as your primary calibration technique [39]. |
| Lateral force calibration seems inaccurate. | The common "wedge method" can suffer from load-dependent errors and requires careful analysis [42]. | Use a more direct method, such as a glass fiber standard [40] or a calibrated microforce sensor (MEMS) [42]. |
Issue: Damage to or Destabilization of Hydrated Biofilm During Imaging or Force Measurement
| Observation | Possible Cause | Solution |
|---|---|---|
| Biofilm structure is swept away or deformed during scanning. | Excessive imaging force; use of contact mode on a soft, weakly adhered sample. | Switch to Tapping Mode. Use softer cantilevers (spring constant < 0.1 N/m) and reduce the imaging setpoint to minimize tip-sample force [1]. |
| Cells detach from the substrate during force mapping. | Inadequate immobilization of the sample. | Chemically functionalize your substrate (e.g., with poly-L-lysine or using a photocatalytically active surface) to improve cell adhesion. Alternatively, use a porous membrane to physically trap cells [1]. |
| Unstable laser deflection signal in liquid. | Drift in the laser alignment on the cantilever; air bubbles in the liquid cell. | Ensure the system is thermally equilibrated. Carefully clean and fill the liquid cell to avoid bubbles. Use a cantilever with a reflective coating optimized for liquid environments. |
Quantitative Data and Calibration Methods
Comparison of Common Calibration Methods
The following table summarizes key techniques for calibrating the normal spring constant of AFM cantilevers.
| Method | Principle | Key Advantages | Key Limitations | Typical Uncertainty |
|---|---|---|---|---|
| Theoretical [40] | Calculates spring constant from cantilever dimensions & material properties. | Quick; no additional equipment needed. | Requires accurate knowledge of dimensions and modulus; difficult for irregular shapes or coatings. | High (can be >100%) [39] |
| Thermal Noise [39] [43] | Analyzes cantilever's Brownian motion using Equipartition Theorem. | Non-destructive; fast; works for various lever shapes; built into many AFM software packages. | Less accurate for very stiff levers; requires accurate sensitivity calibration. | Medium (~5-15%) |
| Laser Doppler Vibrometry (LDV) Thermal [43] | Advanced thermal method with ultra-precise displacement measurement. | Very high accuracy and SI traceability; low uncertainty. | Requires specialized, expensive equipment (LDV). | Very Low (~1-2%) |
| Added Mass (Cleveland) [39] | Measures frequency shift from attached known masses. | Conceptually straightforward. | Destructive; time-intensive; uncertainty in added mass and position. | Medium |
| Reference Cantilever [39] [41] | Measures deflection against a lever of known spring constant. | Direct method. | Requires a set of accurately pre-calibrated reference levers. | Depends on reference |
Standard Reference Materials for Calibration
For the highest accuracy, traceable standards are available.
| Material / Standard | Function | Key Features |
|---|---|---|
| NIST SRM 3461 [41] | Array of 7 pre-calibrated silicon cantilevers for validating or performing calibration methods. | SI-traceable calibration; spring constants from 0.5 N/m to 100 N/m. |
| Glass Fiber Standard [40] | A known structure for direct lateral force calibration. | Inexpensive, easy-to-make; direct conversion of signal to force; transferable. |
| MEMS Microforce Sensor [42] | External sensor for direct lateral force calibration. | Directly measures friction force; high precision and accuracy; eliminates need for grating. |
Experimental Protocols
Detailed Protocol: Thermal Noise Method for Normal Spring Constant Calibration
This is a step-by-step guide for one of the most common and practical calibration methods [39].
1. Equipment and Setup:
- Atomic Force Microscope.
- Rigid calibration substrate (e.g., clean silicon wafer, freshly cleaved mica).
- Software capable of acquiring a thermal tune spectrum (common in modern AFMs) or a high-speed analog-to-digital converter (ADC) card to record the deflection signal.
2. Measurement Procedure:
- Mount the Substrate and Cantilever: Place the rigid substrate and the cantilever to be calibrated in the AFM.
- Laser Alignment: Align the laser on the end of the cantilever to get a strong, stable signal with a compact spot size.
- Approach: Perform a coarse approach and auto-approach until the tip is in contact with the rigid substrate.
- Sensitivity Calibration:
- Obtain a force curve on the rigid substrate.
- In the contact portion of the force curve, the deflection signal (in Volts) changes linearly with the piezo displacement (in meters).
- Measure the slope of this linear region (dV/dz). The inverse of this slope is the sensitivity factor, ( S ) (in m/V), which relates the photodiode signal to the actual cantilever deflection: ( \text{Deflection} = S \times \text{Voltage} ).
- Thermal Spectrum Acquisition:
- Withdraw the tip several microns from the surface, away from any influence of surface forces.
- Record the thermal fluctuations of the cantilever's deflection signal at the highest possible sensitivity for several seconds to obtain a good statistical average.
3. Data Analysis:
- Calculate the Power Spectral Density (PSD) of the recorded thermal noise data.
- Identify the fundamental resonant peak in the PSD plot.
- Fit the peak to a simple harmonic oscillator model to determine the area under the peak, ( P ) (in V²/Hz). This represents the mean squared amplitude of the oscillation.
- Apply the thermal calibration formula to calculate the spring constant, ( k ): ( k = \frac{0.8174 kB T}{S^2 P} ) where ( kB ) is Boltzmann's constant and ( T ) is the absolute temperature in Kelvin [39].
Workflow Diagram: Force Calibration and Application
The following diagram illustrates the logical workflow for cantilever calibration and its application in biofilm force measurement experiments.
The Scientist's Toolkit: Essential Research Reagents and Materials
| Item | Function in AFM Biofilm Research |
|---|---|
| Soft Cantilevers (0.01 - 0.1 N/m) | Essential for high-resolution imaging of delicate, hydrated biofilms in tapping mode to prevent structural damage [1]. |
| Poly-L-Lysine | A common chemical immobilization agent. Coating a substrate (e.g., glass) with poly-L-lysine promotes electrostatic adhesion of microbial cells, preventing them from being swept away by the AFM tip [1]. |
| NIST SRM 3461 | A Standard Reference Material consisting of an array of pre-calibrated cantilevers. Used to validate in-house calibration methods or to perform the reference lever method, ensuring SI-traceable accuracy [41]. |
| Silicon Wafer / Mica | An atomically flat, rigid substrate. Critical for accurately performing the sensitivity calibration step required for most spring constant calibration methods [39]. |
| MEMS Microforce Sensor | A microelectromechanical system device that acts as a force transducer. Provides a direct and accurate method for calibrating lateral (friction) forces, bypassing the uncertainties of the wedge method [42]. |
| Glass Fiber Standard | A simple, inexpensive, and transferable artifact for direct calibration of the AFM's lateral force sensitivity [40]. |
Validating AFM Data and Comparing it with Other Biofilm Imaging Modalities
Troubleshooting Guides
Common Integration Challenges and Solutions
Table 1: Troubleshooting AFM-CLSM Integration
| Problem | Possible Cause | Solution | Preventive Measure |
|---|---|---|---|
| Poor image registration/overlay | Different resolution and field of view between techniques [5] | Use standardized calibration grids; apply software-based image stitching and registration algorithms [5] | Establish a correlative workflow with common fiduciary markers |
| AFM probe interference with CLSM detection | Probe shadow or reflection obstructs optical path [44] | Use reflective or specially coated probes; adjust CLSM laser angle relative to AFM probe position | Characterize probe-optics interaction during setup |
| Sample deformation or drift during sequential imaging | Sample hydration changes or mechanical instability [5] [4] | Use liquid cell for hydrated imaging; minimize time between AFM and CLSM measurements | Implement rapid correlative systems or environmental control chambers |
| Inconsistent data from different length scales | Mismatch in scanning areas (AFM: μm-scale; CLSM: mm-scale) [5] [26] | Adopt large-area automated AFM to bridge the scale gap [5] [26] | Pre-define regions of interest (ROIs) for both techniques |
Table 2: Troubleshooting AFM-SEM Integration
| Problem | Possible Cause | Solution | Preventive Measure |
|---|---|---|---|
| AFM electronics interference with SEM beam | Electromagnetic noise from AFM controllers [45] | Use shielding; optimize grounding schemes; employ sequential rather than simultaneous imaging | Invest in dedicated integrated AFM-SEM systems [45] |
| Vacuum incompatibility for bio-samples | Dehydration in SEM vacuum alters native biofilm structure [30] | For hydrated biofilms, use environmental SEM (ESEM) or dedicated hydration cells [45] | Plan experiments: use SEM for high-res surface data after AFM mechanical mapping |
| Limited space for AFM inside SEM chamber | Physical size of AFM scanner and sample stage [45] | Use miniaturized AFM detectors designed for in-situ SEM integration [45] | Verify system compatibility and chamber size before integration attempts |
| Sample contamination during transfer | Exposure to atmosphere or handling between instruments [45] | Use integrated systems or anoxic transfer chambers to maintain sample condition | Implement glove boxes or clean room environments for sample handling |
FAQ: Addressing Researcher Queries
Q1: What is the most critical step for successfully correlating AFM with CLSM for hydrated biofilm studies?
A: The most critical step is maintaining biofilm hydration and physiological conditions throughout the correlative process. This requires using a liquid cell for both AFM and CLSM imaging. For AFM, this allows nanomechanical property mapping under native conditions [5] [4]. For CLSM, it preserves cell viability and the structure of the extracellular polymeric substance (EPS). The workflow must be optimized to minimize transition time between instruments.
Q2: Can we perform true simultaneous AFM and CLSM imaging?
A: Yes, true simultaneous imaging is possible and is a powerful approach for observing dynamic processes. It requires a specially designed integrated setup where the AFM is mounted onto the CLSM stage. This allows for directly correlating nanomechanical data from AFM (e.g., stiffness, adhesion) with functional fluorescence data from CLSM (e.g., metabolic activity, specific labels) from the exact same location and time [44]. The main challenge is avoiding optical interference from the AFM cantilever.
Q3: How can we overcome the significant scale difference between conventional AFM scans and CLSM images?
A: The key is to use automated large-area AFM. Traditional AFM is limited to scans of ~100x100 μm, while biofilms organize over millimeter scales. Automated large-area AFM platforms overcome this by stitching hundreds of high-resolution AFM images to create a millimeter-scale map, seamlessly bridging the resolution and scale gap with CLSM [5] [26]. Machine learning algorithms are then used to analyze these large datasets and extract quantitative features like cell count, orientation, and surface coverage [5].
Q4: What are the advantages of integrating AFM with SEM instead of using them separately?
A: In-situ AFM/SEM integration provides complementary, multiparametric data from the same sample location. SEM offers high-resolution surface topography and composition, while AFM adds quantitative nanomechanical properties (e.g., elasticity, adhesion) and can operate in liquid [45] [4]. An integrated system eliminates the uncertainty of relocating specific features after transfer, providing a direct link between the sample's ultrastructure (SEM) and its mechanical function (AFM) [45].
Experimental Protocols
Protocol: Correlative AFM-CLSM for Hydrated Biofilm Mechanics and Structure
Objective: To correlate the nanomechanical properties of a hydrated biofilm measured by AFM with its 3D structure and chemical composition obtained by CLSM.
Materials:
- Bacterial strain (e.g., Pantoea sp. YR343 [5])
- Growth medium
- Suitable substrate (e.g., glass coverslip, PFOTS-treated coverslip [5])
- AFM with liquid cell and optical access
- CLSM
- Fluorescent stains (e.g., for live/dead cells, EPS components)
Method:
- Sample Preparation: Grow biofilms on the substrate to the desired maturity. For staining, incubate with appropriate fluorescent dyes according to the manufacturer's protocol.
- Region of Interest (ROI) Selection: Place the sample in the AFM/CLSM integrated setup. Use low-magnification CLSM to identify several ROIs based on biofilm architecture.
- Fiduciary Marking: Use a focused laser or micro-ablation to create small, gentle fiduciary marks near the ROI to enable re-location if needed. Alternatively, use unique topological features as natural markers.
- Simultaneous/Sequential Data Acquisition:
- Simultaneous: Engage the AFM probe in the liquid on the ROI. Start the CLSM Z-stack acquisition and simultaneously run the AFM force mapping or imaging sequence.
- Sequential: First, acquire the CLSM Z-stack. Then, without moving the sample, perform AFM nanomechanical mapping on the exact same region.
- Data Correlation: Use software to overlay the AFM mechanical maps (e.g., Young's modulus) with the CLSM fluorescence channels. Correlate local stiffness variations with high EPS density or specific cell arrangements.
Protocol: In-situ AFM-SEM for High-Resolution Surface and Mechanical Characterization
Objective: To obtain high-resolution SEM surface images and AFM nanomechanical data from the same micron-scale region of a biofilm.
Materials:
- Biofilm sample
- Integrated AFM-SEM system [45]
- Sample holder compatible with both techniques
Method:
- System Setup: Install the miniaturized AFM detector inside the SEM chamber. Ensure the AFM and SEM coordinate systems are calibrated [45].
- Sample Loading and Pump Down: Mount the biofilm sample on the stage. Evacuate the SEM chamber. For hydrated samples, use an ESEM mode or a specialized hydration stage.
- ROI Identification with SEM: Use the SEM at low magnification (e.g., 500X) to navigate the sample and locate a region of interest for correlation.
- In-situ AFM Analysis: At high SEM magnification, navigate the AFM probe to the exact ROI. Retract the SEM beam or blank it to minimize interference. Perform the AFM scan (topography or force spectroscopy) to collect mechanical data.
- SEM Imaging: Once the AFM scan is complete, retract the probe and acquire high-resolution SEM images of the same region.
- Data Correlation: Use the software provided with the integrated system to precisely overlay the AFM and SEM data channels, correlating surface features with mechanical properties.
Workflow and Relationship Visualizations
Correlative Microscopy Decision Pathway
AFM-CLSM Integration Setup
Research Reagent and Material Solutions
Table 3: Essential Materials for Correlative Biofilm Experiments
| Item | Function/Description | Example Application in Protocols |
|---|---|---|
| PFOTS-treated Glass Coverslips | Creates a hydrophobic surface to study specific biofilm attachment patterns [5]. | Used as a substrate to observe honeycomb-like cellular organization in Pantoea sp. YR343 biofilms [5]. |
| Liquid Cell (AFM) | A sealed chamber that allows the AFM probe to scan samples immersed in liquid, preserving hydration [4]. | Essential for all AFM-CLSM correlative work on live, hydrated biofilms to measure native mechanical properties. |
| Fluorescent Stains (e.g., SYTO9, PI, ConA) | Binds to specific biofilm components (nucleic acids, polysaccharides) for visualization in CLSM. | Used in the AFM-CLSM protocol to distinguish live/dead cells or visualize EPS matrix alongside AFM mechanics. |
| Calibration Grid | A standard sample with a known pattern (e.g., grating) for calibrating and aligning both microscopes. | Critical first step in both protocols to ensure precise spatial correlation between AFM and CLSM/SEM images. |
| Engineered Nanostructured Surfaces | Substrates with nanoscale ridges or patterns to study how surface topography influences biofilm growth [26]. | Used to test antifouling surfaces; AFM-SEM correlation reveals how structures disrupt bacterial attachment [26]. |
Benchmarking AFM Data Against Traditional Methods (Crystal Violet, CFU)
Atomic Force Microscopy (AFM) provides nanoscale resolution for probing biofilm topography and mechanical properties under physiological conditions. However, researchers often need to benchmark these high-resolution findings against established, bulk quantification methods like Crystal Violet (CV) staining and Colony Forming Unit (CFU) counts to contextualize their data within the existing body of literature. This technical guide addresses the common challenges and solutions in correlating data from these disparate methods, framed within the complexities of imaging hydrated biofilm structures.
Quantitative Comparison of Biofilm Assessment Methods
The table below summarizes the core characteristics, outputs, and key benchmarking metrics of AFM, Crystal Violet, and CFU methods, based on interlaboratory studies and methodological reviews.
Table 1: Key Methodological Characteristics and Quantitative Reproducibility of Biofilm Assessment Techniques
| Method | Primary Measurement | Data Output | Reproducibility (Reproducibility Standard Deviation, S_R) | Key Advantage | Key Limitation |
|---|---|---|---|---|---|
| Atomic Force Microscopy (AFM) | Topography, Nanomechanical properties | Height maps, Adhesion force, Stiffness (Modulus) | Not formally quantified in interlab studies [46] | Nanoscale resolution under hydrated conditions [5] [4] | Small scan area, sensitive to vibration, tip artifacts [2] |
| Crystal Violet (CV) | Total biomass (cells & matrix) | Absorbance (570-600 nm) | S_R = 0.44 (log10 scale) in control experiments [46] | Cost-effective, high-throughput compatibility [46] [47] | Does not distinguish live/dead cells [46] [30] |
| Colony Forming Unit (CFU) | Viable, culturable cells | Log10(CFU/well) | S_R = 0.92 (log10 scale) in control experiments [46] | Direct measure of cultivable viability [46] [47] | Labor-intensive, misses viable-but-non-culturable cells [46] [30] |
Frequently Asked Questions (FAQs) and Troubleshooting
FAQ 1: Why is there a poor correlation between AFM biomass coverage and Crystal Violet absorbance in my replicates?
Issue: AFM measures biovolume or surface coverage from a very small, nanoscale area, while CV staining measures the total biomass from the entire well. This difference in scale can lead to perceived discrepancies.
Solution:
- Increase AFM Sampling: Do not rely on a single AFM scan. Use automated large-area AFM to collect multiple images across different locations of your sample (e.g., millimeter-scale areas) to capture the inherent heterogeneity of the biofilm and get a more representative dataset [5].
- Standardize CV Protocol: Ensure consistent staining, destaining, and elution times across all replicates. Even small variations can significantly impact the final absorbance reading [46].
FAQ 2: My AFM images show abundant cells, but the corresponding CFU counts are low. What does this mean?
Issue: This is a common and informative discrepancy. AFM detects all surface-associated structures (live cells, dead cells, and extracellular polymeric substance (EPS)), while CFU counts only quantify cells that are viable and able to grow on the selected agar medium [47].
Solution and Interpretation:
- Confirm Antimicrobial Efficacy: If this result occurs after an antimicrobial treatment, it indicates the treatment was effective at killing cells but not at removing cellular debris. The AFM is visualizing the "ghosts" of the inactivated biofilm [30].
- Check for VBNC State: The biofilm may contain a population of viable but non-culturable (VBNC) cells, which are metabolically active but do not form colonies on plates. AFM can image these cells, but CFU counts will not detect them [30].
FAQ 3: I am getting persistent streaks and noise in my AFM images of hydrated biofilms. How can I fix this?
Issue: Streaks and noise are often caused by environmental interference or tip contamination, which is particularly problematic when imaging soft, dynamic samples like hydrated biofilms.
Solution:
- Check for Vibration: Ensure the AFM is on a functioning anti-vibration table. Perform imaging during quiet hours (e.g., early morning or late evening) to minimize building vibrations [2].
- Inspect the AFM Tip: Contamination or a blunt tip is a common cause of image artifacts. If you see repeating, irregular shapes, replace the probe with a new, sharp one [2].
- Verify Laser Interference: If the sample is highly reflective, laser interference can cause noise. Use a probe with a reflective coating to mitigate this issue [2].
FAQ 4: What is the best way to benchmark the efficacy of an anti-biofilm treatment using these methods?
Solution: Use a combination of methods, as they provide complementary information. The interlaboratory study found that for treatment experiments, plate counts (CFU) had the best responsiveness and reproducibility (Slope/S_R = 1.02) for evaluating killing efficacy [46].
Recommended Workflow:
- Use CV for initial, high-throughput screening of treatment impact on total biomass.
- Use CFU to quantify the log reduction in viable cells, which is the gold standard for antimicrobial efficacy testing [46].
- Use AFM to investigate the structural and mechanical changes induced by the treatment, such as collapse of the 3D architecture or changes in biofilm stiffness, which provides mechanistic insights [4].
Experimental Protocols for Integrated Analysis
Protocol 1: Correlative AFM and Crystal Violet Staining on the Same Sample
This protocol allows you to correlate nanoscale surface morphology with total biomass.
- Biofilm Growth: Grow biofilm in a sterile, optical-grade Petri dish or on a coverslip suitable for AFM according to your standard protocol [46].
- AFM Imaging (Live Biofilm): Carefully transfer the hydrated sample to the AFM. Using a soft cantilever (e.g., 0.1 N/m spring constant), acquire multiple large-area scans in non-contact or quantitative imaging mode in the same medium used for growth to maintain biofilm structure [5] [4].
- Fixation and Staining: After AFM imaging, gently rinse the sample with phosphate-buffered saline (PBS) to remove non-adherent cells. Fix the biofilm with 2.5% glutaraldehyde for 30 minutes. Then, stain with 0.1% crystal violet solution for 20 minutes [46] [47].
- Destaining and Elution: Gently rinse the stained sample with water until the water runs clear. Elute the bound dye with 30% acetic acid (200 µL) for 15 minutes with gentle shaking [46].
- Quantification: Transfer 100 µL of the eluent to a clean 96-well plate and measure the absorbance at 570-600 nm using a plate reader [46].
Protocol 2: Benchmarking Antimicrobial Efficacy Against CFU
This protocol outlines a standardized method for comparing AFM-observed structural damage with the gold standard for cell viability.
- Treatment Design: In a 96-well plate, grow biofilms and apply your antimicrobial treatment (e.g., sodium hypochlorite) across a range of concentrations. Include wells with growth medium only as negative controls [46].
- Neutralization and Homogenization: After treatment, remove the antimicrobial agent and neutralize it (e.g., with Dey-Engley broth or by washing twice with PBS). For the CFU assay, add sterile PBS to the well and disrupt the biofilm by vigorous scraping, pipetting, or brief sonication to create a homogeneous cell suspension [46] [47].
- CFU Enumeration: Perform serial dilutions of the homogenized biofilm suspension. Plate aliquots onto nutrient agar plates and incubate for 24-48 hours. Count the colonies and back-calculate to Log10(CFU/well) [46].
- AFM Sample Preparation: From a separate, identically treated well (not homogenized), carefully extract the biofilm-covered surface. Gently rinse it with PBS or a compatible buffer to remove loose debris while preserving the treated biofilm structure.
- AFM Imaging (Treated Biofilm): Image the treated, hydrated biofilm using AFM. Focus on capturing changes in the EPS matrix integrity, cell morphology, and overall biofilm architecture compared to the untreated control [4].
Essential Research Reagent Solutions
Table 2: Key Materials and Reagents for Integrated Biofilm Analysis
| Reagent / Material | Function | Considerations for Benchmarking |
|---|---|---|
| Polystyrene 96-well Plates | Substrate for high-throughput biofilm growth for CV and CFU [46] | Use flat-bottomed, untreated plates for consistent AFM sampling if used as a substrate. |
| Crystal Violet Dye | Stains total biomass (cells and EPS) [46] [47] | Ensure consistent staining and destaining times for reproducible absorbance values. |
| Soft Cantilevers (e.g., 0.1 N/m) | AFM probes for imaging soft, hydrated biofilms without damage [4] | Critical for obtaining accurate topographical and mechanical data. |
| High-Aspect-Ratio (HAR) AFM Probes | Probes with sharp, tall tips for accurate imaging of rough biofilms [2] [48] | Prevents tip artifacts and enables better resolution of deep biofilm structures. |
| Tryptic Soy Broth (TSB) / Agar | Standard nutrient medium for growing S. aureus and other biofilms [46] | Medium composition can significantly influence biofilm structure and matrix production. |
| Sodium Hypochlorite (NaOCl) | A common antimicrobial agent for biofilm challenge studies [46] | Titrate to know the exact concentration of chlorine; allows for creating dose-response curves. |
Diagnostic and Workflow Diagrams
Diagram 1: Diagnosing AFM-CFU Data Mismatches
Diagram 2: Troubleshooting Common AFM Imaging Problems
Technical Support Center
Frequently Asked Questions (FAQs)
FAQ 1: What is the primary advantage of using AFM over other microscopy techniques for hydrated biofilm studies? AFM provides quantitative, nanoscale resolution imaging of topographical features and nanomechanical properties under physiological, liquid conditions without the need for extensive sample preparation that can introduce artifacts, such as dehydration, staining, or metal coating required by electron microscopy [5] [49] [50].
FAQ 2: Our biofilm samples are consistently displaced or damaged during AFM scanning. How can we improve sample stability? Sample displacement is a common challenge when imaging soft, hydrated biological materials. We recommend improving immobilization through chemical or mechanical methods:
- Chemical Immobilization: Use substrates functionalized with poly-L-lysine or other adhesion-promoting molecules to strengthen the attachment of cells to the surface [1].
- Mechanical Immobilization: Utilize porous membranes or micro-fabricated polydimethylsiloxane (PDMS) stamps with pore sizes tailored to your specific microbial cells to physically trap them during scanning [1].
FAQ 3: How can we quantitatively measure the cohesive strength of a hydrated biofilm using AFM? The cohesive energy of a biofilm can be quantified in situ using an AFM-based abrasion method. This involves:
- Acquiring a topographical image of a biofilm region at a low applied force (~0 nN) to establish a baseline [19].
- Scanning a smaller sub-region repeatedly at a high load (e.g., 40 nN) to abrade and displace biofilm material [19].
- Re-imaging the original larger area at low force and calculating the volume of displaced biofilm from the height difference [19].
- The cohesive energy (in nJ/μm³) is calculated as the frictional energy dissipated during abrasion divided by the volume of biofilm displaced. This technique has been validated, showing that cohesive energy increases with biofilm depth and with the addition of calcium ions [19].
FAQ 4: What AFM imaging mode is most suitable for visualizing the fine structure of hydrated biofilms without causing damage? Tapping mode (also called intermittent contact mode) is highly recommended for hydrated biofilms. In this mode, the tip oscillates and only briefly contacts the sample, minimizing lateral (dragging) forces that can damage or displace soft, delicate structures like extracellular polymeric substances (EPS) and flagella [1] [5]. Simultaneously acquired phase images can also help distinguish between different material components, like cells and the surrounding EPS matrix, based on variations in surface mechanical properties [1].
FAQ 5: Can AFM be used to assess the effect of an antimicrobial treatment on single cells within a biofilm? Yes, AFM is an excellent tool for this. You can conduct nanoindentation experiments on individual cells before and after treatment. By performing force-distance curves on a cell's surface and applying a mechanical model (e.g., the Hertz model), you can quantify changes in nanomechanical properties, such as the elastic (Young's) modulus [1]. A significant increase in cell stiffness often indicates successful antimicrobial action, as it can correlate with cell death or physiological stress [49].
Troubleshooting Guides
Problem: Poor Quality Images with Streaks or Blurs in Liquid
- Potential Cause 1: Contaminated AFM tip or sample debris.
- Solution: Ensure thorough cleaning of the fluid cell and cantilever. Filter buffers and solutions to remove particulates.
- Potential Cause 2: Unstable thermal or acoustic drift.
- Solution: Allow the AFM system sufficient time to thermally equilibrate after introducing liquid. Perform imaging in a quiet, draft-free location, or use an acoustic enclosure.
- Potential Cause 3: Incorrect setpoint or feedback gains in tapping mode.
- Solution: Optimize the amplitude setpoint and slowly adjust the proportional and integral gains to achieve stable feedback without oscillating.
Problem: Inconsistent Force Curve Measurements
- Potential Cause 1: Tip contamination.
- Solution: Clean the tip with a UV-ozone cleaner or plasma cleaner before use. Confirm tip cleanliness by performing force curves on a clean, reference surface in a clean fluid.
- Potential Cause 2: Nonspecific adhesion between the tip and the biofilm matrix.
- Potential Cause 3: Variability in the biofilm surface itself.
- Solution: Acquire a large number of force curves (e.g., 256x256 grid) over the area of interest to create a force volume map, allowing you to account for and visualize spatial heterogeneity [1].
Experimental Protocols & Data Presentation
Detailed Protocol: Assessing Antimicrobial Efficacy via Nanoindentation
This protocol measures changes in the nanomechanical properties of biofilm cells after exposure to an antimicrobial agent.
- Biofilm Growth: Grow a hydrated biofilm on a suitable substrate (e.g., PFOTS-treated glass, mica, or functionalized silicon) in your chosen growth medium for the desired time [5].
- Baseline Measurement (Pre-treatment):
- Mount the biofilm in the AFM liquid cell with appropriate growth medium or buffer.
- Locate a region of interest using optical microscopy or a large-area AFM scan [5].
- On several individual cells within this region, acquire force-distance curves using a sharp, non-functionalized tip. Use a minimum trigger force and a slow approach rate (e.g., 0.5-1 µm/s) to avoid damaging the cells.
- Fit the retraction portion of the force curve with the Hertz model to calculate the initial Young's modulus for each cell [1].
- Antimicrobial Treatment:
- Gently perfuse the liquid cell with a solution of the antimicrobial agent at the desired concentration. Avoid introducing air bubbles.
- Incubate for the required treatment time.
- Post-treatment Measurement:
- Return the liquid cell to the original buffer solution to remove any unbound antimicrobial.
- Relocate the same cells or a similar population and repeat the force-distance curve measurements under identical conditions.
- Data Analysis:
- Compare the average Young's modulus of the cells before and after treatment using a statistical test (e.g., Student's t-test). A statistically significant increase in stiffness is a strong indicator of antimicrobial-induced cell damage or death [49].
Quantitative Data from Literature
The following table summarizes key quantitative findings from AFM studies on biofilms and antimicrobial effects.
Table 1: Quantitative AFM Measurements on Biofilms
| Measurement Type | Sample/Context | Quantitative Finding | Significance |
|---|---|---|---|
| Cohesive Energy [19] | 1-day biofilm from activated sludge | Increased from 0.10 ± 0.07 nJ/µm³ (top) to 2.05 ± 0.62 nJ/µm³ (with depth) | Quantifies increasing biofilm strength and stability with depth. |
| Cohesive Energy (Ca²⁺ effect) [19] | Biofilm with 10 mM CaCl₂ added | Increased from 0.10 ± 0.07 nJ/µm³ to 1.98 ± 0.34 nJ/µm³ | Demonstrates calcium's role in enhancing biofilm matrix strength. |
| Cell Dimensions [5] | Pantoea sp. YR343 cells | ~2 µm in length and ~1 µm in diameter | Establishes baseline cellular morphology for the studied organism. |
| Flagella Dimensions [5] | Pantoea sp. YR343 | ~20–50 nm in height, extending tens of micrometers | Highlights AFM's capability to resolve fine extracellular structures. |
Research Reagent Solutions
Table 2: Essential Materials for AFM Biofilm Studies
| Item | Function/Application | Examples & Notes |
|---|---|---|
| Functionalized Substrates | Promotes strong, irreversible adhesion of cells for stable imaging. | PFOTS-treated glass [5], poly-L-lysine coated surfaces [1], mica. |
| Micro-fabricated Stamps | Physically traps microbial cells of specific sizes for high-resolution imaging. | PDMS stamps with 1.5–6 µm wide pits [1]. |
| Sharp AFM Probes | High-resolution topographical imaging in tapping mode. | Silicon nitride or silicon tips with high resonance frequency for liquid. |
| Colloidal Probes | Measures average interaction forces and nanomechanical properties over a larger area. | Cantilevers with a glued spherical particle (e.g., silica) [1]. |
| Liquid Cell | Enables AFM operation under fully hydrated, physiological conditions. | Sealed fluid cell to maintain buffer environment around the sample [49]. |
Workflow Visualization
Experimental Workflow for Antimicrobial Assessment
AFM Antimicrobial Assessment Workflow
Biofilm Immobilization Strategies
Biofilm Immobilization Decision Tree
Atomic Force Microscopy (AFM) is a powerful tool in biofilm research, enabling nanoscale topographical imaging and force measurements under physiological conditions. However, its application to hydrated biofilm structures presents unique challenges that can limit data interpretation and experimental success. This technical support guide addresses common limitations and provides troubleshooting methodologies to help researchers obtain reliable, high-quality data for their studies on biofilm mechanics, structure, and function.
FAQs: Addressing Common AFM Challenges in Biofilm Research
Q1: What is the fundamental trade-off between resolution and field of view when imaging biofilms with AFM?
Conventional AFM has a limited scanning range, typically below 100×100 µm, restricted by piezoelectric actuator constraints [5]. This creates a scale mismatch where high-resolution cellular and subcellular details cannot be linked to the millimeter-scale organization of functional biofilm architectures [5]. While AFM provides nanometer-scale resolution, you can only image small, isolated sections of a biofilm, making it difficult to study heterogeneity or representative areas.
Q2: How does sample preparation for hydrated biofilms often lead to experimental artifacts?
Immobilizing soft, hydrated biofilms securely enough to withstand scanning forces without altering their native properties is a significant challenge [1]. Inadequate immobilization results in cells being swept away by the tip, while overly aggressive chemical fixation can alter nanomechanical properties and viability [1]. Biofilms are mechanically robust but easily disrupted during AFM scanning, requiring careful optimization of attachment protocols.
Q3: Why does my AFM data show inconsistent mechanical properties for similar biofilm samples?
Variations in measurement parameters, tip geometry, and environmental conditions significantly influence nanomechanical results. The Hertz model, commonly used to calculate elastic moduli, assumes perfectly homogeneous smooth bodies, while biofilms are inherently heterogeneous [1]. Using different cantilevers, loading rates, or indentation depths will yield different modulus values for the same material.
Q4: What are the primary limitations for imaging dynamic processes in living biofilms?
Standard AFM imaging is slow compared to biofilm dynamics. Capturing high-resolution images can take minutes, during which cells can grow, move, or alter their surface properties [1]. Furthermore, continuous scanning can mechanically stimulate biofilms, potentially influencing the very processes you are trying to observe.
Troubleshooting Guides
Issue 1: Limited Field of View and Non-Representative Sampling
Problem: The scanned area is too small to represent the heterogeneous architecture of a mature biofilm.
Solution: Implement Automated Large-Area AFM Mapping [5].
- Methodology: Automate the AFM to collect multiple adjacent high-resolution images over a millimeter-scale area. Use machine learning-driven stitching algorithms to create a seamless composite image.
- Protocol:
- Program your AFM software for automated sequential imaging of predefined positions.
- Ensure a minimal (~5-10%) overlap between adjacent scan areas.
- Acquire images in tapping mode to reduce lateral forces on the soft biofilm.
- Use computational stitching (e.g., with ML-based algorithms) to merge tiles into a large-area map.
- Apply automated cell detection and classification to quantitatively analyze spatial organization from the large dataset [5].
Table 1: Comparison of AFM Imaging Modes for Biofilms
| Imaging Mode | Principle | Best for Biofilms | Key Limitations |
|---|---|---|---|
| Contact Mode | Tip in constant contact with surface [51] | Hard, well-immobilized samples; force spectroscopy [51] | High lateral forces can distort or remove soft biofilm material [1] |
| Tapping Mode | Tip oscillates and intermittently contacts surface [51] [1] | Most biofilms; reduces lateral forces; phase imaging for material contrast [1] | Can still deform soft surfaces in liquid; slower than contact mode [51] |
| Non-Contact Mode | Tip oscillates near surface without contact [51] | Delicate surface structures | Lower resolution; susceptible to noise in liquid environments [51] |
| Force Spectroscopy | Measures force-distance curves at points [51] [1] | Quantifying adhesion, elasticity, and turgor pressure [1] | Point-by-point measurement; slow for mapping; model-dependent analysis [1] |
Issue 2: Unstable Imaging and Sample Damage on Hydrated Biofilms
Problem: The AFM tip detaches or damages the biofilm during scanning in liquid.
Solution: Optimize Biofilm Immobilization Techniques [1].
- Methodology: Use a combination of mechanical trapping and benign chemical adhesion to securely immobilize biofilms without altering their native state.
- Protocol:
- Mechanical Entrapment: For single cells or early biofilms, use porous membranes or micro-fabricated PDMS stamps with pore dimensions matching the cell size to physically trap cells [1].
- Chemical Adhesion: Treat substrates (e.g., glass, mica) with poly-L-lysine or functionalize with carboxyl groups to improve adhesion.
- Bio-friendly Additives: Add divalent cations (e.g., Mg²⁺, Ca²⁺) to the immersion liquid. These cations can bridge negative charges on the bacterial and substrate surfaces, enhancing attachment without fixatives [1].
- Validate Immobilization: Start with low scanning forces and amplitude setpoints in tapping mode, gradually increasing to stable imaging parameters.
Issue 3: Interpreting Complex Force Curves and Mechanical Properties
Problem: Force-distance curves on biofilms are complex and difficult to interpret quantitatively.
Solution: Adopt a Standardized Force Spectroscopy and Analysis Workflow [1] [4].
- Methodology: Systematically collect and analyze force-volume maps (arrays of force curves) using appropriate contact mechanics models and control experiments.
- Protocol:
- Calibration: Precisely calibrate the cantilever's spring constant and sensitivity on a clean, rigid substrate (e.g., glass or silicon) in the same liquid.
- Acquisition: Collect force-volume maps over the area of interest. Use a sufficient number of points and curves to account for heterogeneity.
- Analysis:
- Use the Hertz contact model for initial analysis of initial elastic response, but be aware it assumes homogeneity [1].
- For more accurate modeling, consider the Oliver-Pharr method or other models that account for adhesion (e.g., JKR, DMT models) [4].
- Always report the model used, along with all parameters (tip radius, indentation depth, Poisson's ratio assumption).
- Control: Functionalize the AFM tip with specific chemical groups (e.g., -CH3, -COOH) or biomolecules to probe specific interactions within the biofilm matrix [51].
The following workflow outlines the key steps and decision points for performing AFM-based mechanical characterization of biofilms.
Issue 4: Low Throughput and Inefficient Data Analysis
Problem: AFM data acquisition is slow, and analyzing large datasets (e.g., from force maps or large areas) is prohibitively time-consuming.
Solution: Integrate Machine Learning (AI) for Automated Operation and Analysis [5] [7].
- Methodology: Leverage AI and machine learning algorithms to automate scanning, enhance image quality, and segment features in large datasets.
- Protocol:
- Automated Scanning: Use AI-driven software for optimal site selection and scanning parameter adjustment, enabling continuous, multi-day experiments without supervision [5].
- Image Enhancement: Apply deep learning models (e.g., convolutional neural networks) to denoise images, correct distortions, and even reconstruct high-resolution images from faster, sparse scans [7].
- Feature Analysis: Train machine learning models to automatically detect, segment, and classify cells, flagella, and EPS components from topographical and phase images [5]. This is essential for analyzing datasets containing thousands of cells [5].
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for AFM Biofilm Studies
| Item | Function in AFM Experiment | Key Considerations |
|---|---|---|
| Poly-L-Lysine | Coats substrate to improve cell adhesion via electrostatic interactions [1] | Can alter surface chemistry and potentially affect cell physiology; use low concentrations. |
| Micro-fabricated PDMS Stamps | Mechanically traps individual cells for stable single-cell analysis [1] | Must be fabricated with pore sizes specific to the microbe being studied. |
| Functionalized AFM Tips | Probes specific chemical interactions (e.g., protein-protein, lectin-carbohydrate) within the biofilm [51] | Requires careful chemistry; specificity must be validated with control experiments. |
| Divalent Cations (Mg²⁺, Ca²⁺) | Added to liquid medium to enhance biofilm attachment to substrates by charge bridging [1] | A bio-friendly method that minimally interferes with native cell state. |
| ML-Ready Analysis Software | Enables automated segmentation, classification, and analysis of large AFM datasets [5] | Requires initial training but drastically increases analysis throughput and objectivity. |
Conclusion
AFM imaging of hydrated biofilms, while challenging, is an indispensable tool for understanding the fundamental properties that underpin biofilm resilience in medical environments. By mastering advanced immobilization techniques, operating modes, and leveraging new technologies like AI and large-area automation, researchers can overcome traditional limitations. The integration of AFM with other multimodal techniques provides a more holistic view, validating findings and enriching data interpretation. Future directions point toward increasingly automated, intelligent systems capable of long-term, dynamic monitoring of live biofilms under physiological conditions. These advancements will significantly accelerate the discovery of novel therapeutic interventions and biofilm control strategies, ultimately impacting drug development and clinical outcomes in the fight against persistent infections.
References
- 1. Atomic Force Microscopy of Biofilms—Imaging, Interactions ... [https://www.intechopen.com/chapters/50739]
- 2. 4 Common Imaging Problems (and how to fix them) - NuNano [https://www.nunano.com/blog/2020/11/23/4-common-imaging-problems-and-how-to-fix-them]
- 3. Atomic Force Microscopy (AFM) on Biopolymers and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8949482/]
- 4. Advances in biofilm characterization: utilizing rheology and ... [https://link.springer.com/article/10.1007/s42114-024-00950-2]
- 5. Analysis of biofilm assembly by large area automated AFM [https://www.nature.com/articles/s41522-025-00704-y]
- 6. DOE Scientists Develop Novel AFM Method to Transform ... [https://www.labcompare.com/617-News/620684-DOE-Scientists-Develop-Novel-AFM-Method-to-Transform-Biofilm-Research/?catid=61]
- 7. Artificial intelligence-empowered imaging technologies for ... [https://www.sciencedirect.com/science/article/abs/pii/S1385894725084311]
- 8. How To Tackle Artifacts In Atomic Force Microscopy Imaging [https://eureka.patsnap.com/report-how-to-tackle-artifacts-in-atomic-force-microscopy-imaging-solutions]
- 9. A guide for nanomechanical characterization of soft matter ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12166430/]
- 10. Atomic Force Microscopy, a Powerful Tool in Microbiology - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC135344/]
- 11. Tips and Tricks for Frustrated AFM Users [https://www.afmworkshop.com/newsletter/frustrated-with-your-afm?srsltid=AfmBOoo51okdXdhLi7A1OUTXpUX1NUtQAOQHWuc2WydkZW6m1A-rCefA]
- 12. A Brief Guide to AFM Probe Selection - Emma Benjaminson [https://sassafras13.github.io/ProbeSelection/]
- 13. True Non-Contact™ Mode [https://www.parksystems.com/en/products/research-afm/AFM-modes/Imaging-modes/TrueNon-ContactMode]
- 14. Choosing the Right Nanoscale Imaging Technique [https://www.nanosurf.com/insights/choosing-the-right-nanoscale-imaging-technique]
- 15. Tissue section AFM: In situ ultrastructural imaging of native ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2877882/]
- 16. Contact Mode vs. Tapping Mode AFM Comparison [https://www.nanoandmore.com/contact-vs-tapping-mode-afm?srsltid=AfmBOootA66tWddvyu7QrPAIvHMGPUNVDCXM13ufpiI9_wKD-PpKhZ3P]
- 17. Modes of Operation [https://www.doitpoms.ac.uk/tlplib/afm/modes_operation.php]
- 18. Tips and Tricks for Frustrated AFM Users [https://www.afmworkshop.com/newsletter/frustrated-with-your-afm?srsltid=AfmBOoqf9evufvQiS2ZG7Qq4m47Sl2BDU8wnihfQz-gge5skdQtH8OHa]
- 19. Biofilm Cohesiveness Measurement Using a Novel Atomic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1892862/]
- 20. Novel Combination of Atomic and Epifluorescence... Force Microscopy [https://pmc.ncbi.nlm.nih.gov/articles/PMC2223268/]
- 21. Dynamic immobilization of bacterial cells on biofilm in a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10992621/]
- 22. Nanomechanical Characterization with AFM [https://www.bruker.com/en/products-and-solutions/microscopes/materials-afm/resource-library/an-160-nanomechanical-characterization-with-afm.html]
- 23. Atomic Force Microscopy FAQs [https://www.bruker.com/en/products-and-solutions/microscopes/materials-afm/faq.html]
- 24. Tips and Tricks for Frustrated AFM Users [https://www.afmworkshop.com/newsletter/frustrated-with-your-afm?srsltid=AfmBOorwfnkaCnJVEyZ1bokf5aYzwJgU3l7wf6v4j9evH9vbo-SE0sID]
- 25. A guide for nanomechanical characterization of soft matter ... [https://www.sciencedirect.com/science/article/pii/S2666166725002151]
- 26. New imaging approach transforms study of bacterial biofilms [https://www.ornl.gov/news/new-imaging-approach-transforms-study-bacterial-biofilms]
- 27. Tips and Tricks for Frustrated AFM Users [https://www.afmworkshop.com/newsletter/frustrated-with-your-afm?srsltid=AfmBOoqyHSG2pt53JRSQf4IFpAUFmpSDR5H5S2ERnX3gcE6ObcovHS2U]
- 28. Atomic Force Microsocopy: Key Unconventional Approach for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11603319/]
- 29. Automated morphological classification and quantification of ... [https://pubs.rsc.org/en/content/articlehtml/2025/na/d5na00665a]
- 30. Biofilms Exposed: Innovative Imaging and Therapeutic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12466655/]
- 31. Overcoming Challenges and Limitations Regarding the ... [https://www.mdpi.com/2079-6439/11/10/83]
- 32. Water-solid interfaces probed by high-resolution atomic ... [https://www.sciencedirect.com/science/article/pii/S0167572921000340]
- 33. Quantification of solvation forces with amplitude ... [https://www.sciencedirect.com/science/article/pii/S0021979725001456]
- 34. Cohesiveness and hydrodynamic properties of young ... [https://www.sciencedirect.com/science/article/abs/pii/S0043135411007858]
- 35. 5 Image Analysis Frustrations (and How to Fix Them) [https://mediacy.com/blog/frustrating-realities-image-analysis/]
- 36. How to Fix Vision AI API Image Analysis Failures [https://medium.com/@Adekola_Olawale/how-to-fix-vision-ai-api-image-analysis-failures-0642286a0bac]
- 37. Recognizing Panoramas: The Keystone of Automated ... [https://medium.com/the-modern-scientist/recognizing-panoramas-the-keystone-of-automated-image-stitching-355b45037bf7]
- 38. Question about sudden deprecation behavior of Image ... [https://learn.microsoft.com/en-us/answers/questions/2258821/question-about-sudden-deprecation-behavior-of-imag]
- 39. Calibration of AFM Cantilevers [https://physics.uwo.ca/~jhutter/afmcal.shtml]
- 40. Easy and direct method for calibrating atomic force ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3018784/]
- 41. SRM 3461: An Accurate and Precise Force Calibration ... [https://www.nist.gov/news-events/news/2023/01/srm-3461-accurate-and-precise-force-calibration-artifact]
- 42. Method for lateral force calibration in atomic ... [https://www.sciencedirect.com/science/article/abs/pii/S0304399116303941]
- 43. Accurate Calibration of Atomic Force Microscope ... [https://www.nist.gov/mml/mmsd/nanomechanical/accurate-calibration-atomic-force-microscope-cantilever-spring-constants]
- 44. In-situ microscopy and digital image correlation to study the ... [https://link.springer.com/article/10.1007/s43939-025-00208-8]
- 45. A Comprehensive Analysis of Combined AFM/SEM ... [https://pubmed.ncbi.nlm.nih.gov/41222976/]
- 46. Interlaboratory study for the evaluation of three microtiter plate-based biofilm quantification methods | Scientific Reports [https://www.nature.com/articles/s41598-021-93115-w]
- 47. Quantitative and Qualitative Assessment Methods for Biofilm Growth: A Mini-review - PMC [https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6133255/]
- 48. Three-dimensional atomic force microscopy for ultra-high- ... [https://www.sciencedirect.com/science/article/pii/S0169433218330940]
- 49. The use of atomic force microscopy for studying ... [https://www.sciencedirect.com/science/article/abs/pii/S0927776501002338]
- 50. Microscopy Methods for Biofilm Imaging: Focus on SEM ... [https://www.mdpi.com/2079-7737/10/1/51]
- 51. Atomic Force Microscopy (AFM) on Biopolymers and ... [https://www.mdpi.com/2073-4360/14/6/1267]