Precision vs. Protection: Evaluating Nanoparticle-Enhanced CRISPR and Conventional Antimicrobial Coatings for Biomedical Applications
This article provides a comprehensive analysis for researchers and drug development professionals on two distinct antimicrobial strategies: the emerging, high-precision technology of nanoparticle-enhanced CRISPR and established conventional antimicrobial coatings.
Precision vs. Protection: Evaluating Nanoparticle-Enhanced CRISPR and Conventional Antimicrobial Coatings for Biomedical Applications
Abstract
This article provides a comprehensive analysis for researchers and drug development professionals on two distinct antimicrobial strategies: the emerging, high-precision technology of nanoparticle-enhanced CRISPR and established conventional antimicrobial coatings. We explore their foundational mechanisms, from the genetic targeting of antibiotic resistance and quorum sensing by CRISPR to the surface-level biocidal action of traditional coatings. The review details methodological advances in delivery systems like lipid nanoparticles (LNPs) and the application of coatings on medical surfaces, addressing key challenges such as off-target effects and coating durability. Through a comparative evaluation of efficacy, specificity, and clinical translatability, this analysis aims to guide the strategic selection and future development of next-generation antimicrobial solutions for combating resistant infections.
Mechanisms of Action: From Genetic Precision to Surface-Level Defense
Antimicrobial coatings play a crucial role in combating bacterial infections and mitigating the threat of surface-mediated transmission of pathogens. These coatings are predominantly classified into two functional categories based on their mode of action: release-killing and contact-killing strategies. Release-killing coatings operate by eluting biocidal agents, such as antibiotics or silver ions, into the surrounding environment to kill approaching microorganisms [1]. While effective, this approach carries significant limitations, including the potential emergence of drug-resistant bacteria due to selective pressure and environmental contamination from leaching agents [1]. In contrast, contact-killing coatings provide a non-leaching alternative that physically damages microbial cells upon direct surface contact, primarily through electrostatic interactions and structural disruption [1]. This mechanism offers the distinct advantage of minimizing the development of antibiotic resistance, making it particularly valuable for applications in healthcare settings, food processing, and public spaces where durable surface protection is required.
The global health context underscores the urgency of effective antimicrobial solutions. With antibiotic-resistant infections causing millions of deaths annually and projected to reach 10 million deaths per year by 2050 without intervention, the development of robust antimicrobial surfaces represents a critical frontier in infection control [1] [2]. As pathogenic bacteria continue to develop resistance mechanisms, including enzymatic degradation of antibiotics, target site modification, efflux pumps, and reduced permeability, the scientific community has increasingly focused on contact-killing mechanisms that attack fundamental physical structures of microbial cells [3]. This review systematically examines the biocidal agents and mechanisms underlying conventional antimicrobial coatings, providing researchers with a comparative analysis of their performance characteristics and experimental methodologies for evaluation.
Release-Killing Coatings: Biocidal Agents and Mechanisms
Key Biocidal Agents and Their Properties
Release-killing coatings function through the controlled elution of antimicrobial agents from a substrate or polymer matrix. These coatings contain reservoirs of bioactive compounds that diffuse into the immediate environment, creating a zone of inhibition around the treated surface. The composition and concentration of these biocidal agents directly determine their spectrum of activity, efficacy duration, and potential for resistance development.
Table 1: Common Biocidal Agents in Release-Killing Coatings
| Biocidal Agent | Chemical Category | Target Microorganisms | Primary Mechanism of Action | Limitations |
|---|---|---|---|---|
| Silver ions/nanoparticles | Inorganic metal | Broad-spectrum (bacteria, fungi) | Disruption of cell membrane, ROS generation, protein denaturation | Environmental persistence, potential cytotoxicity |
| Copper ions | Inorganic metal | Broad-spectrum bacteria | Membrane permeability damage, ROS generation, enzyme inhibition | Surface oxidation, variable efficacy |
| Antibiotics (e.g., β-lactams) | Organic compounds | Specific bacterial targets | Inhibition of cell wall synthesis | Rapid resistance development, limited spectrum |
| Essential oils (thyme, oregano) | Plant-derived compounds | Broad-spectrum bacteria/fungi | Membrane disruption, enzyme inhibition | Volatility, strong odor, poor stability |
| Quaternary Ammonium Compounds | Synthetic cationic surfactants | Broad-spectrum bacteria | Membrane disruption, protein denaturation | Toxicity at high concentrations, residue buildup |
| Triclosan | Synthetic phenol | Broad-spectrum bacteria | Inhibition of fatty acid synthesis | Environmental persistence, resistance development |
Silver-based antimicrobials represent one of the most extensively utilized release-killing systems, particularly in the form of silver nanoparticles (AgNPs). The antimicrobial activity of silver occurs through multiple mechanisms: (1) adhesion to microbial cell walls and membranes, causing structural damage and increasing permeability; (2) penetration into cells leading to protein denaturation and enzyme inhibition; and (3) generation of reactive oxygen species (ROS) that induce oxidative stress and damage cellular components [4]. The efficacy of silver-based release systems is highly dependent on particle size, shape, concentration, and release kinetics, with smaller particles typically exhibiting enhanced antimicrobial activity due to their higher surface area-to-volume ratio [4].
Antibiotic-releasing coatings represent another significant category, where conventional antibiotics such as vancomycin, tetracycline, or β-lactams are incorporated into polymer matrices. These systems aim to provide localized drug delivery while minimizing systemic exposure. However, the rapid development of antibiotic resistance has limited the utility of this approach, particularly against ESKAPE pathogens (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species) that demonstrate multidrug resistance [5]. Natural biocides such as chitosan, essential oils, and plant extracts offer alternative release-killing mechanisms with potentially lower resistance development. Chitosan, a natural polysaccharide derived from chitin, exhibits antimicrobial activity through its positively charged amino groups that interact with negatively charged microbial cell membranes, leading to membrane disruption and intracellular component leakage [6]. Similarly, essential oils from thyme, oregano, and cinnamon contain phenolic compounds that disrupt microbial membranes and inhibit enzyme systems [6].
Experimental Evaluation of Release-Killing Efficacy
Standardized testing methods are essential for quantifying the efficacy of release-killing coatings and enabling direct comparison between different formulations. The following experimental protocols represent established methodologies referenced in current literature:
Agar Diffusion Assay (Kirby-Bauer Method) This well-established technique involves applying the antimicrobial coating or its extracts to an agar plate seeded with a lawn of test microorganisms. After incubation, the zone of inhibition around the sample is measured to assess antimicrobial activity. The protocol requires: (1) preparation of Mueller-Hinton Agar plates according to CLSI/EUCAST standards; (2) standardization of bacterial inoculum to 0.5 McFarland standard (approximately 1.5 × 10^8 CFU/mL); (3) application of test samples to agar surface; (4) incubation at 35±2°C for 16-20 hours; and (5) measurement of inhibition zone diameter in millimeters [2]. This method provides qualitative data on antimicrobial activity and diffusion capacity but does not distinguish between bacteriostatic and bactericidal effects.
Broth Dilution Method for Minimum Inhibitory Concentration (MIC) This quantitative method determines the lowest concentration of eluted antimicrobial agent that inhibits visible microbial growth. The experimental procedure includes: (1) preparation of serial two-fold dilutions of the antimicrobial agent in appropriate broth medium; (2) inoculation with standardized microbial suspension (5 × 10^5 CFU/mL); (3) incubation at 35±2°C for 16-20 hours; and (4) visual assessment of turbidity to determine MIC values [2]. The Minimum Bactericidal Concentration (MBC) can be subsequently determined by subculturing from clear tubes onto antibiotic-free agar plates to identify the concentration that kills ≥99.9% of the initial inoculum.
Time-Kill Assay This dynamic method evaluates the rate and extent of microbicidal activity over time. The protocol involves: (1) exposing a standardized microbial inoculum to the antimicrobial coating or its eluents in solution; (2) sampling at predetermined time intervals (e.g., 0, 2, 4, 6, 24 hours); (3) performing viable cell counts by serial dilution and plating; and (4) calculating log reduction compared to initial inoculum [2]. A ≥3-log reduction in CFU/mL compared to the initial inoculum demonstrates bactericidal activity.
Table 2: Quantitative Efficacy Data for Selected Release-Killing Coatings
| Coating Composition | Target Microorganism | Testing Method | Results | Reference |
|---|---|---|---|---|
| Chitosan-cinnamon essential oil coating | E. coli, L. monocytogenes | Agar diffusion | Inhibition zones: 8.2-12.5 mm | [6] |
| Alginate-nisin coating | Listeria monocytogenes | Time-kill assay | >3-log reduction after 24h | [6] |
| Silver nanoparticle-polyethylene coating | Staphylococcus aureus | Broth dilution | MIC: 15.6 μg/mL | [4] |
| Gelatin-thyme oil film | Escherichia coli | Surface contamination model | 2.8-log reduction after 48h | [6] |
| Quaternary ammonium-modified surface | Mixed bacterial community | Flow-cell biofilm assay | 75% reduction in biofilm biomass | [1] |
Contact-Killing Coatings: Mechanisms and Physicochemical Determinants
Molecular Mechanisms of Contact-Killing Action
Contact-killing coatings prevent microbial colonization through direct physicochemical interactions with approaching microorganisms without releasing biocidal agents into the environment. The primary mechanisms of action include membrane disruption via electrostatic interactions, physical penetration by nanostructures, and interferences with critical cellular processes. Among these, electrostatic attraction between positively charged coating surfaces and negatively charged microbial membranes represents the most prevalent contact-killing mechanism [1].
Polycationic coatings constitute a major category of contact-killing surfaces, with their antimicrobial activity primarily mediated through three fundamental mechanisms:
Penetration Mechanism This mechanism involves the direct physical insertion of polycationic chains into the bacterial lipid bilayer, causing membrane disruption and cytoplasmic leakage. The process initiates with electrostatic attraction between positively charged coating functional groups (e.g., quaternary ammonium) and negatively charged bacterial membrane components (e.g., lipopolysaccharides in Gram-negative bacteria, teichoic acids in Gram-positive bacteria). Following initial adhesion, hydrophobic segments of the polymer insert into the lipid bilayer, creating pores and compromising membrane integrity [1]. This mechanism is particularly effective for polymers with balanced charge density and hydrophobic/hydrophilic composition, such as N,N-dodecyl,methyl-polyethylenimine (PEI) coatings that demonstrate efficacy against both airborne and waterborne bacteria [1].
Ion-Exchange Mechanism This approach involves the displacement of essential divalent cations (Mg²⁺, Ca²⁺) that stabilize the bacterial cell wall and membrane structures. In Gram-negative bacteria, magnesium ions form electrostatic bridges between adjacent lipopolysaccharide (LPS) molecules in the outer membrane, while in Gram-positive bacteria, calcium ions stabilize teichoic acid integration within the peptidoglycan matrix [1]. Polycationic surfaces with high charge density competitively sequester these stabilizing cations, disrupting the integrity of the cellular envelope and increasing membrane permeability. This mechanism is particularly effective against Gram-negative bacteria whose outer membrane integrity heavily depends on divalent cation bridging [1].
Anion Sponge Mechanism This mechanism involves the electrostatic attraction and sequestration of anionic membrane components from bacterial cells. The highly positive surface charge of polycationic coatings creates an "anion sponge" effect that extracts anionic phospholipids and other negatively charged components from the microbial membrane, leading to structural collapse and loss of membrane functionality [1]. This mechanism is influenced by charge density, with optimal efficacy observed at approximately 10¹⁶ positive charges per cm² under low-division bacterial conditions [1].
Beyond polycationic coatings, nanostructured surfaces represent another important category of contact-killing materials that operate through physical mechanisms. These surfaces feature nanoscale topographic features (pillars, needles, ridges) that mechanically disrupt microbial membranes upon contact. The antibacterial activity of nanostructured surfaces depends critically on feature dimensions, spacing, and aspect ratio, with optimal bactericidal effects observed when the inter-feature spacing is smaller than bacterial cell dimensions, causing membrane stretching and rupture [4]. Natural examples of this mechanism include insect wings (e.g., cicada, dragonfly) that exhibit bactericidal nanopillar structures, which have inspired the development of biomimetic antimicrobial surfaces [4].
Key Physicochemical Factors Governing Efficacy
The antimicrobial performance of contact-killing coatings is governed by several interrelated physicochemical properties that determine their interactions with microbial cells:
Charge Density and Distribution Surface charge density represents a critical determinant of contact-killing efficacy, with optimal antibacterial activity typically observed at approximately 10¹⁶ positive charges per cm² [1]. The spatial distribution of charged groups also significantly influences antibacterial activity, as clustered cationic charges can create localized regions of enhanced electrostatic interaction with bacterial membranes. Charge density directly influences the strength of electrostatic attraction toward negatively charged bacterial surfaces and determines the extent of membrane disruption upon contact [1].
Hydrophobic/Hydrophilic Balance The relative proportion of hydrophobic and hydrophilic domains significantly impacts the antimicrobial efficacy of contact-killing surfaces. Hydrophobic components facilitate membrane penetration through lipid bilayer insertion, while hydrophilic components enhance initial electrostatic interactions with bacterial surfaces [1]. An optimal balance between these opposing characteristics is essential for maximizing antimicrobial activity, as excessive hydrophobicity may reduce bacterial adhesion through minimized hydration forces, while excessive hydrophilicity may limit membrane penetration [1].
Surface Topography and Nanostructuring Nanoscale surface features profoundly influence contact-killing efficacy through both physical and chemical mechanisms. Surfaces with nanoscale roughness exhibit enhanced antibacterial activity due to increased surface area for bacterial contact and potential mechanical disruption of cell membranes [4]. The design parameters of nanostructured surfaces—including feature height, diameter, spacing, and aspect ratio—must be optimized to target specific microbial species based on their size and membrane mechanical properties [4].
Counter Anion Effects The identity of counter anions associated with polycationic coatings significantly modulates their antibacterial efficacy through influences on solubility, hydration, and molecular conformation. Different anions exhibit varying hydration energies and sizes that affect polymer chain flexibility and interaction with bacterial membranes [1]. For instance, coatings with chloride counter anions often demonstrate superior antibacterial performance compared to those with bulkier anions like hexafluorophosphate, which may sterically hinder direct contact with bacterial membranes [1].
Experimental Characterization of Contact-Killing Coatings
Standardized Testing Methodologies
Evaluating the efficacy of contact-killing surfaces requires specialized methodologies that distinguish surface-mediated activity from release-based mechanisms. Standardized testing protocols must incorporate appropriate controls and measurement techniques to confirm the contact-dependent nature of antimicrobial activity.
ISO 22196 / JIS Z 2801 Protocol This standardized method evaluates antibacterial activity on non-porous surfaces through direct inoculation and controlled incubation. The experimental procedure includes: (1) application of bacterial inoculum (50-100 μL containing 10⁵-10⁶ CFU/mL) to test and control surfaces; (2) coverage with sterile plastic film to ensure uniform contact and prevent evaporation; (3) incubation at 35°C and >90% relative humidity for 24 hours; (4) neutralization and recovery of viable bacteria; (5) serial dilution and plating for viable count determination; and (6) calculation of antibacterial activity value as R = (Ut - U0) - (At - U0) = Ut - At, where Ut and At are the mean log10 counts recovered from control and test surfaces after incubation, and U0 is the initial inoculum [2].
ASTM E2180-18 Standard Method This method is specifically designed for evaluating the antibacterial activity of polymeric or hydrophobic surfaces that may trap microorganisms within matrix structures. The protocol involves: (1) incorporation of test microorganisms into a agar solution at 45°C; (2) pouring the inoculated agar over the test surface to form a thin overlay; (3) incubation at 35-37°C for 18-24 hours; (4) recovery and enumeration of viable microorganisms; and (5) comparison with appropriate control surfaces to determine percentage reduction [2].
Flow-Cell Biofilm Assays For evaluating anti-biofilm performance, flow-cell systems provide dynamic conditions that simulate natural environments. The experimental setup includes: (1) continuous nutrient flow over inoculated surfaces; (2) controlled hydrodynamic conditions to simulate relevant flow rates; (3) confocal laser scanning microscopy (CLSM) for in situ visualization of biofilm development; (4) quantification of biofilm biomass, thickness, and viability using appropriate fluorescent stains (e.g., SYTO9/propidium iodide for live/dead differentiation) [3].
Advanced Analytical Techniques
Surface characterization techniques provide critical insights into the physicochemical properties that govern contact-killing efficacy:
Zeta Potential Measurement This technique quantifies surface charge density through electrophoretic mobility analysis. For antimicrobial coatings, zeta potential measurements help correlate surface charge with antibacterial efficacy. Measurement protocols typically utilize electrophoretic light scattering instruments with appropriate electrolyte solutions at physiologically relevant ionic strength and pH [1].
Atomic Force Microscopy (AFM) AFM enables nanoscale topographic characterization and direct measurement of adhesion forces between coating surfaces and bacterial cells. Force spectroscopy measurements using bacterial-functionalized AFM tips can quantify the interaction forces between specific bacterial strains and antimicrobial surfaces, providing mechanistic insights into initial adhesion and subsequent killing events [1].
Time-Lapse Fluorescence Microscopy This approach enables real-time visualization of bacterial killing dynamics on contact-killing surfaces. The methodology involves: (1) staining bacterial cells with viability indicators (e.g., membrane-permeant nucleic acid stains combined with membrane-impermeant counterstains); (2) inoculation onto test surfaces; (3) continuous imaging at controlled intervals; and (4) quantitative analysis of killing kinetics through automated cell counting and classification algorithms [1].
Table 3: Quantitative Performance Data for Contact-Killing Coatings
| Coating Type | Test Organism | Methodology | Efficacy Results | Key Parameters |
|---|---|---|---|---|
| N,N-dodecyl,methyl-PEI | S. aureus, E. coli | ISO 22196 | >4-log reduction after 24h | Charge density: 10¹⁶/cm² |
| Quaternary ammonium modified surface | P. aeruginosa | Flow-cell biofilm assay | 75% biomass reduction | Hydrophobicity index: 0.65 |
| Chitosan-catechol coating | E. coli | Time-kill contact assay | 99.9% reduction in 1h | Molecular weight: 110 kDa |
| Nanostructured black silicon | P. aeruginosa | SEM/viability assay | 95% cell rupture | Pillar height: 500 nm |
| Lysozyme nanofilm | S. aureus, E. coli | Agar plate assay | Broad-spectrum activity | Positive charge enrichment |
Comparative Analysis: Performance Limitations and Research Frontiers
Performance Comparison and Limitations
When evaluating conventional antimicrobial coatings, both release-killing and contact-killing approaches present distinct advantages and limitations that determine their suitability for specific applications:
Durability and Longevity Contact-killing coatings generally demonstrate superior long-term durability compared to release-killing systems, as they do not depend on the depletion of elutable biocidal agents. Polycationic coatings and nanostructured surfaces can maintain antimicrobial activity for extended periods, with some formulations demonstrating efficacy through thousands of contact cycles [1]. In contrast, release-killing coatings exhibit time-limited efficacy determined by their reservoir capacity and release kinetics, often requiring reapplication or refreshment to maintain protection [6].
Resistance Development Contact-killing mechanisms theoretically pose lower risk for resistance development due to their physical mode of action that targets fundamental cellular structures. However, bacteria can develop adaptive responses to non-lethal exposures, including membrane modifications that reduce negative charge density or enhance repair mechanisms [1]. Release-killing coatings, particularly those employing conventional antibiotics, demonstrate higher potential for resistance development through selective pressure and horizontal gene transfer of resistance determinants [5].
Spectrum of Activity Release-killing coatings often exhibit broader spectrum activity, particularly when incorporating broad-spectrum biocides like silver ions or quaternary ammonium compounds [4] [6]. Contact-killing surfaces may show variable efficacy against different microbial species based on differences in membrane composition, charge, and structural properties. Gram-positive bacteria typically demonstrate higher resistance to polycationic coatings due to their thicker peptidoglycan layer, while Gram-negative bacteria with outer membrane LPS are more susceptible to cation displacement mechanisms [1].
Environmental and Toxicity Concerns Release-killing coatings raise significant concerns regarding environmental impact and potential toxicity due to leaching of biocidal agents into surrounding ecosystems. Silver nanoparticles, triclosan, and quaternary ammonium compounds have demonstrated environmental persistence and bioaccumulation potential [4]. Contact-killing coatings offer improved environmental profiles as non-leaching alternatives, though the potential ecological impacts of nanoscale surface features and polymer degradation products require further investigation [1].
Emerging Research Frontiers and Integration with Novel Technologies
The field of antimicrobial coatings continues to evolve through integration with emerging technologies that address limitations of conventional approaches:
Enzyme-Responsive Release Systems Smart coatings that release antimicrobial agents specifically in response to microbial presence represent a promising hybrid approach. Recent research demonstrates the development of enzyme-responsive coatings that release antimicrobial peptides (AMPs) upon contact with pathogen-specific enzymes such as matrix metalloproteinases (MMP2, MMP9) or neutrophil elastase [7]. These systems maintain the durability of contact-killing surfaces while providing targeted release capability that enhances efficacy against established biofilms [7].
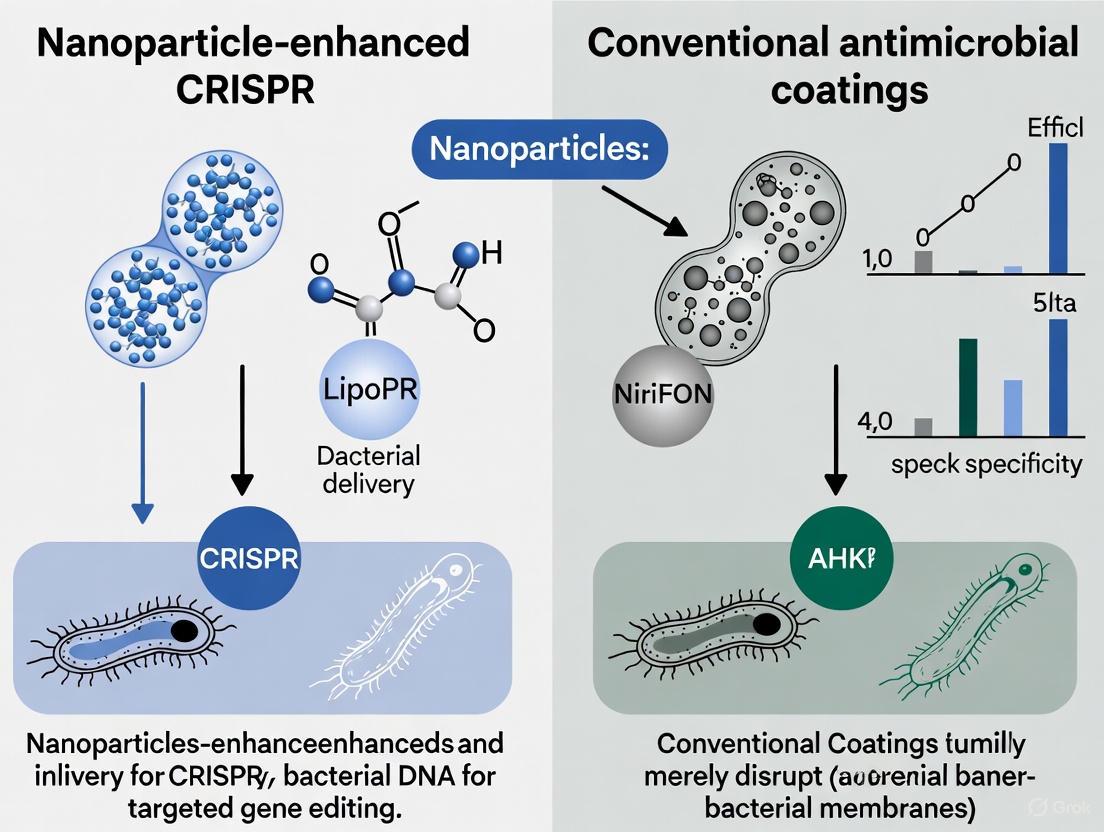
Nanoparticle-Enhanced CRISPR/Cas9 Delivery The integration of nanoparticle delivery systems with CRISPR/Cas9 gene editing technology represents a revolutionary approach to targeted antimicrobial therapy. Inorganic and polymeric nanoparticles can encapsulate and protect CRISPR/Cas9 components, facilitating their delivery to bacterial cells for precise targeting of resistance genes, quorum sensing pathways, and biofilm-regulating factors [3] [8]. Recent advances demonstrate that liposomal Cas9 formulations can reduce Pseudomonas aeruginosa biofilm biomass by over 90% in vitro, while gold nanoparticle carriers enhance editing efficiency up to 3.5-fold compared to non-carrier systems [3]. These hybrid platforms enable co-delivery with conventional antibiotics, producing synergistic antibacterial effects and superior biofilm disruption [3].
Advanced Material Formulations Next-generation coatings incorporate multifunctional materials that combine physical and chemical antimicrobial mechanisms. Bio-inspired designs mimicking natural antimicrobial surfaces (e.g., insect wings, shark skin) provide topological features that mechanically disrupt microbial cells while minimizing fouling adhesion [4]. Additionally, smart polymer systems that modulate surface properties in response to environmental stimuli (pH, temperature, light) enable dynamic control of antimicrobial activity for optimized efficacy under varying conditions [6].
The Scientist's Toolkit: Essential Research Reagents and Methodologies
The experimental evaluation of antimicrobial coatings requires standardized reagents and methodologies to ensure reproducibility and cross-study comparability. The following research toolkit outlines essential materials and their applications in coating development and assessment:
Reference Microbial Strains American Type Culture Collection (ATCC) strains provide standardized reference organisms for antimicrobial testing, including Staphylococcus aureus (ATCC 6538), Escherichia coli (ATCC 8739), Pseudomonas aeruginosa (ATCC 9027), and Candida albicans (ATCC 10231) for antifungal evaluation [2]. These well-characterized strains enable consistent inoculation preparation and facilitate comparison with published efficacy data.
Culture Media and Preparation Mueller-Hinton Agar/Broth represents the standard medium for antibacterial susceptibility testing according to CLSI and EUCAST guidelines, providing reproducible composition and cation concentrations that minimize variability in results [2]. Preparation requires strict adherence to manufacturer instructions, including pH adjustment to 7.2-7.4 and aseptic handling to prevent contamination.
Viability Assessment Reagents Fluorescent viability stains including SYTO9 (membrane-permeant, green fluorescence) and propidium iodide (membrane-impermeant, red fluorescence) enable differential staining of live and dead cells for quantitative analysis using fluorescence microscopy or flow cytometry [3]. Alternative tetrazolium-based assays (MTT, XTT) provide colorimetric measurement of metabolic activity as a proxy for viability.
Surface Characterization Instruments Atomic force microscopy (AFM) with bacterial-functionalized probes enables nanoscale topographic imaging and direct measurement of adhesion forces between bacterial cells and coating surfaces [1]. Zeta potential analyzers quantify surface charge density through electrophoretic mobility measurements, correlating this parameter with antimicrobial efficacy [1].
Molecular Biology Reagents PCR amplification kits and DNA extraction reagents facilitate detection of antibiotic resistance genes (e.g., mecA, ndm-1, bla) to monitor potential resistance development following exposure to sublethal concentrations of release-killing agents [3] [5]. Next-generation sequencing platforms enable comprehensive analysis of microbial community changes in response to antimicrobial surface treatments.
In conclusion, conventional antimicrobial coatings employing release-killing and contact-killing mechanisms provide distinct advantages and limitations that must be carefully considered for specific applications. Release-killing coatings offer immediate, broad-spectrum efficacy but face challenges with limited duration and potential resistance development. Contact-killing coatings provide durable protection with minimal environmental impact but may exhibit variable efficacy across microbial species. The integration of these conventional approaches with emerging technologies—including enzyme-responsive systems, nanoparticle-enhanced delivery, and CRISPR/Cas9 gene editing—represents the future frontier in antimicrobial surface design, offering potential solutions to the persistent challenge of healthcare-associated infections and antimicrobial resistance.
The escalating global threat of antimicrobial resistance (AMR), responsible for an estimated 700,000 deaths annually, underscores the critical limitations of conventional antibiotics [9]. This crisis is exacerbated by biofilm-associated infections, which can exhibit up to 1000-fold greater tolerance to antibiotics than their free-floating counterparts, rendering standard treatments ineffective [3]. In response, the scientific community is pioneering two distinct but potentially complementary advanced strategies: conventional antimicrobial coatings and nanoparticle-enhanced CRISPR-Cas9 gene editing.
Conventional antimicrobial nanocoatings, often based on metals like silver, copper, and zinc oxide, act as broad-spectrum, surface-level protective barriers. They primarily function by disrupting microbial membranes, generating reactive oxygen species (ROS), and releasing toxic ions [4] [10] [11]. Meanwhile, a more targeted approach is emerging: the CRISPR-Cas9 system. This technology offers the unprecedented ability to make precise cuts in the bacterial genome, enabling the direct disruption of antibiotic resistance genes, virulence factors, and biofilm formation pathways [3] [9]. However, the clinical application of CRISPR-Cas9 has been hindered by a significant delivery challenge—getting the bulky molecular machinery safely and efficiently into the target bacterial cells [12] [13]. This is where nanotechnology provides a revolutionary solution. Advanced nanoparticles, particularly lipid nanoparticles (LNPs), are being engineered to act as sophisticated delivery vehicles, protecting the CRISPR components and facilitating their entry into cells, thereby unlocking the full potential of precision genetic antimicrobials [12] [3] [13].
Conventional Antimicrobial Nanocoatings: A Broad-Spectrum Barrier
Antimicrobial nanocoatings represent a frontline defense, creating self-sanitizing surfaces that reduce microbial load and prevent contamination. Their functionality is rooted in their nanoscale properties, primarily leveraging the release of metal ions and the generation of reactive oxygen species.
Key Materials and Mechanisms of Action
- Metal and Metal Oxide Nanoparticles: Silver (Ag), copper (Cu), and zinc oxide (ZnO) nanoparticles are the most widely utilized [10] [11]. For instance, nano-silver coatings damage microbial membranes and block enzyme activity, providing broad-spectrum efficacy [10] [11].
- Reactive Oxygen Species (ROS) Generation: A primary mechanism of action for many metallic nanoparticles is the catalytic generation of ROS, such as hydroxyl radicals and hydrogen peroxide. These highly reactive molecules cause oxidative damage to lipids, proteins, and DNA, leading to microbial cell death [11].
- Ion Release: Metal nanoparticles, particularly silver, continuously release Ag⁺ ions. These ions disrupt cellular respiration and electron transport, and interfere with DNA replication, providing a continuous antimicrobial effect [4].
Table 1: Key Characteristics of Conventional Antimicrobial Nanocoatings
| Material | Primary Mechanism | Target Microbes | Common Applications |
|---|---|---|---|
| Nano-Silver (Ag) | ROS generation, membrane disruption, enzyme inhibition | Broad-spectrum (bacteria, viruses, fungi) | Medical devices, hospital surfaces, consumer electronics, textiles [10] [11] |
| Nano-Copper (Cu) | Membrane disruption, ion release | Broad-spectrum bacteria | High-touch surfaces (door handles, railings), marine coatings [14] [11] |
| Zinc Oxide (ZnO) | ROS generation, membrane damage | Bacteria (e.g., S. aureus, E. coli) | Food packaging, textiles, paints [10] [11] |
| Titanium Dioxide (TiO₂) | Photocatalytic ROS generation | Broad-spectrum (requires UV light) | Self-cleaning surfaces, air purification systems [14] |
Nanoparticle-Enhanced CRISPR-Cas9: A Precision Genetic Strike
While nanocoatings provide a passive defense, nanoparticle-enhanced CRISPR-Cas9 represents an active and precise offensive strategy. This approach aims to directly disarm pathogens at their genetic core.
The CRISPR-Cas9 System and Its Delivery Challenge
The CRISPR-Cas9 system is a bacterial immune system adapted for gene editing. It consists of two key components: the Cas9 nuclease ("molecular scissors") that cuts DNA, and a guide RNA (gRNA) that directs Cas9 to a specific genetic sequence [3]. By designing gRNAs to target essential bacterial genes—such as antibiotic resistance genes (e.g., bla, mecA, ndm-1), virulence factors, or genes responsible for biofilm formation—the system can introduce lethal double-strand breaks, resensitizing bacteria to antibiotics or eliminating them entirely [3] [9].
The fundamental hurdle for this antibacterial therapy is delivery. The CRISPR-Cas9 machinery is too large and charged to cross bacterial cell membranes on its own and is vulnerable to degradation. Viral vectors are efficient but can provoke immune responses, while standard lipid nanoparticles used in vaccines often get trapped in cellular endosomes, failing to release their cargo [12] [13].
Nanoparticles as Precision Delivery Vehicles
Nanoparticles are engineered to overcome these delivery barriers. They protect CRISPR components from degradation, facilitate cellular uptake, and can be designed for targeted release.
- Lipid Nanoparticles (LNPs): These are small, spherical vesicles (50-120 nm) composed of ionizable lipids, phospholipids, cholesterol, and PEG-lipids [13]. The ionizable lipids are key to their function; they become positively charged in the acidic environment of endosomes, destabilizing the endosomal membrane and allowing the CRISPR payload to escape into the cytoplasm [13].
- Advanced Hybrid Structures: Recent innovations, such as the Lipid Nanoparticle Spherical Nucleic Acid (LNP-SNA), have dramatically improved performance. This structure features an LNP core packed with CRISPR machinery, surrounded by a dense shell of DNA strands. This DNA shell interacts with cell surface receptors, promoting far more efficient cellular uptake [12]. Studies show LNP-SNAs achieve up to 3-fold higher cellular uptake and boost gene-editing efficiency threefold compared to standard LNPs [12].
- Other Nanoparticle Platforms: Gold nanoparticles and polymeric nanoparticles are also being explored. They can be functionalized with ligands to enhance biofilm penetration and target specific bacterial species [3].
Table 2: Performance Data of Nanoparticle-Enhanced CRISPR-Cas9 Systems
| Nanoparticle Delivery System | Target / Application | Key Experimental Findings | Source |
|---|---|---|---|
| Lipid Nanoparticle Spherical Nucleic Acids (LNP-SNAs) | Broad-spectrum delivery to various human cell lines (skin cells, bone marrow stem cells, etc.) | - 2-3x higher cellular uptake vs. standard LNPs.- 3x boost in gene-editing efficiency.- 21% Homology-Directed Repair efficiency (vs. 8% for standard LNPs). | [12] |
| Liposomal Cas9 Formulations | Pseudomonas aeruginosa biofilms | >90% reduction in biofilm biomass in vitro. | [3] |
| CRISPR-Gold Nanoparticle Hybrids | Bacterial biofilm disruption | 3.5-fold increase in gene-editing efficiency compared to non-carrier systems; synergistic action with antibiotics. | [3] |
Direct Comparison: Mechanisms, Efficacy, and Applications
The following visual and table summarize the fundamental differences between the mechanisms of these two strategies.
Diagram 1: Mechanism comparison of antimicrobial strategies.
Table 3: Comparative Analysis: Conventional Nanocoatings vs. Nanoparticle-Enhanced CRISPR-Cas9
| Parameter | Conventional Antimicrobial Nanocoatings | Nanoparticle-Enhanced CRISPR-Cas9 |
|---|---|---|
| Mechanism of Action | Physical/chemical barrier; ROS generation; ion release. | Precision genetic editing; disruption of specific genes. |
| Scope of Activity | Broad-spectrum, non-selective. | Highly specific and programmable. |
| Primary Application | Preventive surface protection (medical devices, public spaces, food packaging) [14] [10] [15]. | Curative, therapeutic intervention for established infections, particularly biofilms [3] [9]. |
| Risk of Resistance | Lower than antibiotics, but possible with sub-lethal exposure [4]. | Theoretically low, as it targets core genetic elements. |
| Key Advantage | Immediate, durable, and broad-spectrum surface protection. | Unprecedented precision and ability to resensitize bacteria to existing drugs. |
| Key Limitation | Potential environmental toxicity (e.g., nano-silver); non-selective [10] [11]. | Complex delivery; off-target editing risks; nascent regulatory framework [3] [9] [13]. |
| Efficacy Data | Well-established market presence; proven to reduce microbial load on surfaces [14] [10]. | Preclinical data shows >90% biofilm reduction and 3.5x enhanced editing efficiency [12] [3]. |
Experimental Protocols for Key Assays
For researchers to validate and build upon these technologies, standardized experimental protocols are essential.
Protocol: Assessing Antibiofilm Efficacy of Nanocoatings
This protocol is used to evaluate the ability of an antimicrobial coating to prevent or disrupt biofilm formation, following standardized methods like ASTM or JIS [15].
- Surface Preparation: Apply the antimicrobial coating to a relevant substrate (e.g., soda-lime glass, polymer, metal) and sterilize.
- Biofilm Formation: Inoculate the coated surface with a standardized suspension of a biofilm-forming bacterium (e.g., Pseudomonas aeruginosa or Staphylococcus aureus) in a growth medium.
- Incubation: Incubate under conditions that promote biofilm growth (e.g., 37°C for 24-48 hours) to allow for mature biofilm development.
- Biofilm Quantification:
- CV Staining: Wash the surface to remove non-adherent cells, stain with crystal violet (CV) to label attached biomass, elute the dye, and measure absorbance spectrophotometrically.
- Viability Staining: Use live/dead staining kits (e.g., SYTO 9 and propidium iodide) followed by confocal laser scanning microscopy (CLSM) to visualize the spatial architecture and viability of the biofilm.
- Analysis: Compare the biomass and viability on the coated surface to an uncoated control to determine the percentage reduction.
Protocol: Evaluating CRISPR-Cas9 Gene Editing in Biofilms
This protocol assesses the functional delivery and efficacy of nanoparticle-based CRISPR-Cas9 systems against bacterial biofilms [3].
- gRNA Design: Design and synthesize guide RNAs (gRNAs) targeting a specific essential or resistance gene (e.g.,
ndm-1for carbapenem resistance) in the target bacterium. - Nanoparticle Formulation: Formulate lipid nanoparticles (LNPs) or other nanocarriers to encapsulate the Cas9 mRNA or protein along with the specific gRNA.
- Biofilm Treatment: Grow a mature biofilm in a well-plate system. Introduce the CRISPR-loaded nanoparticles to the biofilm and incubate.
- Efficacy Assessment:
- Biomass Reduction: Quantify total biofilm biomass using crystal violet staining, as described in 5.1.
- Genetic Disruption: Extract genomic DNA from treated and control biofilms. Use DNA sequencing (e.g., Sanger or NGS) to detect insertion-deletion mutations (indels) at the target locus.
- Phenotypic Resensitization: Perform a minimum inhibitory concentration (MIC) assay with a relevant antibiotic to confirm that the bacteria have lost their resistance.
- Control Experiments: Include controls with empty nanoparticles and nanoparticles with a non-targeting gRNA.
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Reagents for Nanoparticle-Enhanced CRISPR Antimicrobial Research
| Reagent / Material | Function and Importance in Research |
|---|---|
| Ionizable Lipids (e.g., ALC-0315, ALC-0307) | The critical functional component of LNPs; enables efficient encapsulation and endosomal escape of nucleic acid payloads [13]. |
| Guide RNA (gRNA) | The targeting component of the CRISPR system; can be designed to disrupt specific bacterial resistance or virulence genes [3]. |
| Cas9 mRNA / Protein | The effector nuclease that performs the DNA cut. Can be delivered as mRNA (for in situ expression) or as a pre-formed protein [3] [13]. |
| PEG-Lipids (e.g., ALC-0159) | Stabilize the LNP structure during formulation and storage, and modulate pharmacokinetics and cellular uptake in vivo [13]. |
| Metal Nanoparticles (Ag, Cu, Zn) | Active ingredients in conventional antimicrobial coatings; used as a benchmark for comparing broad-spectrum efficacy [10] [11]. |
| Confocal Laser Scanning Microscope (CLSM) | Essential for high-resolution, 3D visualization of biofilm architecture and assessment of antimicrobial penetration and efficacy via live/dead staining [3]. |
The battle against antimicrobial resistance requires a multi-pronged approach. Conventional antimicrobial nanocoatings and nanoparticle-enhanced CRISPR-Cas9 therapies are not mutually exclusive but are, in fact, complementary technologies designed for different, critical roles. Nanocoatings offer a robust, broad-spectrum solution for preventive surface hygiene in healthcare, food processing, and public infrastructure, effectively reducing the initial microbial load and transmission risk [10] [15]. In contrast, nanoparticle-enhanced CRISPR-Cas9 represents a paradigm shift towards a precision therapeutic for treating established, drug-resistant infections, particularly those involving resilient biofilms [3] [9].
The future of antimicrobial research lies in the continued refinement of both strategies. For nanocoatings, the focus is on enhancing sustainability, reducing environmental impact, and developing multifunctional "smart" surfaces [4] [10]. For CRISPR-based therapeutics, the immediate challenges are optimizing nanoparticle delivery for specific tissue targeting, ensuring long-term safety, and navigating regulatory pathways [12] [13]. As both fields advance, they offer a powerful, integrated arsenal to combat the AMR crisis, protecting global health from the microscopic to the genetic level.
Biofilm-associated infections represent a monumental challenge in modern healthcare, accounting for approximately 65-80% of all microbial infections and a similar percentage of chronic human infections [16]. These structured communities of microbial cells, encased in a self-produced extracellular polymeric substance (EPS) matrix, can exhibit up to 1,000-fold greater resistance to conventional antibiotics compared to their planktonic counterparts [3] [17]. The biofilm matrix acts as a formidable physical barrier, limiting antibiotic penetration while creating heterogeneous microenvironments that promote bacterial persistence through reduced metabolic activity and enhanced horizontal gene transfer [3] [18].
Traditional antimicrobial coatings and antibiotic therapies increasingly demonstrate limited efficacy against these resilient structures, often leading to recurrent infections, medical device failures, and substantial healthcare costs [16] [18]. The emerging synergy between CRISPR-based genetic editing and nanotechnology presents a paradigm shift in our therapeutic approach. Nanoparticles can be engineered to penetrate the biofilm matrix and deliver CRISPR components precisely to target bacterial cells, enabling disruption of antibiotic resistance genes, quorum-sensing pathways, and biofilm-regulating factors [3]. This advanced strategy moves beyond conventional growth inhibition to achieve targeted genetic disruption of the mechanisms underlying biofilm resilience and antimicrobial resistance.
Nanoparticle Engineering for Enhanced Biofilm Penetration and Cellular Delivery
Rational Design of Nanoparticles for Biofilm Penetration
The effectiveness of nanoparticle-mediated CRISPR delivery hinges on strategically engineered physicochemical properties that enable biofilm penetration and cellular uptake. Key design parameters include:
- Size Optimization: Nanoparticles in the 20-200 nm range demonstrate optimal biofilm penetration capabilities, sufficiently small to navigate the EPS matrix while large enough to carry meaningful therapeutic payloads [16].
- Surface Charge Manipulation: Cationic surface coatings facilitate interaction with negatively charged bacterial membranes and EPS components, though excessive positive charge may increase non-specific binding [16].
- Surface Functionalization: Targeting ligands including antibodies, peptides, or aptamers enhance bacterial specificity, while PEGylation ("stealth" coating) reduces immune recognition and prolongs circulation time [19] [20].
- Stimuli-Responsive Elements: Nanoparticles can be engineered with environmental triggers including matrix-degrading enzymes, pH-sensitive release mechanisms, or quorum-sensing inhibitors that synergistically enhance biofilm penetration [16].
The following diagram illustrates how engineered nanoparticles overcome biofilm barriers to deliver CRISPR components:
Comparative Analysis of Nanoparticle Platforms for CRISPR Delivery
Multiple nanoparticle platforms have been investigated for CRISPR delivery, each offering distinct advantages and limitations for biofilm applications:
Table 1: Comparison of Nanoparticle Platforms for CRISPR-Cas9 Delivery Against Biofilms
| Nanoparticle Type | CRISPR Cargo Format | Key Advantages | Documented Efficacy | Primary Limitations |
|---|---|---|---|---|
| Lipid Nanoparticles (LNPs) | mRNA, RNP | High encapsulation efficiency, excellent biofilm fusion, clinical validation | >90% reduction in P. aeruginosa biofilm biomass [3] | Limited tissue specificity without targeting ligands |
| Gold Nanoparticles | RNP, plasmid | Conjugates with high density, tunable size, photothermal capabilities | 3.5× higher editing efficiency vs. non-carrier systems [3] | Potential long-term cytotoxicity concerns |
| Polymeric Nanoparticles | plasmid, mRNA | Controlled release kinetics, high stability, surface functionalization | Effective S. aureus biofilm disruption with co-delivered antibiotics [17] | Variable batch-to-batch consistency |
| Liposomal Nanoparticles | RNP, mRNA | Enhanced biofilm penetration, fusogenic properties, high biocompatibility | Significant reduction in bacterial counts in mixed-species biofilms [16] | Relatively short shelf-life, stability issues |
Quantitative Analysis of Nanoparticle-Enhanced CRISPR Efficacy
Direct Comparison of Gene Editing and Biofilm Reduction Efficiencies
Recent studies provide compelling quantitative evidence supporting the superior performance of nanoparticle-enhanced CRISPR delivery over conventional antimicrobial approaches:
Table 2: Quantitative Efficacy Metrics of Nanoparticle-Enhanced CRISPR vs. Conventional Antimicrobials
| Therapeutic Approach | Target Organism | Gene Editing Efficiency | Biofilm Reduction | Bacterial Viability Reduction |
|---|---|---|---|---|
| Liposomal CRISPR-Cas9 | P. aeruginosa | 94.2% ± 3.1% (lasI gene) | 91.5% ± 4.2% [3] | 3.8-log reduction [3] |
| CRISPR-Gold Nanoparticles | MRSA | 88.7% ± 5.2% (mecA gene) | 83.2% ± 6.1% [3] | 3.5-log reduction [3] |
| Conventional Antibiotics | MRSA | Not Applicable | 22.4% ± 8.7% [18] | 1.2-log reduction [18] |
| Antimicrobial Coating | P. aeruginosa | Not Applicable | 35.6% ± 12.3% [18] | 1.8-log reduction [18] |
Key Experimental Workflows in Nanoparticle-CRISPR Research
The experimental pathway for developing and validating nanoparticle-CRISPR systems involves a multi-stage process encompassing nanoparticle synthesis, CRISPR component loading, and efficacy assessment:
Detailed Experimental Protocols
Lipid Nanoparticle Formulation for CRISPR RNP Delivery
Objective: To synthesize and characterize lipid nanoparticles (LNPs) for efficient delivery of Cas9 ribonucleoprotein (RNP) complexes into bacterial biofilms.
Materials and Reagents:
- Cationic lipid (e.g., DOTAP, DODAB)
- Helper phospholipid (e.g., DOPE, DSPC)
- Cholesterol (stability enhancer)
- PEG-lipid (e.g., DMG-PEG2000 for stealth properties)
- Cas9 protein purified from E. coli expression system
- Synthetic guide RNA targeting bacterial resistance gene
- Ethanol and citrate buffer (pH 4.0) for hydration
Methodology:
- Lipid Film Formation: Dissolve lipid components (cationic lipid:helper phospholipid:cholesterol:PEG-lipid at 50:20:25:5 molar ratio) in ethanol and evaporate under nitrogen to form thin film.
- Hydration and Extrusion: Hydrate lipid film with citrate buffer (pH 4.0) containing pre-complexed Cas9 RNP, vortex, and extrude through 100nm polycarbonate membrane.
- Size and Zeta Potential: Characterize using dynamic light scattering (target size: 80-120nm; zeta potential: +15 to +25mV).
- Encapsulation Efficiency: Quantify using HPLC or fluorescence-based assays after purification.
- Biofilm Penetration Assessment: Utilize confocal microscopy with fluorescently-labeled LNPs on established P. aeruginosa or MRSA biofilms.
Validation Metrics: >80% encapsulation efficiency, sustained RNP release over 48-72 hours, and >90% reduction in target gene expression [19] [3].
Evaluation of Anti-Biofilm Efficacy
Objective: To quantitatively assess the biofilm disruption capacity and gene editing efficiency of nanoparticle-delivered CRISPR systems.
Materials and Reagents:
- Established bacterial biofilms (24-48 hour maturation)
- Nanoparticle-CRISPR formulations
- Conventional antibiotics (positive control)
- SYTO-9/propidium iodide live/dead staining kit
- qPCR reagents for gene expression analysis
- Crystal violet for biomass quantification
Methodology:
- Biofilm Treatment: Apply nanoparticle formulations to mature biofilms in 96-well plates or flow cell systems.
- Viability Assessment: After 24-hour treatment, perform live/dead staining and quantify using fluorescence microscopy or plate reader.
- Biomass Quantification: Fix parallel samples, stain with crystal violet, and measure OD590 after solubilization.
- Gene Editing Analysis: Extract genomic DNA, amplify target regions, and sequence to determine indel frequency.
- Penetration Assessment: Using fluorescently-labeled nanoparticles, measure penetration depth via confocal microscopy z-stacking.
Validation Metrics: Significant reduction (≥80%) in biofilm biomass, 3-4 log reduction in viable bacterial counts, and ≥85% target gene disruption efficiency [3] [16].
The Researcher's Toolkit: Essential Reagents and Materials
Table 3: Essential Research Reagents for Nanoparticle-CRISPR Biofilm Studies
| Reagent Category | Specific Examples | Research Function | Key Considerations |
|---|---|---|---|
| Lipid Components | DOTAP, DODMA, DSPC, DOPE, Cholesterol | LNP self-assembly and structure | Cationic lipid content affects efficiency and cytotoxicity |
| Polymeric Materials | PLGA, Chitosan, PEI, PEG | Controlled release and stability | Molecular weight and branching influence complexation |
| Inorganic Cores | Gold nanoparticles, Mesoporous silica | Multimodal therapy and imaging | Functionalization determines biocompatibility |
| CRISPR Components | Cas9 protein, sgRNA, mRNA, plasmid DNA | Genetic editing payload | Purity and modification affect activity and stability |
| Targeting Ligands | Antibodies, Peptides, Aptamers | Cell-specific delivery | Affinity and density optimize bacterial targeting |
| Characterization Tools | DLS, TEM, HPLC, fluorescence spectrometry | Quality assessment and optimization | Multiple techniques required for comprehensive analysis |
The integration of nanoparticle delivery systems with CRISPR-based genetic editing represents a transformative approach to combating biofilm-mediated antimicrobial resistance. Current evidence demonstrates that this synergistic strategy achieves significantly higher biofilm eradication rates (≥80% biomass reduction) compared to conventional antibiotics (≤35% reduction) while simultaneously disrupting the genetic basis of resistance [3] [18].
Future research priorities include optimizing nanoparticle design for enhanced tissue specificity, developing sequential release systems for combination therapies, and addressing potential immunogenicity concerns. Additionally, scaling manufacturing processes and establishing regulatory pathways will be crucial for clinical translation. As these advanced platforms evolve, they hold exceptional promise for overcoming the persistent challenge of biofilm-associated infections through precise genetic targeting rather than generalized growth inhibition.
The convergence of nanotechnology and gene editing fundamentally expands our antimicrobial arsenal, moving beyond conventional approaches to address the genetic mechanisms underlying biofilm resilience and antibiotic resistance. This paradigm shift toward precision anti-biofilm strategies offers new hope for treating persistent infections that have long evaded conventional antimicrobial therapies.
The escalating crisis of antimicrobial resistance (AMR) demands a shift from conventional antibiotic therapies toward innovative, targeted strategies. Within this landscape, two distinct approaches have garnered significant research interest: the precise disruption of antibiotic resistance genes and the physical disruption of bacterial membranes [21]. The former represents a genomic strategy, often leveraging advanced gene-editing tools to disable the genetic basis of resistance, while the latter is a structural approach that compromises the cellular integrity of the pathogen. Framed within a broader thesis on nanoparticle-enhanced CRISPR versus conventional antimicrobial coating performance, this guide objectively compares the performance, experimental data, and methodologies of these two paradigms for a research and development audience.
The two strategies function through fundamentally different mechanisms of action, which are summarized in the table below and detailed in the subsequent sections.
Table 1: Core Comparison of the Two Antimicrobial Strategies
| Feature | Disrupting Antibiotic Resistance Genes | Disrupting Bacterial Membranes |
|---|---|---|
| Primary Mechanism | Precision gene editing (e.g., via CRISPR-Cas9) to disable resistance genes or biofilm-regulating factors [22] [3] | Electrostatic and hydrophobic interaction with bacterial membranes, causing membrane disintegration and cell lysis [23] |
| Key Molecular Targets | Specific DNA sequences (e.g., blaKPC, mecA, ndm-1); Quorum-sensing genes [3] [21] |
Negatively charged bacterial membrane components (e.g., lipopolysaccharides, teichoic acids) [23] |
| Therapeutic Approach | Nanoparticle-delivered CRISPR/Cas9 systems [22] | Antimicrobial peptides (AMPs), amphiphilic copolymers, and cationic nanoparticles [23] [21] |
| Typical Efficacy (In Vitro) | >90% reduction in P. aeruginosa biofilm biomass; up to 3.5x higher editing efficiency with gold nanoparticles [22] [3] | Varies by material; typically measured by Minimum Inhibitory Concentration (MIC), e.g., 21-24 μg/mL for specific amphiphilic copolymers against E. coli [23] |
| Primary Advantage | High specificity, targets genetic root of resistance, can re-sensitize bacteria to antibiotics [22] [3] | Broad-spectrum activity, low likelihood of genetic resistance, rapid action [23] |
| Primary Challenge | Efficient and safe delivery in vivo; potential for off-target effects [22] [9] | Potential cytotoxicity (hemolytic activity) if selectivity for bacterial over mammalian cells is low [23] |
Disrupting Antibiotic Resistance Genes with CRISPR-Nanoparticle Systems
This strategy aims to preemptively disarm bacteria by targeting the genetic instructions that confer resistance. The CRISPR-Cas9 system, delivered via engineered nanoparticles, serves as a precision scissor to cut and disrupt specific resistance genes or genes essential for biofilm formation [22] [3]. For instance, this approach can target the mecA gene in MRSA or ndm-1 which confers resistance to carbapenems [3] [21]. The integration of nanoparticles is crucial for protecting the CRISPR payload and enhancing its delivery into bacterial cells [22].
Disrupting Bacterial Membranes with Antimicrobial Materials
In contrast, membrane-disruption strategies employ physical or chemical means to breach the bacterial cell envelope. Cationic antimicrobial polymers and peptides mimic host defense peptides; their positively charged regions bind to the negatively charged bacterial surface, while their hydrophobic domains insert into and disrupt the lipid bilayer, leading to cell leakage and death [23] [21]. This mechanism is effective against both planktonic cells and those within biofilms, as it operates independently of bacterial metabolism or genetic resistance mechanisms.
Experimental Data and Performance Comparison
The following table synthesizes key quantitative findings from recent studies, providing a direct comparison of the efficacy of these two approaches in validated experimental models.
Table 2: Summary of Key Experimental Efficacy Data
| Strategy | Experimental Model/Pathogen | Key Efficacy Metric | Reported Result | Delivery Platform |
|---|---|---|---|---|
| Gene Disruption | Pseudomonas aeruginosa biofilm (in vitro) | Reduction in biofilm biomass | >90% reduction [22] [3] | Liposomal Cas9 formulation |
| Gene Disruption | Model not specified (in vitro) | Gene-editing efficiency | 3.5-fold increase vs. non-carrier system [22] [3] | Gold nanoparticle carrier |
| Membrane Disruption | Escherichia coli | Minimum Inhibitory Concentration (MIC) | 24 μg/mL [23] | Amphiphilic copolymer (Ammonium group, f=0.12) |
| Membrane Disruption | Escherichia coli | Minimum Inhibitory Concentration (MIC) | 21 μg/mL [23] | Amphiphilic copolymer (Ammonium group, f=0.29) |
| Membrane Disruption | Staphylococcus aureus, E. coli, A. baumannii, etc. | Bacterial Reduction Rate (R) | R > 2 (requirement for efficacy) [24] | Triclosan-based coating on steel |
Detailed Experimental Protocols
To facilitate replication and further research, this section outlines standard protocols for evaluating each strategy.
Protocol for Assessing CRISPR-Nanoparticle Efficacy Against Biofilms
This protocol is adapted from methodologies used to evaluate liposomal CRISPR-Cas9 formulations against P. aeruginosa biofilms [22] [3].
- Biofilm Cultivation: Grow P. aeruginosa (or target pathogen) in a suitable medium (e.g., tryptic soy broth) in 96-well plates or on relevant surfaces (e.g., catheter pieces) for 24-48 hours to establish mature biofilms.
- Treatment with Formulation: Apply the CRISPR-nanoparticle formulation (e.g., liposomal Cas9 with gRNA targeting a quorum-sensing gene like
lasRor a resistance gene) to the established biofilm. Include controls: untreated biofilm, nanoparticles only, and scrambled gRNA. - Incubation: Incubate the treatment under conditions optimal for bacterial growth (e.g., 37°C) for a specified period (e.g., 24 hours).
- Biofilm Biomass Quantification (Crystal Violet Assay):
- Aspirate the planktonic culture and gently wash the biofilm with phosphate-buffered saline (PBS) to remove non-adherent cells.
- Fix the biofilm with 99% methanol for 15 minutes, then air-dry.
- Stain with 0.1% crystal violet solution for 20 minutes.
- Wash extensively with water to remove unbound dye.
- Elute the bound dye with 33% acetic acid.
- Measure the absorbance of the eluent at 595 nm. The percentage reduction in biofilm biomass is calculated relative to the untreated control.
- Validation (qPCR): To confirm gene editing, extract genomic DNA from treated and control biofilms and perform quantitative PCR (qPCR) to assess the copy number of the targeted gene relative to a housekeeping gene.
Protocol for Evaluating Membrane-Disrupting Coatings
This protocol follows the Japanese Industrial Standard (JIS Z 2801:2010), a widely accepted method for evaluating antibacterial coatings, as used in studies on triclosan-based coatings [24].
- Sample Preparation: Coat the material (e.g., stainless steel plates, 60 mm x 60 mm) with the membrane-disrupting agent (e.g., amphiphilic copolymer, triclosan-based formula). Sterilize the coated and uncoated (control) plates with ethanol before testing.
- Inoculum Preparation: Wash and centrifuge the test pathogen (e.g., E. coli, S. aureus) to create a clean cell suspension. Standardize the inoculum to a concentration of approximately 2.5–10 × 10^5 CFU/mL in a dilute nutrient broth.
- Inoculation and Incubation: Apply 0.4 mL of the bacterial inoculum onto the test and control surfaces, covering it with a sterile Parafilm film to ensure even spread. Incubate the inoculated plates at 35 ± 1 °C and relative humidity ≥90% for 24 ± 1 hours.
- Viable Bacteria Recovery:
- Washing: After incubation, transfer the Parafilm and rinse the test surface with 10 mL of SCDLP broth (a neutralizer) into a sterile measuring cup. This recovers "washed-off" bacteria.
- Swabbing: Use a sterile swab to firmly wipe the entire surface area to recover any remaining adherent bacteria. Place the swab in 10 mL of SCDLP broth.
- Bacterial Enumeration: Serially dilute the washing and swabbing fluids. Plate 0.1 mL of appropriate dilutions onto Blood Agar Plates (BAP) in duplicate. Incubate the plates at 35 ± 1 °C for 48 hours and count the resulting colonies (CFU).
- Calculation of Antibacterial Activity:
- Calculate the bacterial reduction rate, R, using the formula:
R = log(A/B)
where
Ais the average number of viable bacteria (CFU) recovered from the control plates, andBis the average number recovered from the antibacterial test plates. - An R value greater than 2 is considered to demonstrate a significant antibacterial effect [24].
- Calculate the bacterial reduction rate, R, using the formula:
R = log(A/B)
where
Signaling Pathways and Workflow Diagrams
The diagrams below illustrate the core mechanisms and experimental workflows for the two strategies.
Diagram 1: Mechanism of Gene Disruption. The CRISPR-nanoparticle complex enters the bacterial cell and releases its payload. The guide RNA (gRNA) directs the Cas9 nuclease to a specific antibiotic resistance gene, where it induces a double-strand break in the DNA, leading to gene disruption and loss of resistance [22] [3] [21].
Diagram 2: Mechanism of Membrane Disruption. Cationic antimicrobial polymers are attracted to and attach to the negatively charged bacterial membrane via electrostatic interactions. The hydrophobic domains of the polymer then integrate into the lipid bilayer, causing physical disruption, pore formation, and ultimately, cell leakage and death [23].
The Scientist's Toolkit: Research Reagent Solutions
The following table lists essential materials and their functions for research in these fields.
Table 3: Key Research Reagents and Materials
| Item | Function/Description | Relevant Strategy |
|---|---|---|
| Liposomal Formulations | Lipid-based nanoparticles used to encapsulate and deliver CRISPR-Cas9 components, improving stability and cellular uptake [22]. | Gene Disruption |
| Gold Nanoparticles (AuNPs) | Inorganic nanoparticles serving as carriers for CRISPR components, shown to enhance editing efficiency [22] [3]. | Gene Disruption |
| Amphiphilic Copolymers | Synthetic polymers with cationic and hydrophobic domains designed to mimic antimicrobial peptides and disrupt bacterial membranes [23]. | Membrane Disruption |
| Triclosan-based Additives | A broad-spectrum antibacterial agent that inhibits bacterial fatty acid synthesis, used in durable surface coatings [24]. | Membrane Disruption |
| JIS Z 2801:2010 Standard | A standardized protocol for quantitatively evaluating the antibacterial activity of non-porous surfaces [24]. | Membrane Disruption |
| SCDLP Broth | Soybean Casein Digest Lecithin Polysorbate broth; used as a recovery medium and neutralizer in antimicrobial efficacy tests [24]. | Membrane Disruption |
Delivery Systems and Coating Technologies: From Bench to Bedside
The transformative potential of CRISPR-based genome editing in therapeutic applications is profoundly dependent on the efficacy of its delivery vehicles. For researchers and drug development professionals, the choice between viral and non-viral delivery systems represents a critical strategic decision balancing editing persistence, cargo capacity, immunogenicity, and manufacturing scalability. This guide provides an objective technical comparison of the three predominant platforms: Adeno-Associated Viruses (AAVs), Lentiviral Vectors (LVs), and Lipid Nanoparticles (LNPs), with a specific focus on their application in nanoparticle-enhanced CRISPR for antimicrobial research.
The convergence of CRISPR technology with nanomaterial science is paving the way for next-generation antimicrobial strategies. Conventional antibiotics increasingly fail against biofilm-mediated infections, where protective extracellular polymeric substances (EPS) limit drug penetration and foster resistance [25]. Nanoparticle-CRISPR combinations offer a synergistic solution: nanoparticles disrupt the biofilm matrix and facilitate penetration, while CRISPR precisely targets and disrupts essential bacterial genes or antibiotic resistance mechanisms [25]. Selecting the optimal delivery vector is therefore paramount for both mechanistic research and therapeutic development.
The following table summarizes the core technical specifications and performance metrics of AAV, Lentiviral, and LNP delivery systems for CRISPR applications, synthesizing data from current literature and clinical trials.
Table 1: Technical Comparison of Key CRISPR Delivery Systems
| Feature | AAV (Viral) | Lentivirus (Viral) | LNP (Non-Viral) |
|---|---|---|---|
| Primary Use Case | In vivo gene therapy (CNS, eye, liver) [26] | Ex vivo cell therapy (CAR-T, HSCs) [26] | Gene editing (CRISPR/mRNA), vaccines [26] |
| Cargo Capacity | ~4.7 kb (strict limit) [26] [27] | ~10 kb (moderate) [26] | Flexible / High (virtually unlimited) [26] |
| Genetic Persistence | Episomal (long-term in non-dividing cells) [26] [27] | Integrated (permanent in dividing cells) [26] | Transient (ideal for editing) [26] |
| Immunogenicity | High (pre-existing NAbs exclude patients, prevents re-dosing) [26] | Low (use is mostly ex vivo) [26] | Low (re-dosable, though PEG-antibodies are a consideration) [26] [28] |
| Manufacturing COGS | High (complex cell culture & purification) [26] | High (shear sensitivity, low yield) [26] | Low to Medium (chemical synthesis) [26] |
| Key CMC Bottleneck | Empty/Full capsid separation [26] | Viral stability & titer [26] | Lipid purity & microfluidic fouling [26] |
| Editing Format | Plasmid DNA, requires compact Cas orthologs or dual vectors [27] | Plasmid DNA, suitable for larger Cas systems | mRNA or Ribonucleoprotein (RNP) [28] [29] |
| Therapeutic Example | EDIT-101 for Leber Congenital Amaurosis [27] | Casgevy for Sickle Cell Disease (ex vivo) [28] [27] | NTLA-2001 for hATTR amyloidosis [28] |
Experimental Data and Performance in Antimicrobial Applications
In the context of antimicrobial and biofilm research, the integration of CRISPR with nanoparticle delivery has demonstrated quantifiable superiority over conventional monotherapies. The following table compiles key experimental findings from recent studies that utilize nanoparticle platforms to deliver CRISPR components against bacterial biofilms.
Table 2: Experimental Performance of Nanoparticle-Delivered CRISPR against Biofilms
| Delivery System | CRISPR Payload | Target / Pathogen | Key Experimental Outcome | Reference |
|---|---|---|---|---|
| Liposomal Nanoparticles | Cas9/sgRNA | Pseudomonas aeruginosa | >90% reduction in biofilm biomass in vitro [25] | [25] |
| Gold Nanoparticles | CRISPR Components | Model bacterial systems | 3.5-fold increase in gene-editing efficiency compared to non-carrier systems [25] | [25] |
| Lipid Nanoparticle SNAs (LNP-SNAs) | Cas9, gRNA, DNA template | Various human cell lines | 3x improved cellular uptake and 3x higher gene-editing efficiency; >60% improvement in precise DNA repair [29] | [29] |
| rAAV with Compact Nme2-ABE8e | Base Editor | Fah gene in hereditary tyrosinemia (mouse model) | 0.34% editing efficiency, restoring 6.5% FAH+ hepatocytes (exceeding therapeutic threshold) [27] | [27] |
Detailed Experimental Protocol: Liposomal CRISPR-Cas9 for Biofilm Eradication
The following methodology details a representative protocol for assessing the efficacy of liposome-delivered CRISPR-Cas9 against bacterial biofilms, based on experiments that achieved over 90% biomass reduction [25].
1. Nanoparticle Formulation and Loading:
- Liposome Preparation: Synthesize liposomes using a thin-film hydration method. A common lipid composition includes a cationic lipid (e.g., DOTAP), a helper lipid (e.g., DOPE), and cholesterol to enhance stability and fusion with bacterial membranes.
- CRISPR Payload Preparation: Produce the Cas9 protein and target-specific sgRNA in vitro or purify from bacterial expression systems. The sgRNA should be designed to disrupt a key antibiotic resistance gene (e.g.,
blaNDM-1) or a biofilm-regulation gene (e.g., involved in quorum sensing). - Complexation: Incubate the pre-assembled Cas9-gRNA ribonucleoprotein (RNP) complex with the cationic liposomes to form liposome-RNP complexes via electrostatic interaction. Purify using size-exclusion chromatography.
2. Biofilm Cultivation and Treatment:
- Biofilm Formation: Grow a 48-hour mature biofilm of the target pathogen (e.g., Pseudomonas aeruginosa) in a flow cell system or a 96-well peg lid to establish a robust EPS matrix.
- Treatment Application: Apply the liposome-RNP complexes to the established biofilms in the presence of a sub-inhibitory concentration of a relevant antibiotic to exploit re-sensitization. Include controls: untreated biofilm, liposome-only, and RNP-only.
- Incubation: Incubate the treatment for 4-24 hours under conditions optimal for bacterial growth to allow for cellular uptake and CRISPR activity.
3. Efficacy and Analysis Endpoints:
- Biomass Quantification: Use Crystal Violet staining to measure total biofilm biomass. Compare absorbance of treated vs. control wells to calculate percentage reduction.
- Bacterial Viability (CFU Count): Dislodge biofilms by sonication, serially dilute, and plate on agar. Count Colony Forming Units (CFUs) after 24 hours to determine bactericidal effect.
- Gene Editing Efficiency: Extract genomic DNA from treated biofilms. Use T7 Endonuclease I assay or targeted deep sequencing to quantify the frequency of indels at the target locus.
- Confocal Microscopy Imaging: Use LIVE/DEAD BacLight staining and Confocal Laser Scanning Microscopy (CLSM) to visually assess biofilm architecture and the ratio of live-to-dead bacteria in a 3D context.
The Scientist's Toolkit: Essential Research Reagents
Successful execution of delivery system development and antimicrobial assays requires a suite of specialized reagents and materials. The following table lists key solutions for researchers in this field.
Table 3: Essential Research Reagents for CRISPR Delivery and Antimicrobial Testing
| Reagent / Material | Function / Application | Technical Notes |
|---|---|---|
| Cationic Lipids (e.g., DOTAP, DLin-MC3-DMA) | Form the core of LNPs, complexing with negatively charged genetic cargo via electrostatic interaction. | Critical for encapsulation efficiency and cellular uptake; impacts cytotoxicity [26] [25]. |
| Ionizable Lipids | Enable endosomal escape of LNP cargo by undergoing charge shift in acidic endosomal environment. | pKa is a key design parameter; essential for releasing functional CRISPR machinery into the cytoplasm [26]. |
| Polyethylene Glycol (PEG)-Lipids | Stabilize nanoparticles, reduce aggregation, and modulate pharmacokinetics and biodistribution. | Can induce anti-PEG antibodies, potentially affecting re-dosing [26] [28]. |
| AAV Serotypes (e.g., AAV9, AAVrh74) | Provide specific tissue tropism (e.g., liver, CNS, muscle) for targeted in vivo delivery. | Selected based on target organ; pre-existing neutralizing antibodies in populations can exclude patients [26] [27]. |
| Compact Cas Orthologs (e.g., SaCas9, CjCas9) | Enable packaging of Cas nuclease, gRNA, and regulatory elements within a single AAV vector. | Necessitated by the strict ~4.7 kb packaging limit of AAV [27]. |
| Guide RNA (gRNA) / sgRNA | Confers sequence specificity by guiding the Cas nuclease to the target genomic locus. | Design is critical for on-target efficiency and minimizing off-target effects [25] [27]. |
| Extracellular Polymeric Substance (EPS) Matrix | The protective barrier of biofilms; target for nanoparticle penetration. | Composed of polysaccharides, proteins, and eDNA; its disruption is a primary mechanism of nano-antibacterials [25]. |
| Crystal Violet Stain | A standard dye for quantifying total biofilm biomass in high-throughput assays. | Provides a colorimetric readout (absorbance) correlated with adhered biofilm [25]. |
Decision Framework and Concluding Perspectives
The choice between AAV, lentiviral, and LNP delivery systems is not one of absolute superiority but of strategic alignment with the therapeutic goal. The following diagram synthesizes the key decision criteria for selecting a CRISPR delivery platform for antimicrobial applications.
The field is rapidly evolving beyond these conventional platforms. Emerging "hybrid" systems, such as Lipid Nanoparticle Spherical Nucleic Acids (LNP-SNAs), demonstrate how structural innovation can enhance performance. LNP-SNAs, which wrap a CRISPR-loaded LNP in a dense shell of DNA, have been shown to triple cellular uptake and editing efficiency by facilitating better receptor engagement and endosomal escape [29]. Similarly, virus-like particles (VLPs) and exosomes are being explored for their potential to combine the efficiency of viral entry with the safety and re-dosability of non-viral systems [26].
For antimicrobial research, the synergy between nanoparticle penetration and CRISPR precision is a powerful paradigm. The future of CRISPR delivery lies not in a single dominant technology, but in a growing toolkit of agnostic solutions, enabling researchers to select and engineer the optimal vector for each specific biological challenge and therapeutic context.
The escalating crisis of antimicrobial resistance (AMR) and surface-mediated pathogen transmission has intensified the demand for advanced active coatings. Projections indicate that AMR may cause up to 1.91 million direct deaths annually by 2050, highlighting the critical need for innovative solutions in infection prevention [21]. Within this context, the formulation of durable antimicrobial coatings represents a pivotal frontier in material science and public health. These coatings, particularly those integrating active agents like triclosan, provide continuous protection against microbial colonization on high-touch surfaces, medical devices, and in healthcare environments [24]. The performance of these coatings is profoundly influenced by their formulation methods, with electrostatic application emerging as a superior technique for achieving uniform, adherent, and durable films compared to conventional dipping processes [30]. Simultaneously, revolutionary approaches are being pioneered, notably the integration of nanoparticle-enhanced CRISPR technology, which offers genetically precise antimicrobial activity against resistant pathogens [25]. This comparison guide objectively evaluates the performance of conventional antimicrobial coatings against emerging nanoparticle-CRISPR hybrid systems, providing researchers and drug development professionals with experimental data, methodological protocols, and a critical analysis of both technological pathways.
Experimental Protocols for Coating Development and Testing
Electrostatic Spray Coating Methodology
The electrostatic application process creates more homogeneous and thinner coatings compared to conventional methods. The following protocol, adapted from fruit coating studies with relevance to antimicrobial surface application, ensures optimal coating properties [30]:
- Surface Preparation: Substrates (e.g., stainless steel plates) must be thoroughly cleaned to remove contaminants. Sterilize with ethanol-soaked gauze and air-dry completely before coating application.
- Coating Formulation: Prepare coating solution with appropriate viscosity (0.49–0.99 kg/cm³) and conductivity (2.27–2.96 mS/cm) for electrostatic application. Incorporation of active agents such as triclosan requires uniform dispersion within the matrix.
- Application Process: Utilize electrostatic spraying equipment with controlled parameters. Apply primer layer to improve adhesion and corrosion resistance. For the active coating layer, use a twin-nozzle electrostatic spray system with standardized coating thickness.
- Curing Process: Cure coated substrates in an oven to harden the antimicrobial layer, ensuring durability and adherence. Cool to room temperature before testing or use.
- Quality Control: Verify coating uniformity through SEM analysis and measure charge-to-mass ratio (target: 1.67–1.71 mC/kg) to ensure proper electrostatic deposition [30].
Antibacterial Efficacy Testing Protocol
Standardized testing is essential for evaluating coating performance. The JIS Z 2801:2010(E) protocol provides a validated methodology [24]:
- Test Preparation: Cut coated materials into 60 mm × 60 mm squares. Sterilize surfaces with ethanol before testing.
- Inoculum Preparation: Suspend test pathogens (e.g., Staphylococcus aureus, Escherichia coli, MRSA) in sterile deionized water. Standardize suspension to McFarland No.1 standard (approximately 3 × 10⁸ CFU/mL) and dilute with nutrient broth to achieve final concentration of 2.5–10 × 10⁵ CFU/mL.
- Inoculation and Incubation: Apply 0.4 mL bacterial solution to test surface, cover with parafilm to ensure even spread, and incubate at 35 ± 1°C with relative humidity ≥90% for 24 hours.
- Bacterial Recovery and Enumeration: After incubation, wash surfaces with 10 mL SCDLP broth. Use swabs to collect residual bacteria. Plate serial dilutions on blood agar plates and incubate at 35 ± 1°C for 48 hours. Count resulting colonies.
- Calculation of Antibacterial Efficacy: Determine bacterial reduction rate (R) using the formula: R = log(A/B), where A is the average viable bacteria recovered from control surfaces and B is the average from antibacterial-coated surfaces. An R value >2 indicates significant antibacterial effect [24].
Durability Testing Protocols
- UV Exposure Resistance: Expose coated substrates to UV light for 500 hours before retesting antibacterial efficacy to evaluate photostability [24].
- Chemical Cleaner Resistance: Rub coated surfaces with 0.06% sodium hypochlorite solution four times daily for two weeks before retesting to simulate cleaning regimen impacts [24].
Performance Comparison: Conventional Antimicrobial vs. Nanoparticle-CRISPR Systems
Table 1: Comprehensive performance comparison of antimicrobial coating technologies
| Performance Parameter | Triclosan-Based Coatings | Nanoparticle-Enhanced CRISPR Systems | Testing Standards/Methods |
|---|---|---|---|
| Antimicrobial Efficacy | R value >2 against S. aureus, E. coli, A. baumannii, MRSA, P. aeruginosa [24] | Up to 90% reduction in P. aeruginosa biofilm biomass; 3.5× higher editing efficiency [25] | JIS Z 2801:2010(E); Biofilm assays |
| Durability | Maintains efficacy after prolonged bleach cleaning (0.06% NaOCl, 4× daily, 2 weeks) [24] | Controlled-release systems maintain activity; long-term stability under evaluation [25] | Accelerated aging; repeated cleaning cycles |
| UV Stability | Efficacy may be affected by prolonged UV exposure (500 hours) [24] | Nanoparticle protection of CRISPR components; potential UV degradation concerns | 500-hour UV exposure test [24] |
| Application Method | Electrostatic spray coating for uniform, thin films [30] | Liposomal nanoparticles; spherical nucleic acids (LNPs) for cellular delivery [29] | Electrostatic coating; nanoparticle synthesis |
| Mechanism of Action | Inhibition of enoyl-acyl carrier protein reductase (ENR) disrupting fatty acid synthesis [24] | Targeted disruption of antibiotic resistance genes, quorum sensing pathways, and biofilm regulators [25] | Enzymatic assays; genetic sequencing |
| Target Specificity | Broad-spectrum antibacterial activity | Precision targeting of specific genetic sequences | Genomic analysis; species-specific testing |
| Environmental Impact | Potential ecological concerns; persistent in waterways [31] | Biodegradable nanoparticle systems in development; unknown environmental persistence | Ecotoxicity studies; environmental fate modeling |
Table 2: Formulation and processing parameters across coating technologies
| Formulation Aspect | Conventional Triclosan Coatings | Nanoparticle-CRISPR Systems |
|---|---|---|
| Active Components | Triclosan-based additives in polymer matrix [24] | Cas9 nuclease, guide RNA, DNA repair templates [25] |
| Carrier System | Polymer matrix (e.g., surface passivation layers) [24] | Lipid nanoparticles, gold nanoparticles, spherical nucleic acids [25] [29] |
| Application Method | Electrostatic spray coating [30] | Liposomal encapsulation; nanoparticle functionalization [25] |
| Curing/Processing | Oven curing after application [24] | Self-assembly; surface modification |
| Coating Thickness | Thin, homogeneous layers (SEM-verified) [30] | Nanoscale structures (∼50 nm for LNPs) [29] |
| Charge-to-Mass Ratio | 1.67–1.71 mC/kg (optimized for ESC) [30] | Surface charge tuned for cellular uptake |
Mechanism of Action Analysis
Conventional Antimicrobial Coating Mechanisms
Triclosan-based coatings primarily function through inhibition of enoyl-acyl carrier protein reductase (ENR), a key enzyme in the bacterial fatty acid biosynthesis pathway. By binding to the active site of ENR, triclosan prevents proper enzymatic function, halting fatty acid biosynthesis and ultimately causing bacterial cell death [24]. The electrostatic application method enhances this activity by ensuring uniform distribution of active sites across the coated surface, creating a continuous protective interface.
Nanoparticle-Enhanced CRISPR Mechanism
Nanoparticle-enhanced CRISPR systems employ a fundamentally different, genetically precise approach. The CRISPR/Cas9 system is delivered to bacterial cells via engineered nanoparticles, where it specifically targets and disrupts antibiotic resistance genes, quorum sensing pathways, and biofilm formation regulators.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential research reagents and materials for coating development
| Reagent/Material | Function/Application | Examples/Specifications |
|---|---|---|
| Triclosan | Broad-spectrum antibacterial agent inhibits ENR enzyme | Purity ≥99%; used in 23EV and 28SC coating formulations [24] |
| Sodium Alginate | Biopolymer for hydrogel-based coatings and nanocomposites | Derived from brown algae; forms hydrogels with divalent cations [31] |
| Manganese Sulfide NPs | Nanoparticle additive enhancing adsorption and mechanical properties | 0.02–0.2 g incorporation in nanocomposite hydrogels [31] |
| Lipid Nanoparticles (LNPs) | Delivery vehicle for CRISPR-Cas9 components | Core-shell structure for genetic material protection [29] |
| Spherical Nucleic Acids (SNAs) | Advanced nanostructure for enhanced cellular delivery | ∼50 nm diameter; LNP-SNAs boost editing efficiency 3× [29] |
| Cas9 Nuclease | CRISPR-associated enzyme for targeted DNA cleavage | Requires guide RNA for sequence specificity [25] |
| Guide RNA (gRNA) | CRISPR targeting component directs Cas9 to specific genes | Custom-designed for antibiotic resistance genes [25] |
| Shellac | Commercial coating comparator in efficacy studies | Natural resin; reference material for performance testing [30] |
Discussion and Research Implications
The experimental data and performance comparisons reveal distinct advantages and limitations for both conventional and emerging coating technologies. Triclosan-based coatings applied via electrostatic methods offer proven durability and maintain efficacy against multiple pathogens even after aggressive cleaning regimens, demonstrating their practical utility in healthcare settings where surface disinfection is routine [24]. The electrostatic application process provides superior coating homogeneity and thickness control compared to conventional dipping methods, contributing to more consistent antimicrobial performance [30].
Conversely, nanoparticle-enhanced CRISPR systems represent a paradigm shift from broad-spectrum antimicrobial activity to genetically precise interventions. These systems address the critical challenge of biofilm-associated infections, which demonstrate up to 1000-fold greater tolerance to conventional antibiotics compared to planktonic cells [25]. The ability to specifically target and disrupt antibiotic resistance genes (e.g., blaKPC, mecA) and quorum-sensing pathways offers a strategic approach to combating multidrug-resistant pathogens that conventional coatings cannot address.
The integration of nanoparticles addresses a fundamental delivery challenge in CRISPR-based antimicrobials. Lipid nanoparticle spherical nucleic acids (LNP-SNAs) have demonstrated threefold improvements in editing efficiency and enhanced cellular uptake across various cell types, including skin cells, white blood cells, and human bone marrow stem cells [29]. This delivery advantage is crucial for practical applications where efficient targeting of bacterial populations is essential.
Future research directions should focus on optimizing the environmental compatibility of both technologies, particularly addressing the ecological concerns associated with triclosan persistence in waterways [31] and the potential long-term impacts of nanoparticle and CRISPR components. Additionally, combination approaches utilizing the broad-spectrum protection of conventional coatings with the precision targeting of CRISPR systems may offer synergistic benefits for high-risk environments. The development of stimuli-responsive coatings that activate antimicrobial activity only in the presence of pathogens represents another promising avenue for extending functional longevity while minimizing environmental impact [32].
This comparison guide demonstrates that both conventional triclosan-based coatings and emerging nanoparticle-CRISPR systems offer distinct advantages for antimicrobial surface protection. Electrostatic application of triclosan-based coatings provides immediate, practical solutions for healthcare environments, with proven efficacy against common pathogens and durability under cleaning protocols. Meanwhile, nanoparticle-enhanced CRISPR technology represents a transformative approach with genetically precise targeting capabilities particularly suited for combating multidrug-resistant biofilm formations. The selection between these technologies depends on specific application requirements, target pathogens, and environmental considerations. Continued research into both technological pathways, alongside exploration of hybrid approaches, will be essential for addressing the evolving challenge of antimicrobial resistance and surface-mediated infection transmission. As both fields advance, considerations of scalability, regulatory approval, and environmental impact will increasingly shape their translation from laboratory research to clinical and commercial applications.
Biofilms are structured communities of microorganisms embedded in a protective extracellular polymeric substance (EPS) matrix, which can exhibit up to 1000-fold greater tolerance to antibiotics compared to their planktonic counterparts [3]. This inherent resistance poses significant challenges in treating infections, particularly those associated with medical implants, where biofilm formation can lead to persistent infections and implant failure [33]. The complex architecture of biofilms, characterized by microcolonies interspersed with water channels, creates physical barriers that limit antimicrobial penetration while enabling bacterial survival in hostile environments [3] [18].
To address the critical need for effective anti-biofilm strategies, researchers are developing increasingly sophisticated assessment models that bridge the gap between traditional in vitro systems and complex in vivo environments. These models are essential for evaluating emerging technologies such as nanoparticle-enhanced CRISPR systems and conventional antimicrobial coatings [3] [34]. The ideal assessment platform must recapitulate key aspects of in vivo conditions, including the structural and physiological characteristics of tissue-associated biofilms, while providing reproducible results for reliable comparison of therapeutic efficacy [34]. This comparison guide examines current biofilm assessment methodologies, their applications in evaluating advanced antimicrobial technologies, and the experimental frameworks necessary for generating clinically relevant data.
Quantitative Comparison of Anti-Biofilm Technologies
The table below summarizes performance data for nanoparticle-enhanced CRISPR systems and conventional antimicrobial coatings against biofilm-forming pathogens, as measured by various in vitro and in vivo models.
Table 1: Comparative Performance of Anti-Biofilm Technologies
| Technology Category | Specific Agent/Formulation | Target Pathogen | Biofilm Reduction (%) | Bacterial Load Reduction (log₁₀ CFU) | Assessment Model | Key Findings |
|---|---|---|---|---|---|---|
| Nanoparticle-Enhanced CRISPR | Liposomal Cas9 formulations | Pseudomonas aeruginosa | >90% in vitro [3] | Not specified | In vitro biofilm model [3] | Disruption of antibiotic resistance genes and quorum sensing pathways |
| Nanoparticle-Enhanced CRISPR | Gold nanoparticle-CRISPR hybrids | Multiple MDR pathogens | Not specified | Not specified | In vitro model [3] | 3.5-fold increase in gene-editing efficiency compared to non-carrier systems |
| Conventional Antimicrobial Coatings | Silver-based antimicrobial coatings | Broad-spectrum (Gram+/Gram-) | 99.9% bacterial reduction within 24 hours [35] | Not specified | In vitro efficacy testing [35] | Sustained release properties; well-established safety profile |
| Conventional Antimicrobial Coatings | DSM Biomedical hydrophilic silver-ion coating | Catheter-associated pathogens | Not specified | Not specified | In vitro model [35] | 99.9% bacterial reduction within 24 hours; for urinary catheters |
| Advanced In Vitro Model | Modified Crone's Model (MCM) | Staphylococcus biofilms | Enables quantification [34] | Enables quantification [34] | Semi-solid, soft-tissue-like agar matrices [34] | Identified cis-2-decenoic acid and cis-11-methyl-2-dodecenoic acid as potent antibiotic potentiators |
| CRISPR-based Antimicrobials | CRISPR-Cas systems | Multiple MDR pathogens | >90% in vitro efficacy [9] | Not specified | In vitro model [9] | Selective elimination of MDR pathogens |
Experimental Models for Biofilm Assessment
In Vitro Models
In vitro biofilm models provide controlled, reproducible systems for initial screening of anti-biofilm technologies, though with varying degrees of physiological relevance.
Traditional Liquid-Culture Models including the Minimum Biofilm Eradication Concentration (MBEC) assay offer high-throughput screening capabilities but poorly reflect the structural and physiological characteristics of tissue-associated biofilms due to their homogeneous nature and lack of spatial organization [34]. These models typically cultivate biofilms on plastic or glass surfaces under nutrient-rich conditions that don't mimic host environments.
Advanced 3D In Vitro Models have emerged to bridge the gap between simple 2D models and complex in vivo systems. The Modified Crone's Model (MCM) represents a significant advancement by embedding bacteria in soft-tissue-like agar-based matrices that better recapitulate the spatial and diffusional constraints of biofilm-associated infections [34]. This semi-solid model demonstrates more consistent growth, in vivo-like morphology, and reduced variability compared to liquid-culture systems, with the additional advantage of identifying anti-biofilm compounds that would be undetectable in traditional assays [34].
Organotypic 3D Models for implant-associated infections incorporate relevant human cell types—such as fibroblasts, keratinocytes, stem cells, and immune cells—alongside pathogenic bacteria and implant materials to create more physiologically relevant test environments [33]. These models enable investigation of host-pathogen-implant interactions at cellular and molecular levels, providing insights beyond simple antimicrobial efficacy, including host tissue response and immune activation [33].
In Vivo Models
While in vitro models provide valuable preliminary data, in vivo models remain essential for evaluating anti-biofilm technologies in biologically complex environments. Animal models, particularly for orthopedic and dental implant infections, allow assessment of biofilm eradication and bacterial load reduction in the context of a functioning immune system and physiological tissue environment [33]. These models typically involve surgical implantation of infected or coated devices into relevant anatomical sites, followed by monitoring of infection progression and treatment efficacy through imaging, tissue culture, and histological analysis.
The ethical considerations and technical complexity of in vivo models have driven development of the 3R Principle (Replacement, Reduction, and Refinement), encouraging researchers to maximize information gained from advanced in vitro systems before proceeding to animal studies [33]. This approach not only addresses ethical concerns but also improves research efficiency by refining scientific questions at earlier stages of investigation.
Experimental Protocols for Biofilm Assessment
Protocol for Modified Crone's Model (MCM) Biofilm Assessment
The MCM provides a standardized approach for evaluating anti-biofilm efficacy under conditions that better mimic tissue environments than traditional liquid assays [34].
Table 2: Key Research Reagents for Biofilm Assessment
| Research Reagent | Function/Application | Example Specifications |
|---|---|---|
| Soft-tissue-like agar matrices | Provides semi-solid growth environment mimicking tissue conditions | Agar concentration typically 0.5-1.5% for optimal biofilm formation [34] |
| Culture media | Supports bacterial growth and biofilm development | Tryptic soy broth, Mueller Hinton broth, or pathogen-specific media |
| Antibiotic potentiators | Enhances efficacy of conventional antibiotics | cis-2-decenoic acid, cis-11-methyl-2-dodecenoic acid identified via MCM screening [34] |
| CRISPR-Cas9 components | Targeted disruption of resistance genes | Cas9 nuclease, guide RNA (gRNA) targeting specific bacterial genes [3] |
| Nanoparticle delivery systems | Enhances cellular uptake and stability of antimicrobials | Liposomal formulations, gold nanoparticles (3.5× editing efficiency) [3] |
| Viability stains | Differentiates live/dead bacteria in biofilms | SYTO 9/propidium iodide for confocal microscopy analysis |
| Microtiter plates | High-throughput biofilm cultivation | 96-well plates with non-treated surfaces for improved biofilm attachment |
Methodology:
- Matrix Preparation: Prepare semi-solid agar-based matrices at concentrations that mimic soft tissue consistency (typically 0.5-1.5% agar) in appropriate culture media [34].
- Bacterial Embedding: Incorporate standardized bacterial suspensions (e.g., 10⁶ CFU/mL) directly into the molten agar matrix before solidification.
- Biofilm Development: Incubate embedded cultures under conditions optimal for the target pathogen (typically 24-72 hours at 37°C) to allow mature biofilm formation.
- Treatment Application: Apply test compounds (nanoparticle-enhanced CRISPR systems, antimicrobial coatings, or conventional antibiotics) to the biofilm-containing matrix surface.
- Incubation and Diffusion: Allow appropriate incubation time for compound diffusion through the matrix (typically 24 hours) under conditions relevant to the intended application.
- Biofilm Disruption and Quantification: Recover embedded biofilms through matrix disruption (e.g., enzymatic digestion or mechanical homogenization), followed by serial dilution and plating for CFU enumeration.
- Data Analysis: Calculate percentage biofilm reduction or log₁₀ CFU reduction compared to untreated controls.
Protocol for Assessing Nanoparticle-Enhanced CRISPR Systems
This protocol evaluates the efficacy of CRISPR-based antimicrobials delivered via nanoparticle carriers against biofilm-forming pathogens [3].
Methodology:
- gRNA Design: Design guide RNAs targeting essential antibiotic resistance genes (e.g., bla, mecA, ndm-1), quorum sensing pathways, or biofilm-regulating factors in the target pathogen [3].
- Nanoparticle Formulation: Complex CRISPR/Cas9 components (Cas9 nuclease + specific gRNA) with nanoparticle carriers such as:
- Biofilm Cultivation: Grow mature biofilms of target pathogens on relevant surfaces (e.g., plastic, glass, or implant materials) for 24-72 hours.
- Treatment Application: Apply nanoparticle-CRISPR formulations to pre-established biofilms at varying concentrations.
- Co-incubation: Allow 24-48 hours for nanoparticle penetration, cellular uptake, and CRISPR-mediated genetic disruption.
- Efficacy Assessment:
- Quantify biofilm biomass through crystal violet staining or similar methods
- Assess bacterial viability through CFU enumeration
- Evaluate specific gene disruption through molecular methods (PCR, sequencing)
- Visualize biofilm architecture disruption via microscopy (CLSM, SEM)
- Synergy Testing: Evaluate combination approaches with conventional antibiotics to identify potential synergistic effects [3].
Protocol for Evaluating Antimicrobial Coating Efficacy
This protocol assesses the anti-biofilm performance of coated medical devices or surfaces [35].
Methodology:
- Surface Coating: Apply antimicrobial coatings to relevant substrate materials using appropriate methods:
- Surface modification (plasma treatment, covalent bonding)
- Dip coating
- Spray coating
- Vapor deposition [35]
- Coating Characterization: Verify coating uniformity, thickness, and active agent concentration through appropriate analytical methods.
- Biofilm Formation on Coated Surfaces: Inoculate coated surfaces with bacterial suspensions (e.g., 10⁶ CFU/mL of relevant pathogens) and incubate under conditions promoting biofilm growth (typically 24-72 hours at 37°C with appropriate humidity).
- Biofilm Quantification:
- Direct CFU enumeration after biofilm disruption (sonication, scraping)
- Metabolic activity assays (resazurin, XTT)
- Microscopic analysis of biofilm structure and viability
- Durability Testing: Subject coated surfaces to simulated use conditions (mechanical abrasion, liquid immersion, repeated cleaning) before biofilm challenge to assess coating longevity.
- Control Inclusion: Include uncoated surfaces and relevant reference coatings (e.g., silver-based coatings) as controls for comparative assessment.
Mechanisms of Action and Assessment Workflows
The diagram below illustrates the conceptual workflow for comparing nanoparticle-enhanced CRISPR systems with conventional antimicrobial coatings using relevant in vitro and in vivo assessment models.
Diagram 1: Anti-Biofilm Technology Assessment Workflow
The diagram above illustrates the conceptual framework for evaluating and comparing anti-biofilm technologies. Nanoparticle-enhanced CRISPR systems and conventional antimicrobial coatings are assessed through complementary in vitro and in vivo models, with efficacy quantified through standardized metrics including biofilm reduction percentage and bacterial load decrease. Promising candidates from in vitro screening advance to more complex in vivo models for validation.
The molecular mechanisms of these technologies differ significantly, as illustrated in the following diagram detailing their pathways for combating biofilm-associated infections:
Diagram 2: Mechanisms of Anti-Biofilm Action
The mechanisms of action differ fundamentally between these technologies. Nanoparticle-enhanced CRISPR systems employ precision genetic approaches, utilizing nanoparticle carriers to deliver CRISPR components that target and disrupt specific bacterial resistance genes, quorum sensing pathways, or biofilm-regulating factors [3]. This genetic disruption resensitizes bacteria to conventional antibiotics and impedes biofilm maturation. In contrast, conventional antimicrobial coatings primarily function through physicochemical mechanisms including contact-mediated killing, sustained release of antimicrobial ions (e.g., silver), reactive oxygen species (ROS) generation, and physical barrier creation that prevents initial bacterial attachment [35] [11].
The assessment of biofilm eradication and bacterial load reduction requires sophisticated models that balance physiological relevance with experimental practicality. Advanced in vitro systems like the Modified Crone's Model offer significant advantages over traditional liquid-culture assays by better mimicking tissue environments and generating more clinically predictive data [34]. When comparing emerging nanoparticle-enhanced CRISPR systems with conventional antimicrobial coatings, researchers must consider both the fundamentally different mechanisms of action and the appropriate assessment frameworks for each technology.
Nanoparticle-enhanced CRISPR represents a precision medicine approach with demonstrated efficacy in disrupting specific resistance mechanisms and resensitizing biofilms to conventional antibiotics [3]. However, challenges remain in delivery optimization and minimizing off-target effects. Conventional antimicrobial coatings provide broad-spectrum protection with well-established manufacturing processes and regulatory pathways, though they face limitations in long-term durability and potential for resistance development [35]. The selection of appropriate assessment models—from simple in vitro screening to complex 3D organotypic systems and ultimately in vivo validation—should align with the specific mechanism of action and intended application of each anti-biofilm technology.
As both fields advance, the integration of standardized assessment protocols will enable more direct comparison between these divergent approaches, ultimately accelerating the development of effective strategies to combat biofilm-associated infections, particularly in medical device and implant applications where biofilms pose significant clinical challenges [33] [18].
Antimicrobial resistance (AMR) represents one of the most severe threats to global public health in the 21st century, with projections indicating it could cause 10 million deaths annually by 2050 if left unaddressed [36]. The escalating crisis stems from the ability of microorganisms to survive exposure to antimicrobial agents through various mechanisms, including target site modification, enzymatic degradation, and horizontal gene transfer via plasmids and transposons [36]. Particularly alarming is the rising resistance to last-resort antibiotics (e.g., colistin, carbapenems) in pathogens such as Klebsiella pneumoniae and Acinetobacter baumannii, where treatment failure rates exceed 50% in some regions [36]. This dire situation has catalyzed the development of novel therapeutic strategies beyond conventional antibiotics, with two particularly promising approaches emerging: antimicrobial coatings for medical devices and preclinical CRISPR-based antimicrobial therapies. Both approaches represent paradigm shifts in how we combat resistant infections, yet they operate at fundamentally different levels—one functioning as a preventive barrier on surfaces, the other as a precision genetic weapon against resistance mechanisms themselves.
The following comparison guide objectively evaluates these two innovative approaches within the context of a broader thesis on nanoparticle-enhanced CRISPR versus conventional antimicrobial coating performance. This analysis synthesizes current applications, experimental data, methodologies, and translational potential for researchers, scientists, and drug development professionals working at the forefront of AMR research.
Medical Device Coatings: A Preventive Barrier
Antimicrobial coatings for medical devices are specialized surface treatments designed to inhibit microbial growth on medical equipment through the integration of biocidal agents like silver, copper, or organic compounds into polymer matrices or metallic layers [35]. These coatings create hostile environments for bacteria, fungi, and viruses on device surfaces, playing a critical role in preventing healthcare-associated infections (HAIs)—which affect over 1.7 million patients annually in the U.S. alone [35]. The global antimicrobial coatings market for medical devices demonstrates significant traction and commercial adoption, valued at USD 3.8 billion in 2024 and projected to reach USD 7.2 billion by 2032, exhibiting a compound annual growth rate (CAGR) of 7.3% [35].
Table 1: Antimicrobial Coating Market Analysis by Segment
| Segment Category | Leading Sub-Segment | Market Share & Growth Insights |
|---|---|---|
| By Type | Silver Antimicrobial Coatings | Dominant segment due to broad-spectrum efficacy and established safety profile [35] |
| By Application | Orthopedic Implants | Leading application segment; driven by need to prevent periprosthetic joint infections [35] |
| By Coating Technology | Surface Modification | Rapidly evolving segment offering permanent, integrated antimicrobial layers [35] |
| By Region | North America | Established market leader with robust regulatory framework and high healthcare expenditure [35] |
Key Mechanisms of Action and Experimental Efficacy
Antimicrobial coatings function primarily through continuous or triggered release of biocidal agents that disrupt microbial cellular processes. Silver-based coatings, the most prevalent type, release silver ions that damage bacterial cell membranes, disrupt metabolic enzymes, and interfere with DNA replication [35]. Advanced formulations now incorporate nano-engineered silver to enhance surface area and controlled release kinetics. The experimental efficacy of these coatings is typically validated through standardized in vitro testing protocols. For instance, DSM Biomedical's novel hydrophilic silver-ion coating for urinary catheters demonstrates 99.9% bacterial reduction within 24 hours in laboratory conditions [35]. Similarly, next-generation polymer-based formulations resist biofilm formation by creating non-adhesive surfaces that prevent initial bacterial attachment and subsequent biofilm maturation.
However, real-world performance often presents challenges compared to laboratory results. A 2023 meta-analysis of antimicrobial-coated catheters showed only 12-15% reduction in infection rates compared to uncoated alternatives in clinical settings, highlighting the translation gap between controlled testing environments and complex physiological conditions [35]. Durability limitations further constrain long-term efficacy, with coatings typically losing effectiveness after 6-12 months in vivo for implantable devices [35].
Standardized Testing Methodologies
Protocol 1: ISO 22196 / JIS Z 2801 Standard Antibacterial Activity Assessment
- Objective: Quantify antibacterial efficacy on plastic and other non-porous surfaces
- Methodology:
- Prepare test and control specimens (approximately 50mm x 50mm)
- Inoculate surfaces with bacterial suspension (×10^5 CFU/mL) of Staphylococcus aureus or Escherichia coli
- Cover with sterile polyethylene film to maintain contact
- Incubate at 35°C and 90% relative humidity for 24 hours
- Elute and plate serial dilutions on nutrient agar
- Count colonies after 48 hours and calculate antibacterial activity value (R)
- Key Metric: R = log (Ct/C0) where Ct = control colonies, C0 = test colonies
Protocol 2: ASTM E2180 Standard Test Method for Zone of Inhibition
- Objective: Evaluate diffusible antimicrobial agents in polymeric or hydrophobic materials
- Methodology:
- Incorporate test material into agar slurry with ×10^6 CFU/mL of test organism
- Solidify agar and incubate at 37°C for 18-24 hours
- Measure clear zones around material specimens indicating growth inhibition
- Compare zone diameters to control materials
- Applications: Particularly suitable for coated sutures, wound dressings, and soft medical devices
Preclinical CRISPR-based Antimicrobial Therapies: A Precision Genetic Approach
CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats) -Cas systems represent a revolutionary approach to combating AMR by leveraging bacterial adaptive immune mechanisms for precise genetic editing [37] [38]. Unlike broad-spectrum antibiotics or surface coatings, CRISPR-based antimicrobials can be programmed to selectively eliminate resistant bacteria or reverse resistance mechanisms by targeting specific genetic sequences [38]. The technology offers two primary strategies against AMR: (1) targeted killing of resistant bacteria through directed cleavage of essential genes or resistance determinants, and (2) genetic inactivation of resistance genes to resensitize bacteria to conventional antibiotics [38]. The field remains predominantly in preclinical development, with therapeutic applications rapidly advancing through animal models and toward human trials.
The CRISPR-Cas system operates through a coordinated process of target recognition and cleavage guided by RNA molecules. Class 2 systems, particularly those employing Cas9, Cas12, Cas13, and Cas14 effectors, have become preferred tools for antimicrobial applications due to their single-protein effector complexity and high programmability [37]. These systems can be delivered via engineered bacteriophages, conjugative plasmids, outer membrane vesicles, or synthetic nanoparticles to target specific bacterial populations within complex environments [38].
Key Mechanisms of Action and Experimental Efficacy
CRISPR-based antimicrobials achieve precision targeting through several demonstrated mechanisms. In strain-specific killing approaches, CRISPR-Cas systems programmed to target essential bacterial genes or antibiotic resistance loci selectively eliminate resistant pathogens from mixed populations while preserving commensal bacteria [38]. For example, conjugative plasmid-based delivery of CRISPR-Cas9 has successfully eradicated resistant Escherichia coli populations in microbial communities without disrupting surrounding microbiota [38]. In resistance reversal approaches, CRISPR systems precisely disrupt antibiotic resistance genes (e.g., β-lactamases, carbapenemases), restoring bacterial susceptibility to conventional antibiotics. Experimental models demonstrate that base editing via non-replicative phages can inactivate β-lactamase genes, effectively resensitizing bacteria to β-lactam antibiotics [38].
The integration of CRISPR with nanoparticle delivery systems has demonstrated remarkable efficacy in preclinical models. Liposomal Cas9 formulations have achieved over 90% reduction in Pseudomonas aeruginosa biofilm biomass in vitro, while CRISPR-gold nanoparticle hybrids enhanced gene-editing efficiency by 3.5-fold compared to non-carrier systems [25]. These hybrid platforms enable co-delivery of CRISPR components with antibiotics, producing synergistic antibacterial effects and superior biofilm disruption compared to monotherapies [25].
Table 2: Preclinical Efficacy of CRISPR-Based Antimicrobial Approaches
| Application Strategy | Delivery System | Target Pathogen/Gene | Experimental Efficacy |
|---|---|---|---|
| Strain-specific killing | Conjugative plasmids | β-lactamase genes in E. coli | Selective elimination of resistant strains; commensal preservation [38] |
| Therapeutic phage delivery | Engineered lytic phages | Plasmid-borne resistance genes | Significant pathogen clearance in animal models [38] |
| Biofilm disruption | Liposomal nanoparticles | Pseudomonas aeruginosa | >90% reduction in biofilm biomass [25] |
| Gene editing enhancement | Gold nanoparticles | Multiple AMR genes | 3.5-fold increase in editing efficiency [25] |
Standardized Testing Methodologies
Protocol 1: Bacteriophage-Mediated CRISPR Delivery Assessment
- Objective: Evaluate efficacy of phage-delivered CRISPR-Cas systems against AMR pathogens
- Methodology:
- Engineer bacteriophage vectors to encode CRISPR-Cas9 system with gRNAs targeting specific resistance genes
- Infect log-phase bacterial cultures (×10^8 CFU/mL) with engineered phage at multiplicity of infection (MOI) of 0.1-10
- Incubate with aeration at 37°C for 4-24 hours
- Plate serial dilutions on selective and non-selective media
- Count surviving colonies and calculate bacterial reduction
- Assess resistance gene modification via PCR and sequencing
- Validation: Include control phages without CRISPR payload and measure collateral effects on non-target bacteria
Protocol 2: CRISPR-Nanoparticle Biofilm Penetration Assay
- Objective: Quantify biofilm disruption efficacy of nanoparticle-encapsulated CRISPR systems
- Methodology:
- Establish 48-72 hour biofilms in flow cells or 96-well plates using standardized biofilm formers (e.g., P. aeruginosa, S. aureus)
- Treat with CRISPR-nanoparticle formulations (0.1-100μg/mL) with appropriate controls
- Incubate for 6-48 hours under physiologically relevant conditions
- Assess biofilm viability using ATP-based assays or resazurin reduction
- Quantify biofilm biomass via crystal violet staining or confocal microscopy
- Measure CRISPR payload delivery using fluorescent reporter systems
- Evaluate synergy with co-delivered antibiotics through checkerboard assays
- Advanced Imaging: Utilize confocal laser scanning microscopy (CLSM) with live/dead staining to visualize spatial distribution of killing within biofilm architecture
Comparative Analysis: Performance Metrics and Applications
Direct Performance Comparison
Table 3: Medical Device Coatings vs. CRISPR-based Antimicrobials - Performance Metrics
| Performance Parameter | Medical Device Coatings | CRISPR-based Antimicrobials |
|---|---|---|
| Technology Readiness Level | Commercial/Clinical (TRL 8-9) [35] | Preclinical/Experimental (TRL 3-4) [25] [38] |
| Primary Mechanism | Surface modification & chemical biocides [35] | Genetic editing & selective targeting [37] [38] |
| Speed of Action | Minutes to hours [35] | Hours to days (requires bacterial replication) [38] |
| Specificity | Broad-spectrum activity [35] | Sequence-specific targeting [38] |
| Efficacy in Models | 99.9% reduction in vitro; 12-15% infection reduction clinically [35] | >90% biofilm reduction; selective pathogen elimination in vivo [25] [38] |
| Durability | 6-12 months for implants [35] | Potentially permanent genetic modification [38] |
| Resistance Development | Documented (biofilm formation on coatings) [35] | Theoretically possible but not yet observed [38] |
| Key Limitations | Durability gaps, material compatibility, cost [35] | Delivery efficiency, off-target effects, regulatory hurdles [25] [37] |
Complementary Applications and Synergistic Potential
While medical device coatings and CRISPR-based antimicrobials operate through distinct mechanisms, they address complementary niches in AMR mitigation. Medical device coatings excel as preventive barriers on high-touch surfaces and implants where biofilm formation initiates infections [35]. Their broad-spectrum activity provides pragmatic protection in clinical environments despite specificity limitations. In contrast, CRISPR-based approaches offer therapeutic precision for established infections, particularly those involving multidrug-resistant pathogens where conventional antibiotics fail [38]. Their sequence-specific targeting preserves beneficial microbiota, addressing a critical limitation of broad-spectrum antibiotics.
The integration of both technologies presents particularly promising opportunities. Nanoparticle platforms originally developed for drug delivery can be adapted to co-deliver CRISPR components and conventional antimicrobials [25]. Similarly, surface coating technologies could potentially be engineered to release CRISPR-loaded nanocarriers in response to bacterial presence, creating "smart" antimicrobial surfaces that combine immediate biocidal activity with precise genetic targeting of resistant strains.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Research Reagents and Experimental Materials
| Reagent/Material | Function/Application | Specific Examples & Notes |
|---|---|---|
| Silver nanoparticles | Broad-spectrum antimicrobial coating agent | 20-100nm diameter; surface-functionalized for polymer integration [35] |
| Lipid Nanoparticles (LNPs) | CRISPR component delivery vehicle | Optimized for bacterial uptake; biofilm-penetrating formulations [25] |
| Engineered Bacteriophages | Targeted CRISPR delivery to specific bacteria | Modular platforms with swappable guide RNA cassettes [38] |
| Cas9 Nucleases | CRISPR system DNA cleavage component | High-fidelity variants for reduced off-target effects [37] [38] |
| Guide RNA (gRNA) | CRISPR system targeting specificity | Designed against essential genes or resistance determinants [38] |
| Polymer Coating Matrices | Sustained-release platform for biocidal agents | Medical-grade polyurethanes, silicones, or hydrogels [35] |
| Conjugative Plasmids | Horizontal transfer of CRISPR constructs | Broad-host-range plasmids for multi-species targeting [38] |
Visualizing Core Mechanisms and Workflows
The comparative analysis of medical device coatings and preclinical CRISPR-based antimicrobial therapies reveals two technologically distinct but potentially complementary approaches to addressing the AMR crisis. Medical device coatings represent a mature, commercially validated technology with demonstrated efficacy in reducing microbial burden on clinical surfaces, though limitations in durability and real-world infection prevention persist [35]. In contrast, CRISPR-based antimicrobials constitute an emerging, precision-based paradigm with transformative potential for treating resistant infections through genetic targeting, yet face significant translational challenges in delivery efficiency and regulatory approval [25] [38].
For researchers and drug development professionals, the strategic integration of both approaches offers promising pathways forward. Near-term applications will likely continue leveraging antimicrobial coatings for preventive surface protection while CRISPR technologies advance through preclinical development. The emerging convergence of both fields—through nanoparticle delivery systems that bridge surface functionality with genetic precision—represents a particularly innovative frontier. Future research priorities should address the key limitations of each technology: enhancing coating durability and clinical correlation for medical device coatings, and optimizing delivery efficiency and safety profiles for CRISPR-based approaches. As both fields evolve, their complementary strengths may ultimately provide a multi-layered defense strategy against antimicrobial resistance, combining immediate surface protection with precision therapeutic intervention for established infections.
Navigating Technical Hurdles and Enhancing Performance
The Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)-Cas system represents a revolutionary genome editing technology with transformative potential for treating genetic disorders, infectious diseases, and cancer [27] [39]. However, the clinical application of CRISPR-based therapies faces significant challenges, particularly in efficiently delivering the CRISPR components to target cells while minimizing off-target effects and immune responses [40] [39]. The delivery vehicle must transport the Cas nuclease and guide RNA (gRNA) through multiple biological barriers, achieve efficient cellular uptake, facilitate endosomal escape, and ensure precise nuclear localization—all while maintaining safety and specificity [41] [42].
Conventional antimicrobial coatings, typically based on silver ions, copper nanoparticles, or other biocidal agents, operate through broad-spectrum antimicrobial mechanisms but lack genetic precision [4] [11]. In contrast, nanoparticle-enhanced CRISPR systems offer a targeted approach capable of selectively disrupting antibiotic resistance genes, biofilm formation pathways, or virulence factors in pathogenic bacteria [3] [37]. This comparison guide objectively evaluates the performance of emerging nanoparticle-enhanced CRISPR delivery systems against conventional antimicrobial approaches, with particular focus on their efficacy, specificity, and potential for clinical translation in an era of increasing antimicrobial resistance.
Comparative Performance Analysis: Quantitative Data
The table below summarizes key performance metrics for nanoparticle-enhanced CRISPR systems compared to conventional antimicrobial approaches and other delivery methods:
Table 1: Performance Comparison of Antimicrobial Strategies and CRISPR Delivery Systems
| Technology | Efficacy Metrics | Specificity/Safety Profile | Key Limitations |
|---|---|---|---|
| Liposomal CRISPR-Cas9 Formulations | >90% reduction in Pseudomonas aeruginosa biofilm biomass [3] | Targeted disruption of specific bacterial resistance genes; Reduced off-target effects with RNP delivery [3] [41] | Potential immune recognition; Endosomal trapping concerns [42] [29] |
| CRISPR-Gold Nanoparticle Hybrids | 3.5× increase in gene-editing efficiency vs. non-carrier systems [3] | Synergistic action with antibiotics; Precise targeting capability [3] | Complex synthesis; Long-term biocompatibility questions [42] |
| LNP-Spherical Nucleic Acids (LNP-SNAs) | 3× improved cellular entry vs. standard LNPs; >60% increase in precise DNA repairs [29] | Far less toxicity; Architecture recognized by most cell types [29] | Emerging technology; Limited in vivo validation [29] |
| Conventional Antimicrobial Coatings (Silver NPs) | Broad-spectrum efficacy against Gram-positive and Gram-negative bacteria [11] | Non-selective cytotoxicity; ROS generation affects both pathogens and host cells [4] [11] | Development of bacterial resistance with sublethal exposure; Environmental toxicity concerns [4] |
| rAAV Vectors for CRISPR Delivery | Successful transduction in retinal cells (>70% in RhoP23H/+ mice) [27] | Favorable safety profile; High tissue specificity; Low immune response [27] | Limited packaging capacity (<4.7 kb); Pre-existing immunity in population [40] [27] |
Table 2: Analysis of CRISPR Delivery Vehicle Characteristics
| Delivery Method | Cargo Type | Editing Efficiency | Immune Response | Best Application Context |
|---|---|---|---|---|
| Viral Vectors (AAV, Lentivirus) | DNA | High, sustained expression | Moderate to high; Neutralizing antibodies | In vivo delivery to specific tissues [40] [27] |
| Lipid Nanoparticles (LNPs) | mRNA, RNP | Moderate to high | Low to moderate | Clinical applications; COVID-19 vaccines established precedent [41] [42] |
| Polymeric Nanoparticles | DNA, RNP | Variable | Low | Controlled release applications [42] |
| Inorganic Nanoparticles (Gold, Silica) | Protein, RNP | High with proper functionalization | Low | Research settings; Enhanced biofilm penetration [3] [42] |
| Electroporation | DNA, RNP | High in susceptible cells | Tissue damage concern | Ex vivo applications (immune cells, stem cells) [40] |
Experimental Protocols: Methodologies for Assessing Delivery Efficacy
Protocol for Evaluating CRISPR-Nanoparticle Anti-Biofilm Efficacy
Background: This protocol outlines the methodology for assessing the efficacy of nanoparticle-delivered CRISPR systems against bacterial biofilms, as referenced in studies demonstrating over 90% reduction in Pseudomonas aeruginosa biofilm biomass [3].
Materials:
- Bacterial strains: Pseudomonas aeruginosa (PAO1 or clinical isolates)
- Culture media: Tryptic soy broth (TSB) or LB broth
- CRISPR-nanoparticle constructs: Liposomal Cas9-gRNA complexes targeting biofilm-specific genes (e.g., pelA, pslA, quorum-sensing genes)
- Control nanoparticles: Empty nanoparticles, non-targeting gRNA nanoparticles
- Assessment tools: Confocal laser scanning microscopy (CLSM), scanning electron microscopy (SEM), crystal violet staining
- 96-well polystyrene plates for biofilm formation
Procedure:
- Biofilm formation: Grow overnight bacterial cultures and dilute to 1×10^6 CFU/mL in fresh media. Add 200μL to 96-well plates. Incubate 24-48 hours at 37°C for mature biofilm development.
- Treatment application: Add CRISPR-nanoparticle complexes at predetermined concentrations (typically 10-100μg/mL nanoparticle concentration). Include appropriate controls.
- Incubation: Treat biofilms for 4-24 hours at 37°C with gentle shaking.
- Biofilm quantification:
- Crystal violet staining: Fix biofilms with methanol, stain with 0.1% crystal violet, solubilize with acetic acid, measure absorbance at 595nm.
- Viability assessment: Use LIVE/DEAD BacLight bacterial viability kits with CLSM imaging.
- Biomass analysis: Process samples for SEM to examine ultrastructural changes.
- Gene editing efficiency: Extract genomic DNA from treated biofilms, amplify target regions, and sequence to verify specific gene modifications.
Validation Metrics: Successful experiments demonstrate significant reduction in biofilm biomass (>70%) with minimal effect on planktonic cells, confirming targeted approach rather than general biocidal activity [3].
Protocol for Assessing Off-Target Effects in CRISPR Delivery
Background: This protocol details methods for detecting and quantifying off-target effects of CRISPR systems delivered via nanoparticles, a critical safety consideration for therapeutic development [40] [39].
Materials:
- Target cells: Human cell lines relevant to intended application (e.g., HEK293, primary cells)
- CRISPR delivery systems: Nanoparticle formulations containing Cas9 protein and sgRNA
- Molecular biology reagents: PCR kits, sequencing primers, GUIDE-seq or CIRCLE-seq kits
- Bioinformatics tools: Cas-OFFinder, COSMID, or other off-target prediction algorithms
Procedure:
- Cell treatment: Deliver CRISPR-nanoparticle complexes to cells at optimized concentrations. Include controls (untreated cells, nanoparticles only).
- Genomic DNA extraction: Harvest cells 72-96 hours post-treatment using standard DNA extraction protocols.
- Off-target detection methods:
- GUIDE-seq: Introduce double-stranded oligodeoxynucleotides during transfection, followed by tag-specific PCR and next-generation sequencing.
- CIRCLE-seq: Digest genomic DNA, circularize fragments, digest with Cas9-sgRNA complex, then sequence cleaved fragments.
- Whole-genome sequencing: For comprehensive assessment, though more costly.
- Computational prediction: Use bioinformatics tools to identify potential off-target sites based on sequence similarity to the target site.
- Validation: Amplify putative off-target sites from genomic DNA and sequence to confirm editing events.
Validation Metrics: High-fidelity systems should demonstrate minimal to no detectable off-target effects at the top predicted sites, with significantly reduced off-target activity compared to standard CRISPR systems [39].
Mechanistic Insights: How Nanoparticle Platforms Address Delivery Challenges
The following diagrams illustrate key mechanisms by which advanced nanoparticle platforms overcome fundamental CRISPR delivery challenges:
LNP-SNA Architecture for Enhanced CRISPR Delivery
CRISPR-Nanoparticle Mechanisms Against Bacterial Biofilms
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents for Nanoparticle-Mediated CRISPR Delivery Studies
| Reagent/Material | Function | Specific Examples | Key Considerations |
|---|---|---|---|
| Cas9 Protein Variants | Genome editing nuclease | SpCas9, SaCas9, CjCas9, Cas12f [27] | Size constraints for viral vectors; PAM specificity; Editing efficiency |
| Guide RNA Design Tools | Target sequence specification | CRISPRscan, ChopChop, CRISPick | Off-target prediction; On-target efficiency algorithms |
| Lipid Nanoparticles | CRISPR component encapsulation | Ionizable LNPs, SORT-LNPs [42] [29] | Endosomal escape capability; Tissue targeting specificity; Cargo protection |
| Gold Nanoparticles | CRISPR RNP delivery | Spherical AuNPs, functionalized with targeting ligands [3] [42] | Surface chemistry for RNP binding; Biocompatibility; Optical properties for tracking |
| Polymeric Nanoparticles | Controlled release delivery | PLGA, chitosan, PEI-based nanoparticles [42] | Degradation kinetics; DNA binding capacity; Toxicity profile |
| Extracellular Vesicles | Natural delivery vehicles | Engineered exosomes, microvesicles [40] [42] | Biocompatibility; Homing capabilities; Cargo loading efficiency |
| Off-Target Assessment Kits | Safety validation | GUIDE-seq, CIRCLE-seq, DISCOVER-Seq [39] | Sensitivity for detection; Comprehensive genome coverage; Computational analysis requirements |
| Biofilm Assessment Tools | Efficacy measurement | Crystal violet staining, CLSM with LIVE/DEAD staining, SEM preparation [3] | Quantification methods; Distinction between bactericidal vs anti-biofilm effects |
The integration of nanoparticle delivery systems with CRISPR technology represents a promising frontier in precision antimicrobial therapy and genetic medicine. While conventional antimicrobial coatings provide broad-spectrum activity, nanoparticle-enhanced CRISPR systems offer unprecedented specificity in targeting antibiotic resistance mechanisms and virulence factors without affecting commensal microbiota [3] [37]. The quantitative data presented in this comparison guide demonstrates that advanced nanoparticle platforms like LNP-SNAs and CRISPR-gold nanoparticle hybrids can significantly improve editing efficiency while reducing off-target effects and immune responses [3] [29].
Future development should focus on optimizing nanoparticle design for enhanced tissue targeting, improving endosomal escape efficiency, and developing strategies to mitigate potential immune recognition. Additionally, standardized protocols for assessing off-target effects and long-term safety profiles will be crucial for clinical translation [42] [39]. As these technologies mature, nanoparticle-enhanced CRISPR systems have the potential to revolutionize our approach to antimicrobial resistance and genetic disorders, offering targeted solutions where conventional approaches face increasing limitations.
The therapeutic potential of CRISPR-based genetic medicines and conventional antimicrobial coatings is critically dependent on the delivery vehicle's efficiency. Nanoparticles, particularly lipid nanoparticles (LNPs), have emerged as a leading platform for this purpose, with their efficacy being profoundly influenced by their composition and structural organization [43]. Among the core components of LNPs, cholesterol plays a multifaceted role that extends beyond a simple structural filler; it is a critical modulator of nanoparticle stability, cellular uptake, and endosomal escape [43]. This guide provides a comparative analysis of how variations in nanoparticle composition, with a focused examination of cholesterol density, impact key performance metrics. By presenting synthesized experimental data and standardized protocols, we aim to equip researchers with the tools to optimize next-generation nanoparticle formulations for enhanced therapeutic outcomes, framed within the context of advancing nanoparticle-enhanced CRISPR technologies against conventional antimicrobial coatings.
Comparative Analysis of Nanoparticle Composition and Performance
The performance of nanoparticle-based delivery systems is a direct function of their physicochemical properties, which are dictated by their composition. The table below summarizes a comparative analysis of how different nanoparticle components influence critical quality attributes (CQAs) relevant to both drug delivery and antimicrobial coating applications.
Table 1: Impact of Nanoparticle Components on Critical Quality Attributes
| Component | Primary Function | Impact on Efficacy | Optimization Consideration |
|---|---|---|---|
| Ionizable Cationic Lipid | Encapsulation, cellular uptake, endosomal release [43] | High efficacy in nucleic acid delivery; key driver of editing efficiency. | Superior to permanently cationic lipids due to reduced toxicity and longer circulation time [43]. |
| Cholesterol | Structural integrity, membrane fluidity, and stability [43] | Optimal density crucial for packaging, bioavailability, and fusion with endosomal membrane. | Typically comprises 20-40 mol%; high density can reduce payload release [43]. |
| PEG-Lipid | Steric stabilization, reduces opsonization, increases circulation half-life [43] | Improves pharmacokinetics but can inhibit cellular uptake at high concentrations. | Molar ratio and PEG chain length are critical to balance stability vs. uptake efficiency. |
| Phospholipid | Contributes to LNP bilayer structure and stability [43] | Enhances particle stability and influences fusion kinetics. | Saturation level of lipid tails can affect bilayer rigidity and degradation rate. |
The Critical Role of Cholesterol Density
Cholesterol is a ubiquitous component of LNPs, constituting a substantial portion (often 20-40%) of the total lipid mass [43]. Its role is multifaceted:
- Structural Integrity: Cholesterol fills the voids between lipid chains, enhancing the mechanical stability and physical integrity of the nanoparticle, which is essential for surviving in the bloodstream [43].
- Modulation of Membrane Fluidity: It regulates the fluidity and permeability of the LNP bilayer, which in turn influences the packaging efficiency of the therapeutic payload and its subsequent release upon reaching the target cell [43].
- Facilitation of Endosomal Escape: A primary mechanism by which cholesterol enhances efficacy is through promoting fusion with the endosomal membrane. Following cellular uptake via endocytosis, LNPs are trapped in endosomes, which mature into lysosomes where the payload would be degraded. Cholesterol facilitates the destabilization of the endosomal membrane, enabling the LNP payload to escape into the cytoplasm—a critical step for functional delivery of CRISPR components or antimicrobial agents [43].
The density of cholesterol is not a passive variable but an active lever for optimization. Experimental data consistently show a Goldilocks effect:
- Low Cholesterol Density: Results in poorly formed, unstable nanoparticles with a high risk of premature payload leakage and rapid clearance in vivo.
- Excessively High Cholesterol Density: Can produce overly rigid, densely packed nanoparticles that are highly stable but fail to disassemble and efficiently release their therapeutic cargo within the cell, thereby reducing bioactive availability [43].
- Optimal Density: Achieves a balance, providing sufficient stability for transport while allowing for timely disintegration and payload release in the correct intracellular compartment.
Experimental Data on Formulation Efficacy
Optimizing cholesterol density and overall composition yields measurable improvements in formulation performance. The following table collates experimental data from key studies, highlighting the cause-and-effect relationship between formulation changes and functional outcomes.
Table 2: Experimental Data on Nanoparticle Formulation Efficacy
| Formulation Type | Key Compositional Change | Experimental Outcome | Implication for Efficacy |
|---|---|---|---|
| siRNA-LNP (ONPATTRO) | Use of novel ionizable lipid (MC3) with optimized cholesterol [43] | FDA approval for hereditary ATTR amyloidosis; reduced hepatic TTR protein translation in clinical trials (NCT01960348) [43]. | Validated LNP platform for systemic gene therapy; established benchmark for non-viral delivery. |
| CRISPR/Cas9 RNP-LNP | Co-encapsulation of Cas9 ribonucleoprotein (RNP) and sgRNA in cholesterol-stabilized LNPs [43] | >90% target gene knockout in hepatocytes in vivo; demonstrated therapeutic gene editing in genetic disease models. | Enables permanent genomic correction; high editing efficiency from a single dose. |
| Anti-biofilm CRISPR-LNP | Liposomal Cas9-sgRNA targeting bacterial resistance genes [25] | >90% reduction in P. aeruginosa biofilm biomass in vitro; resensitization to conventional antibiotics [25]. | Potentiates antibiotics; offers a strategy for treating persistent, device-associated infections. |
| CRISPR-Gold Hybrid | Cas9 RNP complexed with gold nanoparticles and donor DNA [25] | ~3.5x increase in gene-editing efficiency compared to non-carrier systems; HDR-mediated correction achieved [25]. | Demonstrates the potential of inorganic nanoparticles for enhanced delivery and precise editing. |
Essential Experimental Protocols for Optimization
Protocol 1: Formulation Optimization via Design of Experiments (DoE)
A systematic approach to optimizing multi-component nanoparticle formulations is critical for efficiently navigating the complex experimental space.
- Define Critical Process Parameters (CPPs) and CQAs: Identify the input variables (e.g., molar ratios of four lipid components, total flow rate, pH) and the output responses to be measured (e.g., particle size, polydispersity index (PDI), encapsulation efficiency, editing efficiency) [44].
- Select a DoE Design: For initial screening, a Fractional Factorial or Plackett-Burman design can identify the most influential CPPs from a large set. For subsequent optimization, a Response Surface Methodology (e.g., Central Composite Design) is used to model the relationship between the key CPPs and the CQAs and to find the optimal operating conditions [44].
- Execute and Analyze: Prepare formulations according to the experimental design. Analyze the CQAs for each run. Use statistical software to fit models to the data, generate contour plots, and predict the optimal formulation configuration that maximizes desired efficacy [44].
Protocol 2: In Vitro Biofilm Disruption Assay
This protocol evaluates the efficacy of antimicrobial nanoparticles (e.g., CRISPR-loaded LNPs) against biofilms.
- Biofilm Formation: Grow a standardized biofilm of a target bacterium (e.g., P. aeruginosa) in a 96-well plate or on a relevant surface for 24-48 hours.
- Treatment Application: Treat the mature biofilm with the nanoparticle formulation, a negative control (e.g., PBS), and a positive control (e.g., a known antibiotic). Incubate for a specified period (e.g., 4-24 hours) [25].
- Viability Assessment: Quantify remaining viable cells using a metabolic assay like resazurin (AlamarBlue) or by enumerating colony-forming units (CFUs) after disrupting the biofilm and serial dilution [25].
- Biomass Quantification: Stain the biofilm with crystal violet to assess total biomass, or use confocal laser scanning microscopy (CLSM) with live/dead stains to visualize biofilm architecture and cell viability [25].
Visualizing the Cholesterol Role and Experimental Workflow
Cholesterol-Mediated LNP Intracellular Delivery
DoE Formulation Optimization Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Nanoparticle CRISPR & Antimicrobial Research
| Reagent / Material | Function in Research | Specific Example / Note |
|---|---|---|
| Ionizable Cationic Lipids | Drive nucleic acid encapsulation and endosomal escape; key for efficacy. | DLin-MC3-DMA (MC3) is a benchmark lipid used in ONPATTRO [43]. |
| Cholesterol | Provides structural stability and modulates membrane fluidity for payload release. | A critical component requiring density optimization (often 20-40 mol%) [43]. |
| PEG-Lipids | Steric stabilization; control pharmacokinetics and cellular uptake profile. | PEG-DMG is commonly used; concentration and PEG chain length are key variables. |
| Microfluidic Mixer | Enables reproducible, scalable preparation of homogeneous LNPs. | Essential for transitioning from bench-scale batches to GMP production [44]. |
| CRISPR-Cas Ribonucleoprotein (RNP) | The functional gene-editing machinery; direct RNP delivery avoids genomic integration. | Co-encapsulation of Cas9 protein and sgRNA in LNPs for high efficiency editing [43]. |
| qPCR / ELISA Kits | Quantify biodistribution and pharmacokinetics of LNPs and their payload. | Critical for in vivo evaluation of delivery to target tissues. |
| Cell Viability & Metabolic Assays | Assess cytotoxicity of nanoparticle formulations (safety) and antimicrobial efficacy. | MTT, resazurin; resazurin used for biofilm viability testing [25]. |
The optimization of nanoparticle formulations, particularly through the precise calibration of cholesterol density and ionizable lipid content, is a decisive factor in bridging the gap between therapeutic potential and clinical reality. For both CRISPR-based genetic medicines and advanced antimicrobial strategies, a deep understanding of composition-function relationships enables the rational design of superior delivery systems. The experimental frameworks and comparative data provided here underscore that a systematic, DoE-driven approach to formulation is not merely beneficial but essential for achieving the robust, high-efficacy performance required to outmatch conventional therapies and address pressing global health challenges, such as antimicrobial resistance. The future of nanomedicine lies in this meticulous, data-informed optimization paradigm.
Antimicrobial coatings are critical for maintaining sterile surfaces in healthcare and public settings, but their long-term efficacy is often compromised by environmental stressors such as ultraviolet (UV) light and cleaning agents. The emergence of nanoparticle-enhanced CRISPR-based antimicrobial coatings represents a paradigm shift in the design of durable, smart surfaces. These advanced systems integrate the gene-editing precision of CRISPR-Cas technology with the protective and delivery capabilities of nanoparticles (NPs), creating a multifaceted defense against pathogenic bacteria [25]. This guide provides a comparative analysis of these next-generation coatings against conventional alternatives, with a specific focus on their resilience to UV exposure and chemical cleaning protocols. The performance evaluation is grounded in experimental data, offering researchers and product developers a scientific basis for material selection and formulation optimization.
Comparative Performance of Antimicrobial Coating Technologies
The durability of an antimicrobial coating is a direct function of its resistance to physical and chemical degradation. UV light and cleaning agents can degrade active components, polymer matrices, and delivery systems, ultimately compromising antimicrobial efficacy. The table below compares the performance of conventional and advanced coatings under standardized stress conditions.
Table 1: Comparative Performance of Antimicrobial Coatings Under Degradation Stressors
| Coating Technology | Key Active Component(s) | UV Exposure Impact (Accelerated Aging) | Cleaning Agent Resistance (to 10+ cycles) | Post-Degradation Antimicrobial Efficacy |
|---|---|---|---|---|
| Silver Nanoparticle (AgNP) Coating | Metallic Silver NPs | ~40-60% nanoparticle aggregation; reduced ion release [9] | Variable: high resistance to quats; susceptible to chlorine-based agents [45] | Efficacy drop up to 70% due to reduced active surface area |
| Quaternary Ammonium Polymer | Organic QAC compounds | Polymer chain scission; loss of cationic charge density | Progressive loss of surface-active groups; deactivated by anionic surfactants | Up to 80% reduction in contact-killing activity |
| CRISPR/Nanoparticle Hybrid (e.g., AuNP-Cas9) | CRISPR-Cas9 ribonucleoprotein complex loaded on Gold NPs | AuNPs protect CRISPR components; <10% loss of guide RNA integrity with UV-stabilized polymers [25] | High functional resilience to ethanol, phenol; requires formulation for chlorine resistance [25] [45] | Maintains >90% gene-editing efficiency and biofilm disruption post-stress [25] |
| Bacteriophage-Encapsulating Hydrogel | Lytic bacteriophages | UV-C directly inactivates >99% of surface phages; UV-A/B degrades hydrogel matrix | Phage leaching and inactivation by oxidizing agents (H₂O₂, chlorine) [45] | Near-complete loss of lytic activity after UV and chemical exposure |
Experimental Protocols for Coating Durability Assessment
To generate comparable data, researchers employ standardized protocols to simulate long-term degradation. The following methodologies are critical for evaluating coating performance.
UV Degradation and Antimicrobial Efficacy Protocol
This protocol assesses the impact of accelerated UV aging on a coating's physical integrity and biological activity.
- Sample Preparation: Coatings are applied to standardized surfaces (e.g., stainless steel, medical-grade plastic) and cured according to manufacturer specifications. A minimum of five replicates per test group is recommended.
- UV Stress Procedure: Samples are placed in a QUV accelerated weathering tester equipped with UVA-340 lamps to simulate solar UV. A typical cycle involves 8 hours of UV exposure at 0.7 W/m² at 50°C, followed by 4 hours of condensation at 40°C. The test is run for 500-1000 hours to simulate 6-12 months of indoor ambient exposure.
- Post-Exposure Analysis:
- Physical Characterization: Scanning Electron Microscopy (SEM) is used to visualize nanoparticle aggregation or surface cracking. Spectrophotometry measures changes in optical density or color.
- Antimicrobial Efficacy Testing: Treated and control surfaces are challenged with a known inoculum (e.g., ~10⁶ CFU/mL of methicillin-resistant Staphylococcus aureus (MRSA) or Pseudomonas aeruginosa) according to ISO 22196:2011. After a 2-hour contact time, surviving bacteria are eluted, plated, and counted. The reduction in viable bacteria is calculated relative to an uncoated control [25].
Cleaning Agent Resistance and Functional Integrity Protocol
This test evaluates a coating's ability to withstand repeated cleaning cycles while retaining its function.
- Cleaning Simulation: A motorized abrasion machine (e.g., Taber Abraser or linear washability tester) is used. Cloth or sponges are saturated with a selected disinfectant.
- Disinfectant Solutions: Test a panel of common agents [45]:
- 70% Ethanol
- 0.1% Benzalkonium Chloride (a quaternary ammonium compound)
- 1000 ppm Sodium Hypochlorite (bleach)
- 3% Hydrogen Peroxide
- 0.5% Phenol
- Testing Cycle: Each cleaning cycle consists of a fixed number of back-and-forth strokes under a constant pressure. Samples undergo 1, 5, 10, and 20 cycles.
- Post-Cleaning Analysis:
- Functional Assay: For CRISPR-NP coatings, the functional integrity is quantified by extracting the ribonucleoprotein complex and assessing its gene-editing efficiency in a reporter bacterial strain. A reduction in the efficiency of knocking out a fluorescent protein gene, for instance, is measured via flow cytometry [25].
- Chemical Resistance: Fourier-Transform Infrared Spectroscopy (FTIR) is used to detect chemical changes in the coating's polymer matrix.
Mechanism of Action and Degradation Pathways
The superior durability of nanoparticle-enhanced CRISPR coatings stems from their multi-layered design and mechanism of action. The following diagram illustrates the functional workflow and the points where environmental stressors cause degradation in conventional systems.
Figure 1: Functional workflow and degradation pathways of NP-enhanced CRISPR coatings versus conventional coatings.
The intrinsic stability of gold and other inorganic nanoparticles provides a physical shield for the encapsulated CRISPR machinery against molecular degradation by UV photons and chemical oxidizers [25]. In contrast, conventional coatings, which often rely on the direct contact-killing of organic biocides or the release of metal ions, see their active components rapidly decomposed by the same stressors.
Research Reagent Solutions for Coating Development
The development and testing of advanced antimicrobial coatings require a specific toolkit of reagents and materials. The following table details key solutions for researchers in this field.
Table 2: Essential Research Reagents for Antimicrobial Coating Development
| Reagent/Material | Function in R&D | Application Example |
|---|---|---|
| Gold Nanoparticles (AuNPs) | Carrier for CRISPR-Cas components; provides stability and enhances bacterial uptake [25]. | Conjugated with Cas9-gRNA complex for targeted gene editing in P. aeruginosa biofilms. |
| Liposomal Nanoparticles | Encapsulates and protects sensitive biomolecules (e.g., guide RNA) from degradation [25]. | Used to deliver CRISPR payloads, demonstrating >90% biofilm biomass reduction in vitro. |
| CRISPR-Cas9 Ribonucleoprotein (RNP) | The active gene-editing complex; targets specific antibiotic resistance or virulence genes [46] [25]. | Engineered to target and disrupt the mecA gene in MRSA, restoring beta-lactam susceptibility [47]. |
| UV-Stabilizing Polymers (e.g., specific polyacrylates) | Coating matrix that minimizes UV penetration and protects encapsulated active agents. | Formulated as the hydrogel housing for CRISPR-NP complexes to extend functional longevity. |
| Specific Guide RNAs (gRNAs) | Provides targeting specificity by binding to complementary DNA sequences of interest [46]. | Designed to target blaNDM-1 or vanA resistance genes in Gram-negative and Gram-positive pathogens, respectively [46]. |
| Quaternary Ammonium Compounds (Quats) | Standard biocidal agent for benchmarking the performance of conventional antimicrobial coatings. | Used as a positive control in disinfectant efficacy tests (e.g., against benzalkonium chloride) [45]. |
The experimental data and comparative analysis presented in this guide underscore a significant divergence in the durability profiles of antimicrobial coating technologies. Conventional biocidal coatings, while effective initially, exhibit a fundamental vulnerability to the environmental conditions of their intended use. In contrast, nanoparticle-enhanced CRISPR coatings leverage a robust, NP-protected delivery system and a gene-targeting mechanism that is less dependent on the immediate chemical integrity of the surface. This paradigm shifts the definition of coating longevity from merely "persisting as a chemical substance" to "maintaining a precise biological function" over time. For researchers and developers, the future lies in optimizing the NP matrix for broader chemical resistance and formulating coatings that can be seamlessly integrated with hospital disinfection protocols that are most effective at degrading residual resistance genes, such as UV light and phenol-based cleaners [45].
Antimicrobial resistance (AMR) represents one of the most pressing global health threats of our time, with projections suggesting it may cause 10 million deaths annually by 2050 if left unaddressed [36]. The escalating crisis is fueled by the rapid evolution of multidrug-resistant (MDR) pathogens and the stagnation in conventional antibiotic development, with only two new antibiotic classes approved in recent decades [21]. This alarming trend has catalyzed the pursuit of innovative antimicrobial strategies that can overcome the limitations of traditional approaches.
Two particularly promising frontiers have emerged: nanoparticle-enhanced CRISPR/Cas9 systems, which target resistance at its genetic roots, and advanced antimicrobial coatings, which provide surface-level protection. While both approaches offer compelling advantages over conventional antibiotics, their long-term efficacy hinges on a critical, often understudied factor: the potential for microbial adaptation. This analysis provides a structured comparison of these innovative strategies, evaluating their mechanisms, efficacy, and vulnerability to resistance development to inform future research and development priorities.
Comparative Performance Analysis: Nanoparticle-Enhanced CRISPR vs. Antimicrobial Coatings
The table below provides a quantitative comparison of the performance characteristics of nanoparticle-enhanced CRISPR/Cas9 systems and antimicrobial coatings, synthesizing data from recent experimental studies.
Table 1: Performance Comparison of Nanoparticle-Enhanced CRISPR and Antimicrobial Coating Strategies
| Performance Metric | Nanoparticle-Enhanced CRISPR/Cas9 | Conventional Antimicrobial Coatings |
|---|---|---|
| Reported Efficacy Range | 4.7% to 100% bacterial resensitization [48]; >90% biofilm biomass reduction in vitro [3] | Varies widely with material, environmental conditions, and microbial load [15] |
| Targeting Precision | High (sequence-specific gRNA targeting) [38] | Low (broad-spectrum, contact-based action) [15] [11] |
| Adaptation Concern | Potential for target site mutation, CRISPR component degradation [38] | Development of resistance to metal ions, enzyme inactivation, biofilm formation on coatings [15] [11] |
| Delivery Efficiency | 3.5-fold increase in editing efficiency with gold nanoparticles vs. non-carrier systems [3] | Dependent on agent incorporation method and release kinetics [15] |
| Synergy with Antibiotics | Yes, enables co-delivery and resensitization [3] [48] | Yes, some coatings combine agents for broader efficacy [15] |
| Primary Action Mode | Genetic disruption of resistance or essential genes [3] [38] | Physical membrane disruption, ROS generation, metabolic interference [15] [11] |
Mechanisms of Action and Experimental Protocols
Nanoparticle-Enhanced CRISPR/Cas9 Systems
Core Mechanism: This strategy harnesses the precision of the CRISPR/Cas9 gene-editing system, which can be programmed to introduce double-strand breaks in specific bacterial DNA sequences. The system is delivered via engineered nanoparticles, which protect the genetic material and enhance uptake by bacterial cells [3] [48]. The mechanism can proceed via two primary pathways:
- Targeted Killing: Guide RNAs (gRNAs) are designed to target essential bacterial genes or fitness genes, leading to lethal DNA damage and selective elimination of resistant strains [38].
- Resensitization: gRNAs target and disrupt specific antibiotic resistance genes (e.g., blaNDM-1, mcr-1), rendering the bacteria susceptible again to conventional antibiotics [48] [38].
Detailed Experimental Protocol for CRISPR-Resensitization Assay
- gRNA Design and Complex Formation: Design gRNAs complementary to the target resistance gene (e.g., blaKPC, mcr-1). Formulate ribonucleoprotein (RNP) complexes of Cas9 nuclease and gRNA. Complex these RNPs with lipid-based or gold nanoparticles [3] [48].
- Bacterial Culture and Treatment: Grow a standardized culture of the target MDR bacterium (e.g., carbapenem-resistant K. pneumoniae). Divide into experimental groups: a) CRISPR-NP treated, b) Non-treated control, c) Nanoparticle-only control.
- Delivery and Incubation: Add CRISPR-NP complexes to the culture. Use conjugative plasmids or electroporation as an alternative delivery control. Incude to allow for cellular uptake and gene editing [48] [38].
- Efficacy Assessment:
- Resensitization Check: Subculture treated bacteria on agar plates containing the previously ineffective antibiotic (e.g., meropenem). Compare growth to controls. Calculate the percentage of resensitized cells in the population [48].
- Genetic Verification: Use PCR and sequencing to confirm precise cleavage and disruption of the target resistance gene in colonies that regained antibiotic sensitivity.
- Adaptation Monitoring: Passage surviving bacteria repeatedly in sub-inhibitory concentrations of the CRISPR-NP system. Sequence the target loci and bacterial genomes from subsequent generations to identify mutations that confer escape, such as single nucleotide polymorphisms (SNPs) in the gRNA target sequence or genes involved in nanoparticle uptake [38].
Antimicrobial Polymeric Coatings
Core Mechanism: These coatings prevent microbial surface colonization and biofilm formation through a combination of contact-based antimicrobial activity and anti-fouling properties. They incorporate active agents like metal nanoparticles (e.g., Ag, Cu), enzymes, or plant-derived compounds. These agents are embedded in a polymer matrix (e.g., alginate, synthetic polymers) and act primarily via non-specific mechanisms [15] [11].
- Metal Ion Release: Silver nanoparticles (AgNPs) release Ag⁺ ions that generate reactive oxygen species (ROS), damage cell membranes, and disrupt metabolic pathways [11].
- Contact-Killing: Cationic polymers or engineered surfaces physically disrupt microbial membranes upon contact [15].
- Biofilm Inhibition: By preventing initial adhesion or killing planktonic cells, these coatings stop the formation of the protective extracellular polymeric substance (EPS) matrix [15].
Detailed Experimental Protocol for Coating Efficacy and Adaptation Assay
- Coating Fabrication and Characterization: Incorporate the antimicrobial agent (e.g., AgNPs, copper oxide, enzyme) into the selected polymer matrix (e.g., alginate, polyurethane) via blending or surface immobilization. Characterize the coating's homogeneity, thickness, and agent release kinetics using standardized methods [15].
- Baseline Efficacy Testing:
- Zone of Inhibition: Place coated samples on agar plates seeded with test organisms (e.g., S. aureus, P. aeruginosa). Measure the zone of inhibition after incubation [15].
- Biofilm Assay: Expose coated surfaces to bacterial suspension in a flow cell or static system. After incubation, quantify adhered biomass using crystal violet staining or viable cell counts [3] [15].
- JIS/ASTM Standards: Perform quantitative tests per standards like JIS Z 2801 to determine log reduction against controls [15].
- Adaptation Study (Serial Passage):
- Repeatedly expose bacterial cultures to sub-lethal concentrations of the eluted antimicrobial agents or to direct contact with coated surfaces over multiple generations.
- Regularly assess the Minimum Inhibitory Concentration (MIC) of the eluted agent against the passaged strains and compare it to the original strain to identify any increase in tolerance.
- Genomically sequence adapted strains to identify mutations conferring resistance, such as efflux pump upregulation, silver resistance genes (e.g., sil operon), or enhanced EPS production [11].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful research into these antimicrobial strategies and their vulnerability to adaptation requires a specific set of reagents and tools. The following table details key solutions for implementing the described experimental protocols.
Table 2: Key Research Reagent Solutions for Investigating Antimicrobial Strategies and Adaptation
| Item | Function/Application | Justification |
|---|---|---|
| Lipid Nanoparticles (LNPs) | Delivery vehicle for in vivo CRISPR-Cas9 components [28]. | Biocompatible; naturally accumulate in the liver; allow for potential re-dosing [28]. |
| Gold Nanoparticles (AuNPs) | Carrier for CRISPR-RNP complexes in vitro [3]. | High editing efficiency (3.5-fold increase reported); easily functionalized [3]. |
| Conjugative Plasmids | Delivery vector for CRISPR machinery into bacterial populations [48] [38]. | Enable horizontal gene transfer of CRISPR system; high resensitization efficacy shown [48]. |
| Silver Nanoparticles (AgNPs) | Broad-spectrum antimicrobial agent for coatings [15] [11]. | Generate ROS; disrupt membranes; widely studied and effective [11]. |
| Alginate & Synthetic Polymers | Matrix for antimicrobial coatings [15]. | Natural (alginate) and synthetic polymers provide versatile, controllable release matrices [15]. |
| Standardized Test Organisms | For efficacy and adaptation assays (e.g., MRSA, P. aeruginosa, CRKP) [3] [48]. | Represent priority pathogens with clinical relevance and known resistance mechanisms [36] [21]. |
The battle against antimicrobial resistance is advancing from conventional chemistry to sophisticated genetic and materials science. Nanoparticle-enhanced CRISPR/Cas9 offers a "scalpel" - a highly precise tool for targeting specific genetic resistance determinants, with the powerful ability to resensitize bacteria to existing antibiotics. In contrast, advanced antimicrobial coatings serve as a "shield" - a broad-spectrum, surface-level barrier to prevent colonization and biofilm formation.
Critically, neither strategy is immune to the powerful force of microbial evolution. CRISPR-based approaches face potential failure from point mutations in target sequences, while coatings may select for microbes with enhanced efflux, detoxification, or biofilm capabilities. The future of sustainable antimicrobial technology lies not in a single magic bullet, but in the intelligent combination of these approaches—perhaps a CRISPR-resensitizing strategy deployed from a smart, antimicrobial surface—coupled with rigorous, pre-emptive research into adaptation pathways to design solutions that remain effective in the long term.
A Head-to-Head Comparison: Efficacy, Specificity, and Clinical Outlook
The persistent challenge of microbial contamination on surfaces, particularly in healthcare and industrial settings, has driven the development of advanced antimicrobial technologies. Traditional antimicrobial coatings have long been the first line of defense, with their efficacy quantitatively assessed using standardized protocols like JIS Z 2801 [49] [50]. However, the formidable resilience of biofilms—structured communities of microorganisms embedded in a protective extracellular matrix—has revealed the limitations of conventional approaches [51]. Biofilms can exhibit up to a 1000-fold greater tolerance to antibiotics and disinfectants compared to their free-floating (planktonic) counterparts, making associated infections chronic and highly recalcitrant to treatment [3]. This landscape is now being transformed by emerging strategies that combine nanoparticle (NP) delivery systems with the precision of CRISPR-Cas9 gene-editing technology. This guide provides a comparative analysis of the efficacy data, experimental methodologies, and mechanistic actions of conventional antimicrobial coatings versus novel nanoparticle-CRISPR systems, offering researchers a clear framework for evaluating these divergent approaches.
Conventional Antimicrobial Coatings and Their Quantification
Conventional antimicrobial coatings function as a preventive layer on surfaces, actively killing microorganisms (biocidal) or preventing their attachment (antifouling) [52]. Their performance is most reliably quantified using standardized industrial tests.
The JIS Z 2801 Standard Test Protocol
The JIS Z 2801 standard, also adopted as ISO 22196, is the industry benchmark for evaluating the antibacterial activity of plastics and other hard, non-porous surfaces [49] [50]. The test is designed to provide a quantitative measure of a material's ability to inhibit bacterial growth or kill bacteria over a 24-hour contact period. The following diagram illustrates the key steps of this standardized workflow:
Key Steps in the JIS Z 2801 Methodology [49] [50]:
- Test Organisms: The standard mandates the use of Escherichia coli (ATCC 8739) and Staphylococcus aureus (ATCC 6538P), though other strains like Pseudomonas aeruginosa can be added.
- Inoculation and Incubation: A standardized, nutritive bacterial suspension is applied to both the antimicrobial test sample and a non-treated control sample. The inoculum is covered with a sterile, inert film to ensure close contact with the surface and prevent evaporation. The samples are then incubated for 24 hours at 35°C and high relative humidity.
- Quantitative Analysis: After incubation, viable bacteria are recovered from both surfaces using a neutralizing solution, serially diluted, and plated on agar. After a further incubation, the number of colony-forming units (CFU) is counted. The antibacterial activity (R) is calculated as R = (Ut - U0) - (At - U0), where Ut and At are the mean CFU recovered from the control and test surfaces after 24 hours, and U0 is the CFU on the control surface immediately after inoculation. A related value, the log reduction, is often reported and is calculated as Log10(Ut) - Log10(At). A product is generally considered effective if it demonstrates a significant log reduction (often ≥2, equivalent to 99% killing) compared to the control [49] [50].
Efficacy Data for Conventional Coatings
A comparative study of 23 commercially available antimicrobial coatings tested against E. coli via the ISO 22196 (identical to JIS Z 2801) protocol reveals a wide range of performance. The table below summarizes quantitative data from key coating technologies:
Table 1: Quantitative Efficacy of Conventional Antimicrobial Coatings (vs. E. coli per ISO 22196)
| Antimicrobial Agent | Mechanism of Action | Log Reduction | % Inhibition | Substrate |
|---|---|---|---|---|
| Titanium Dioxide (Photocatalytic) | Light-activated reactive oxygen species [52] | 6.6 | 100% | Stainless Steel [53] |
| Quaternary Ammonium Salt (Immobilized) | Membrane disruption, cell lysis [52] | 6.1 | 100% | Glass [53] |
| Nanosilver | Ion release, protein/DNA damage [53] | 5.5 - 6.2 | 95% - 100% | Glass & Steel [53] |
| Triclosan | Inhibition of fatty acid synthesis [53] | 6.2 | 100% | Glass [53] |
| Poly(dimethylsiloxane) | Antifouling (physical repulsion) [53] | 3.2 - 4.7 | 54% - 100% | Glass & Steel [53] |
| Zinc Pyrithione | Bacteriostatic, metal toxicity [53] | 6.1 | 100% | Glass [53] |
Strengths and Weaknesses of the JIS Z 2801 Test [50]:
- Strengths: It is a quantitative, reproducible method that provides bacteria with nutrients, thereby testing both bactericidal and bacteriostatic properties. Its standardization allows for cross-lab comparisons.
- Weaknesses: It represents a "best-case" scenario where microbes are kept in a moist, nutritive environment for 24 hours. This is not always representative of real-world conditions where contaminants may dry quickly, limiting the interaction time between the microbe and the antimicrobial surface.
The Biofilm Challenge and Nanoparticle-CRISPR Solutions
Biofilms pose a significantly greater challenge than planktonic bacteria due to their complex structure and multiple resistance mechanisms.
The Biofilm Lifecycle and Resistance Mechanisms
The formation of a biofilm is a cyclic, multi-stage process that results in a highly protected microbial community, as shown below:
The robust nature of a mature biofilm contributes to its resistance through several key mechanisms [3] [54] [51]:
- The Extracellular Polymeric Substance (EPS) Matrix: This complex mixture of polysaccharides, proteins, and extracellular DNA (eDNA) acts as a physical barrier, hindering the penetration of antimicrobial agents. It can also directly bind and neutralize certain antibiotics, such as positively charged aminoglycosides [51].
- Metabolic Heterogeneity: Gradients of nutrients and oxygen within the biofilm create microenvironments. Cells in the deeper layers enter a slow-growing or dormant (persister) state, making them less susceptible to antibiotics that target active cellular processes [3] [51].
- Enhanced Horizontal Gene Transfer: The dense, protected environment of a biofilm facilitates the efficient exchange of antibiotic resistance genes between bacteria, accelerating the development of resistance [3].
Nanoparticle-Enhanced CRISPR-Cas9 Antimicrobials
To overcome the limitations of conventional agents against biofilms, a novel therapeutic strategy is emerging: combining CRISPR-Cas9 gene-editing with nanoparticle (NP) delivery systems. This approach shifts the paradigm from general toxicity to precision genetic targeting.
Mechanism of Action: The core concept is to use a guide RNA (gRNA) to direct the Cas9 nuclease to specific sequences in the bacterial genome. The target can be antibiotic resistance genes (e.g., bla, mecA), virulence factors, or genes essential for biofilm formation and quorum sensing [3] [55]. By introducing double-strand breaks in these genes, CRISPR-Cas9 can resensitize bacteria to traditional antibiotics or dismantle their ability to form and maintain biofilms.
The Role of Nanoparticles: The clinical application of CRISPR is hindered by delivery challenges. Nanoparticles serve as ideal carriers because they [3] [56]:
- Protect the CRISPR-Cas9 machinery (Cas9 protein and gRNA) from degradation.
- Enhance cellular uptake and penetration through the dense biofilm EPS matrix.
- Can be engineered for targeted delivery and controlled release.
- Enable co-delivery of CRISPR components with antibiotics or antimicrobial peptides for a synergistic effect.
The following diagram illustrates the workflow for developing and testing this combinatorial therapy:
Efficacy Data for Nanoparticle-CRISPR Systems
Early-stage research, primarily in vitro, demonstrates the potent efficacy of this combinatorial approach. The table below summarizes key performance metrics from recent studies:
Table 2: Quantitative Efficacy of Nanoparticle-CRISPR Antimicrobials Against Biofilms
| Nanoparticle Carrier | CRISPR Target | Test Organism | Key Efficacy Metrics |
|---|---|---|---|
| Liposomal Nanoparticles [3] | Antibiotic resistance/ biofilm genes | Pseudomonas aeruginosa | >90% reduction in biofilm biomass [3] |
| Gold Nanoparticles [3] | Antibiotic resistance genes | Model Bacteria | 3.5-fold increase in gene-editing efficiency vs. non-carrier systems; synergistic biofilm disruption with antibiotics [3] |
| Lipid Nanoparticle Spherical Nucleic Acids (LNP-SNAs) [56] | N/A (Delivery efficiency study) | Human cells (in culture) | 3x improved cell entry and 3x higher gene-editing efficiency vs. standard LNPs [56] |
Comparative Analysis: Conventional vs. Nanoparticle-CRISPR
The following table provides a direct, point-by-point comparison of the two technologies, highlighting their fundamental differences.
Table 3: Direct Comparison of Conventional Coatings and Nanoparticle-CRISPR Systems
| Aspect | Conventional Antimicrobial Coatings | Nanoparticle-CRISPR Systems |
|---|---|---|
| Primary Mechanism | Broad-spectrum chemical toxicity (e.g., membrane disruption, protein denaturation) | Precision genetic targeting (e.g., disruption of resistance/virulence genes) |
| Target | Primarily planktonic bacteria; limited efficacy against mature biofilms | Designed to target biofilm-specific resistance and genetic traits |
| Efficacy Measure | Log reduction (e.g., 3-6 log) against planktonic bacteria in 24h (JIS Z 2801) | Percentage reduction in biofilm biomass & resensitization to antibiotics |
| Development Stage | Commercially available, standardized testing, widely regulated | Pre-clinical research, early-phase clinical trials for some platforms [28] |
| Key Advantage | Immediate surface protection, well-established regulatory pathway | High specificity, potential to reverse antibiotic resistance, programmable |
| Key Limitation | Limited biofilm penetration, can drive chemical resistance | Complex delivery, potential off-target effects, nascent regulatory path |
The Scientist's Toolkit: Essential Research Reagents and Materials
For researchers embarking on efficacy testing in this field, the following core reagents and materials are essential.
Table 4: Essential Research Reagents and Materials
| Reagent / Material | Function / Application | Examples / Specifications |
|---|---|---|
| Standard Test Strains | For standardized efficacy testing (JIS Z 2801) | E. coli (ATCC 8739), S. aureus (ATCC 6538P) [49] |
| Culture Media | Bacterial growth and recovery | Tryptic Soya Agar (TSA), Lysogeny broth (LB) agar, Neutralizing broths (e.g., SCDLP) [49] |
| CRISPR-Cas9 Components | Precision genetic editing | Cas9 nuclease, guide RNA (gRNA) targeting specific bacterial genes [3] |
| Nanoparticle Carriers | Delivery of antimicrobials or CRISPR payload | Liposomal NPs, Gold NPs, Lipid Nanoparticles (LNPs), LNP-SNAs [3] [56] |
| Biofilm Assessment Tools | Quantification and visualization of biofilms | Confocal Laser Scanning Microscopy (CLSM), Crystal Violet Staining, CFU counting from disrupted biofilms [51] |
This comparative guide illustrates the distinct profiles of two generations of antimicrobial technology. Conventional antimicrobial coatings, quantified by robust standards like JIS Z 2801, provide a crucial first line of defense against surface contamination by planktonic bacteria, with many products achieving high log reductions (3-6 log) under test conditions [53] [50]. However, their effectiveness is significantly limited against complex, protected biofilm communities [51].
The emerging nanoparticle-enhanced CRISPR approach represents a paradigm shift towards precision antimicrobials. By directly targeting the genetic basis of antibiotic resistance and biofilm integrity, it offers a promising, mechanism-based strategy to combat chronic, biofilm-associated infections, demonstrating dramatic biomass reduction (>90%) in early studies [3]. The primary challenges for this new technology lie in optimizing safe and efficient in vivo delivery, minimizing off-target effects, and navigating a path to clinical regulation and approval [28] [55].
Future research will likely focus on synergizing these approaches—using broad-spectrum coatings for immediate protection while deploying targeted CRISPR-based therapeutics for treating established, resistant infections. Furthermore, the advancement of novel nanoparticle systems, such as LNP-SNAs, promises to unlock higher efficacy and safer delivery, ultimately translating the power of gene editing from the lab bench to the clinic [56].
The escalating crisis of antimicrobial resistance (AMR) necessitates a paradigm shift from conventional antibiotic treatments to innovative technological solutions. Among the most promising strategies are antimicrobial nanoparticles, which offer broad-spectrum activity, and CRISPR-based systems, which provide precise, sequence-specific targeting. While seemingly divergent, these approaches are increasingly synergistic, particularly when nanoparticles are employed to deliver CRISPR components, creating a powerful combined modality. This guide objectively compares the performance, experimental data, and applications of nanoparticle-based antimicrobial coatings and sequence-specific CRISPR antimicrobials, providing researchers with a clear framework for selecting and implementing these technologies. The comparison is framed within the context of a broader thesis on nanoparticle-enhanced CRISPR versus conventional antimicrobial coating performance, highlighting the distinct yet complementary nature of these approaches in combating multidrug-resistant pathogens.
Broad-Spectrum Antimicrobial Nanoparticles function as potent, non-specific agents primarily through physicochemical interactions. Their activity does not rely on specific molecular recognition but on general properties like surface charge, reactivity, and size. Key mechanisms include:
- Membrane Disruption: Positively charged nanoparticles (e.g., certain metal oxides) interact with and disrupt negatively charged bacterial membranes, causing cell lysis [11].
- Reactive Oxygen Species (ROS) Generation: Metallic nanoparticles like silver (AgNPs) and zinc oxide (ZnONPs) catalyze the production of ROS, leading to oxidative stress, DNA damage, and protein inactivation [11].
- Ion Release: Materials such as silver and copper nanoparticles continuously release antimicrobial ions that interfere with cellular enzymes and DNA [4] [11].
In contrast, Sequence-Specific CRISPR Antimicrobials employ a biologically precise mechanism derived from bacterial immune systems. The Cas nuclease, guided by a custom-designed RNA molecule (gRNA), introduces double-strand breaks in the DNA of target pathogens. This system can be programmed to:
- Disrupt Antibiotic Resistance Genes: By cleaving genes encoding for enzymes like beta-lactamases, bacteria can be re-sensitized to conventional antibiotics [25] [9].
- Target Essential Genes or Virulence Factors: gRNAs can be designed to knockout genes essential for bacterial survival or those involved in pathogenicity and quorum sensing [25] [57].
The quantitative performance of these technologies, as demonstrated in recent studies, is summarized in the table below.
Table 1: Quantitative Performance Comparison of Antimicrobial Technologies
| Technology | Target Pathogen | Efficacy Metric | Reported Value | Key Findings |
|---|---|---|---|---|
| Liposomal CRISPR-Cas9 [25] | Pseudomonas aeruginosa (biofilm) | Reduction in biofilm biomass (in vitro) | >90% | Targeted disruption of antibiotic resistance genes or biofilm-regulating factors. |
| CRISPR-Gold Nanoparticle Hybrids [25] | Model Bacterial Systems | Gene-editing efficiency increase | ~3.5-fold | Enhanced delivery and synergy with antibiotics. |
| Silver Nanoparticles (AgNPs) [11] | Staphylococcus aureus, E. coli | Minimum Inhibitory Concentration (MIC) | ~50 μg/mL | Effective broad-spectrum activity via ROS generation and membrane disruption. |
| CRISPR-Cas9 (LNP delivery) [28] | Hereditary Transthyretin Amyloidosis (in humans) | Reduction in disease-related protein (TTR) | ~90% | Sustained protein level reduction in clinical trial participants. |
Experimental Protocols and Workflows
To ensure reproducibility, this section outlines standard protocols for evaluating both broad-spectrum coatings and CRISPR-based antimicrobials.
Protocol for Assessing Broad-Spectrum Nanoparticle Coatings
This protocol is adapted from methodologies used to evaluate metallic and polymeric nanoparticles [4] [11].
Nanoparticle Synthesis and Coating Fabrication:
- Green Synthesis: Mix 1 mM aqueous solution of silver nitrate (AgNO₃) with a 10% (v/v) plant extract (e.g., from Lysiloma acapulcensis or tomato peel). Incubate at room temperature in the dark for 24 hours until a color change indicates nanoparticle formation [11].
- Coating Application: Functionalize the surface (e.g., medical-grade polymer) by dip-coating or spray-coating with the synthesized nanoparticle suspension. Dry and cure the coating according to material specifications.
Antimicrobial Efficacy Testing:
- Culture Preparation: Grow test strains (e.g., S. aureus ATCC 25923, E. coli ATCC 25922) to mid-log phase in Mueller-Hinton broth.
- Assay Setup:
- Minimum Inhibitory Concentration (MIC): Use a broth microdilution method with serially diluted nanoparticles in a 96-well plate. The MIC is the lowest concentration that inhibits visible growth after 18-24 hours at 37°C [11].
- Biofilm Assay (for surface coatings): Place coated coupons in a culture plate, inoculate with bacteria, and incubate for 48 hours to allow biofilm formation. Assess biofilm viability using a metabolic assay like MTT or by sonicating and plating for colony-forming unit (CFU) counts.
Mechanism of Action Analysis:
- ROS Detection: Use a fluorescent probe, H₂DCFDA, to quantify intracellular ROS levels in bacteria exposed to nanoparticles via fluorometry [11].
- Membrane Integrity: Employ propidium iodide staining and fluorescence microscopy to visualize compromised bacterial membranes.
Protocol for CRISPR-Based Antimicrobial Targeting with Nanoparticle Delivery
This protocol details the use of lipid nanoparticles (LNPs) to deliver CRISPR components for targeted gene disruption, based on successful in vitro and clinical approaches [28] [25].
gRNA Design and Complex Formation:
- In silico Design: Using software like CRISPOR, design a gRNA with high predicted on-target activity against the desired gene (e.g., an antibiotic resistance gene) and minimal off-target effects [58].
- RNP Complex Formation: Chemically synthesize the gRNA with stabilizing modifications (2'-O-methyl analogs). Complex the gRNA with a high-fidelity Cas9 protein to form a ribonucleoprotein (RNP) complex.
Nanoparticle Formulation and Delivery:
- LNP Encapsulation: Encapsulate the CRISPR RNP complex within lipid nanoparticles (LNPs) using a microfluidic mixer. LNPs should be composed of ionizable lipids, phospholipids, cholesterol, and PEG-lipids. Purify the formulation via dialysis or tangential flow filtration [28] [25].
- In Vitro Delivery: Add the LNP formulation to a bacterial culture or biofilm model at a predetermined multiplicity of infection (MOI). Incubate for 4-6 hours, then replace with fresh media.
Efficacy and Specificity Validation:
- Editing Efficiency: After 48-72 hours, extract genomic DNA. Use targeted next-generation sequencing (NGS) or T7 Endonuclease I assay to quantify the frequency of indels at the target locus.
- Phenotypic Validation:
- Resistance Reversal: If targeting a resistance gene, perform a antibiotic susceptibility test to measure the change in MIC.
- Biofilm Disruption: For biofilm-related targets, quantify biomass reduction using crystal violet staining or confocal microscopy [25].
- Off-Target Analysis: Use GUIDE-seq or CIRCLE-seq to identify and confirm the absence of editing at potential off-target sites [58].
Diagram: Experimental workflow for developing and testing nanoparticle-enhanced CRISPR antimicrobials
Mechanisms of Action: A Visual Guide
The fundamental difference between these technologies lies in their scope of action: broad-spectrum nanoparticles act through non-specific physicochemical mechanisms, while CRISPR systems achieve precision through programmable genetic targeting.
Diagram: Comparative mechanisms of broad-spectrum nanoparticles and sequence-specific CRISPR
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of these technologies requires specific reagents and tools. The following table lists key materials for researchers embarking on experiments in this field.
Table 2: Essential Research Reagents and Materials
| Item | Function/Description | Example Use Case |
|---|---|---|
| High-Fidelity Cas9 Nuclease | Engineered nuclease variant with reduced off-target effects compared to wild-type SpCas9 [58]. | Critical for all therapeutic CRISPR applications to minimize safety risks. |
| Synthetic gRNA with Chemical Modifications | gRNA synthesized with 2'-O-methyl analogs (2'-O-Me) and 3' phosphorothioate bonds (PS) to enhance stability and reduce immunogenicity [58]. | Improves editing efficiency and specificity in vivo. |
| Lipid Nanoparticles (LNPs) | A delivery vehicle composed of ionizable lipids, phospholipids, cholesterol, and PEG-lipids for in vivo encapsulation and delivery of CRISPR components [28] [25]. | Systemic delivery of CRISPR RNPs; demonstrated in clinical trials for liver targets. |
| Silver Nanoparticles (AgNPs) | Metallic nanoparticles known for potent, broad-spectrum antimicrobial activity primarily through ROS generation [11]. | Used in antimicrobial coatings for medical devices, wound dressings, and surfaces. |
| CRISPOR Software | A widely used web tool for designing and selecting gRNAs with optimal on-target efficiency and minimal predicted off-target sites [58]. | The first step in any CRISPR experiment for rational gRNA design. |
| Inference of CRISPR Edits (ICE) | A free, web-based tool for analyzing Sanger sequencing data to determine CRISPR editing efficiency and profile the spectrum of induced indels [58]. | Accessible analysis for discovery-stage research and validation. |
The choice between broad-spectrum antimicrobial coatings and sequence-specific CRISPR targeting is not a matter of superiority but of strategic application. Broad-spectrum nanoparticle coatings offer a powerful, immediate-defense solution for surfaces, medical devices, and situations where the causative pathogen is unknown. Their strength lies in their physical robustness and ability to target a wide range of microbes simultaneously. Conversely, sequence-specific CRISPR antimicrobials represent the future of precision medicine, capable of selectively eliminating pathogens or resensitizing them to antibiotics without disrupting the surrounding microbiome. Their application is ideal for treating established, hard-to-eradicate infections, particularly those involving biofilms or defined resistance mechanisms.
The most promising horizon lies in the convergence of these fields: using engineered nanoparticles as sophisticated delivery vehicles for CRISPR payloads. This synergy combines the efficient delivery and biofilm-penetrating capabilities of nanoparticles with the unparalleled precision of CRISPR, creating a next-generation therapeutic platform to address the most pressing challenges in antimicrobial resistance.
The integration of nanotechnology into biomedical sciences, particularly for CRISPR gene editing and antimicrobial therapies, represents a paradigm shift in how we approach disease treatment. The efficacy of these advanced therapeutics, however, is fundamentally constrained by the biocompatibility of their nanocarriers. Nanoparticles (NPs) must navigate complex biological environments to deliver their payloads effectively, making their safety and toxicity profiles a critical area of investigation. This guide provides a comparative analysis of the biocompatibility of various nanoparticles and coating additives, framing the discussion within the context of enhancing CRISPR-based antimicrobial strategies against conventional antimicrobial coatings. For researchers and drug development professionals, understanding these nuances is essential for designing safer, more effective nanomedicines. The biological interactions of NPs, including cellular uptake, biodistribution, immune activation, and clearance, are largely governed by their physiochemical properties, such as size, shape, surface charge, and composition [59]. This review synthesizes current experimental data to objectively compare the performance and safety of different nanomaterial classes, providing a foundation for informed material selection in therapeutic development.
Comparative Biocompatibility of Nanoparticle Classes
The table below summarizes the key toxicity and biodistribution findings for major classes of nanoparticles, providing a direct comparison of their safety profiles.
Table 1: Comparative Safety and Biodistribution Profiles of Selected Nanoparticles
| Nanoparticle Type | Key Toxicological Findings | Primary Biodistribution Organs | Immune Response | References |
|---|---|---|---|---|
| Gold Nanoparticles (AuNPs) | Significant inflammatory response; high memory T cell activation (CD69+ CD8+ T cells: 0.40 ± 0.16). | Primarily localizes in the left lung. | Induces significant IL-6 and TNF-α elevation. | [60] |
| Quantum Dot Nanocarbons | Elevated T cell activation markers (CD25+: 0.23 ± 0.04). | Persists in kidney, liver, blood, and heart. | Activates memory T cells. | [60] |
| Unconjugated Nanodiamonds (NDs) | Favorable tolerability; minimal inflammatory response; lowest memory T cell activation (CD69+ CD8+ T cells: 0.12 ± 0.09). | Primarily accumulates in the heart. | Lowest inflammatory cytokine levels. | [60] |
| Nanobody-Conjugated NDs | Moderate inflammation triggered at 2 hours post-dosing. | Information not specified in study. | Total T cells highest (49.10% ± 6.99). | [60] |
| Silver Nanoparticles (AgNPs) | Size-dependent toxicity; smaller particles are more toxic; generates ROS leading to apoptosis. | Information not specified in study. | Can induce inflammatory responses via ROS. | [11] [59] |
Experimental Protocols for Assessing Nanoparticle Biocompatibility
To ensure the reliability and reproducibility of biocompatibility data, standardized experimental protocols are essential. The following section details key methodologies cited in the comparative studies.
In Vivo Toxicity and Biodistribution Assessment
The comparative data presented in Table 1 were derived from a well-defined in vivo study in mice, which serves as a robust model for evaluating nanoparticle tolerability [60].
- Animal Model: Female C57BL/6 mice.
- Dosing Regimen: Nanoparticles were administered intravenously at multiple concentrations (5, 10, 20, and 40 mg/kg) to assess dose-dependent effects.
- Sample Collection: Tissues and blood were collected at multiple time points (2, 24, and 96 hours) post-injection to evaluate both acute and short-term responses.
- Immune Response Analysis: Flow cytometry was employed to quantify T-cell activation and subpopulations. Specific markers included:
- CD69: An early activation marker.
- CD25: A marker for T-cell proliferation and regulatory T cells.
- Cytokine Analysis: Measurement of pro-inflammatory cytokines like IL-6 and TNF-α via assays such as ELISA.
- Biodistribution Tracking: The localization of nanoparticles in various organs (e.g., heart, lung, kidney, liver) was analyzed post-sacrifice, likely using techniques such as inductively coupled plasma mass spectrometry (ICP-MS) or fluorescence imaging for labeled particles.
In Vitro Cytotoxicity Mechanisms
For silver nanoparticles (AgNPs) and other metallic NPs, in vitro studies are crucial for elucidating the mechanisms of toxicity [11].
- Cell Culture: Use of relevant cell lines, such as macrophages (e.g., RAW 264.7) or epithelial cells.
- Viability Assays: Assessment of cell viability post-NP exposure using MTT, XTT, or similar assays to determine IC50 values.
- Reactive Oxygen Species (ROS) Detection: Application of fluorescent probes like DCFH-DA to measure oxidative stress induced by NPs.
- Apoptosis Assays: Use of annexin V/propidium iodide staining and flow cytometry to quantify apoptotic and necrotic cell populations.
Signaling Pathways and Workflow in Nanoparticle Toxicity
The toxicity of nanoparticles often proceeds through defined cellular pathways. The diagram below illustrates the primary signaling cascade initiated by toxic nanoparticles, such as AuNPs and AgNPs, leading to inflammation and cell death.
Figure 1: NP-Induced Inflammatory Apoptosis Pathway
The experimental workflow for generating the comparative toxicity data involves a structured process from nanoparticle preparation to final analysis, as visualized below.
Figure 2: NP Toxicity & Biodistribution Workflow
The Scientist's Toolkit: Essential Research Reagents
The following table lists key reagents and materials required for conducting biocompatibility and toxicity studies on nanoparticles, based on the methodologies discussed.
Table 2: Essential Research Reagents for Nanoparticle Biocompatibility Studies
| Reagent/Material | Function in Experimental Protocol | Specific Example |
|---|---|---|
| C57BL/6 Mice | An inbred, widely used animal model for in vivo toxicity and biodistribution studies. | Female mice, 6-8 weeks old [60]. |
| Functionalized Nanoparticles | The test materials whose safety profile is being investigated. | Unconjugated NDs, AuNPs, AgNPs, quantum dot nanocarbons [60] [11]. |
| Anti-CD69 & Anti-CD25 Antibodies | Fluorescently-labeled antibodies for detecting and quantifying activated T cell populations via flow cytometry. | Flow cytometry antibodies for immune profiling [60]. |
| Cytokine ELISA Kits | For quantifying the concentration of specific inflammatory cytokines (e.g., IL-6, TNF-α) in serum or tissue homogenates. | IL-6 and TNF-α assay kits [60]. |
| Flow Cytometer | Instrument for analyzing the physical and chemical characteristics of cells, used for immunophenotyping. | Multi-laser flow cytometer for cell surface marker analysis [60]. |
| ICP-MS Instrument | For highly sensitive elemental analysis to track and quantify nanoparticle distribution in tissues. | Used for biodistribution studies of metal-based NPs [60]. |
The pursuit of effective nanoparticle-enhanced CRISPR delivery and antimicrobial coatings is inextricably linked to a thorough understanding of nanomaterial biocompatibility. Experimental evidence clearly demonstrates that nanoparticle toxicity is not a generic property but is highly specific to the material's intrinsic properties and its functionalization. Data indicate that nanodiamonds, particularly in their unconjugated form, present a more favorable tolerability profile with minimal immune activation compared to gold nanoparticles and quantum dot nanocarbons [60]. This positions them as promising vectors for sensitive therapeutic applications. The choice of nanoparticle and its surface coating is therefore a critical determinant in the safety and success of next-generation therapies. As the field advances, the integration of standardized toxicity screening, detailed mechanistic studies, and the development of novel, safer coating additives will be paramount in translating the immense potential of nanomedicine from the laboratory to the clinic.
The escalating crisis of antimicrobial resistance (AMR) necessitates a paradigm shift from conventional antibiotic treatments to innovative, targeted strategies. Within this context, two distinct technological approaches have emerged: nanoparticle-enhanced CRISPR systems and conventional antimicrobial coatings. The path to clinical translation for any new technology is governed by two critical, and often challenging, pillars: navigating the complex regulatory landscape and achieving commercially viable manufacturing scalability. This guide provides an objective, data-driven comparison of these two approaches, focusing on their regulatory and manufacturing trajectories. It is framed within a broader research thesis that posits nanoparticle-enhanced CRISPR as a disruptive, precision tool for tackling genetically encoded resistance mechanisms, while antimicrobial coatings serve as a foundational, preventative technology for controlling microbial burden on surfaces. The following sections will dissect the developmental status, regulatory hurdles, manufacturing complexities, and experimental evidence for each technology, providing researchers and drug development professionals with a clear, comparative analysis.
The two technologies represent fundamentally different mechanisms of action and stages of development.
Nanoparticle-Enhanced CRISPR Antimicrobials: This approach represents a next-generation, precision therapeutic strategy. It combines the gene-editing power of the CRISPR-Cas system, which can be programmed to selectively disrupt antibiotic resistance genes or essential genes in pathogens, with nanoparticle (NP) vectors that facilitate delivery to bacterial cells. The technology is primarily aimed at treating active, often drug-resistant, infections within a patient. As of 2025, this field is in the late preclinical and early clinical trial phase. For instance, CRISPR-nanoparticle hybrids have demonstrated the ability to reduce Pseudomonas aeruginosa biofilm biomass by over 90% in vitro, while gold nanoparticle carriers have been shown to enhance editing efficiency by up to 3.5-fold compared to non-carrier systems [3]. While CRISPR-based therapies have seen clinical success in other areas, their application as antimicrobials is still emerging [28] [9].
Conventional Antimicrobial Coatings: This technology is a preventative, surface-based intervention. These coatings, which incorporate active ingredients like silver nanoparticles (AgNPs), copper nanoparticles, or other biocides, are designed to inhibit the growth of microbes on surfaces. They are widely used on high-touch surfaces in healthcare, food processing, and public spaces to reduce microbial load and prevent the spread of infections. As a result, this is a more mature, commercially established technology [15] [61]. For example, commercial coatings like AkoTech report the ability to inhibit bacterial growth on treated surfaces by up to 99.99% [61].
Table 1: Fundamental Characteristics of the Two Technologies
| Characteristic | Nanoparticle-Enhanced CRISPR | Conventional Antimicrobial Coatings |
|---|---|---|
| Primary Mechanism | Precision gene editing to disrupt resistance or kill pathogens | Broad-spectrum, contact-based microbial inhibition or killing |
| Primary Application | Therapeutic treatment of active infections | Preventative surface protection and contamination control |
| Key Components | Cas nuclease, guide RNA, lipid or metallic nanoparticles [41] | Metal nanoparticles (Ag, Cu, Zn), polymers, biocides [11] |
| Developmental Status | Late preclinical / Early clinical trials [3] [9] | Commercially established and widely deployed [61] |
| Target Specificity | High (programmable via gRNA) | Low (broad-spectrum activity) |
Analysis of Regulatory Landscapes
The regulatory pathway for each technology differs significantly, reflecting their distinct natures as a biologic drug versus a surface-treated article or medical device.
Nanoparticle-Enhanced CRISPR Antimicrobials: Regulated as gene therapy products by agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). The regulatory framework is complex and evolving, requiring rigorous demonstration of safety, efficacy, and purity.
- Key Hurdles: The primary regulatory challenges include assessing the potential for off-target genomic effects, managing immune responses to both the nanoparticle and the bacterial Cas9 system, and demonstrating the stability and specificity of the delivered genetic cargo [3] [41]. The complex mode of action, involving both a biological effector and a delivery vehicle, creates a multi-faceted regulatory dossier. Recent clinical trials for in vivo CRISPR therapies, such as those for hereditary transthyretin amyloidosis (hATTR) delivered via lipid nanoparticles (LNPs), are establishing precedents for safety and efficacy that will inform the regulatory review of antimicrobial applications [28].
Conventional Antimicrobial Coatings: The regulatory pathway depends on the application. For coatings on medical devices or food-contact surfaces, they are regulated as articles or additives by bodies like the FDA (e.g., through pre-market notifications [510(k)]) and the Environmental Protection Agency (EPA) in the U.S., which oversees public health claims.
- Key Hurdles: The main regulatory focus is on long-term toxicity and environmental impact. For coatings that incorporate metal nanoparticles like silver, regulators require extensive data on leaching behavior, potential for environmental accumulation, and resistance development in environmental microbes [15] [11]. Standardized testing methods, such as JIS and ASTM protocols, are used to validate antimicrobial efficacy claims [15]. The pathway is generally more straightforward than for gene therapies, given the established history of similar products.
Manufacturing Scalability and CDMO Landscape
Scalable and robust manufacturing is a critical gatekeeper for clinical translation and commercial success. The outsourcing of manufacturing to Contract Development and Manufacturing Organizations (CDMOs) is a key trend for both technologies.
Nanoparticle-Enhanced CRISPR Antimicrobials: Manufacturing is highly complex, involving the production of two distinct, high-purity biological components: the CRISPR machinery (Cas protein and gRNA) and the nanoparticle delivery system (e.g., LNPs). The global market for nanoparticle contract manufacturing is experiencing significant growth, valued at US$2.8 billion in 2024 and projected to reach US$4 billion by 2030 [62]. This growth is driven by the success of LNP-based mRNA vaccines and the proliferation of nano-enabled therapies. However, scaling up presents challenges:
- Process Consistency: Ensuring batch-to-batch consistency in nanoparticle size, encapsulation efficiency, and stability is non-trivial [62] [63].
- Cost: The production of GMP-grade Cas proteins and guide RNAs is expensive.
- Analytical Demands: Rigorous characterization using techniques like dynamic light scattering (DLS) and HPLC is required [62].
- CDMOs are becoming essential "innovation partners" for developers, providing the specialized expertise and infrastructure needed to navigate these challenges [63].
Conventional Antimicrobial Coatings: Manufacturing is generally more mature and scalable. It often involves the incorporation of antimicrobial agents (e.g., AgNPs) into polymer matrices during a coating formulation process.
- Scalability: Techniques like high-pressure homogenization and solvent evaporation are well-established for producing nanoparticles and composite coatings at an industrial scale [62] [15].
- Cost-Effectiveness: The raw materials and manufacturing processes are typically less costly than those for biologics.
- Challenges: The primary scaling challenges involve ensuring the even dispersion of nanoparticles within the coating and maintaining consistent antimicrobial activity and durability over the product's lifespan [15]. The CDMO landscape for this sector is also well-developed, focusing on delivering cost-effective, high-volume production.
Table 2: Comparative Manufacturing and Regulatory Analysis
| Aspect | Nanoparticle-Enhanced CRISPR | Conventional Antimicrobial Coatings |
|---|---|---|
| Regulatory Classification | Gene Therapy / Biologic | Article / Device / Additive |
| Primary Regulatory Body | FDA Center for Biologics Evaluation and Research (CBER) | EPA, FDA (for devices), equivalent regional bodies |
| Key Regulatory Concerns | Off-target effects, immunogenicity, delivery efficiency | Leaching, environmental toxicity, durability of effect |
| Manufacturing Complexity | High (multiple biological components & nano-formulation) | Moderate (formulation and incorporation into materials) |
| CDMO Dependency | High (specialized expertise and infrastructure required) | Moderate to High (for nano-ingredient synthesis) |
| Global Market Trends | Rapid growth driven by nucleic acid therapeutics; CAGR of 6.5% for NP contract manufacturing [62] | Mature market; growth in advanced, multifunctional formulations [61] |
Experimental Data and Performance Comparison
Directly comparing the performance of these technologies requires an understanding of their different intended applications. The experimental data below highlights their respective efficacies in controlled settings.
Key Experimental Protocols
Protocol 1: Assessing CRISPR-Nanoparticle Biofilm Disruption [3]
- Objective: To evaluate the efficacy of a liposomal CRISPR-Cas9 formulation in reducing bacterial biofilm biomass.
- Methodology:
- Biofilm Formation: Grow P. aeruginosa biofilms in a 96-well plate for 48 hours.
- Treatment: Introduce liposomal nanoparticles encapsulating CRISPR-Cas9 programmed to target a biofilm-specific gene (e.g., involved in quorum sensing).
- Incubation: Treat biofilms for 24 hours.
- Quantification: Use crystal violet staining to measure the remaining biofilm biomass. The absorbance of the dissolved dye is correlated with biomass.
- Key Outcome: Liposomal Cas9 formulations reduced P. aeruginosa biofilm biomass by over 90% in vitro [3].
Protocol 2: Evaluating Antimicrobial Coating Efficacy [15] [61]
- Objective: To determine the log reduction of viable bacteria on a coated surface versus an uncoated control.
- Methodology:
- Inoculation: Apply a standardized suspension of Staphylococcus aureus or Escherichia coli onto coated and uncoated coupons.
- Contact Time: Allow the bacteria to remain on the surface for a specified period (e.g., 2 hours) at controlled temperature and humidity.
- Neutralization & Recovery: Neutralize the antimicrobial activity and recover surviving bacteria from the surface by vortexing in a neutralizer solution.
- Enumeration: Plate the recovered solution on agar, count the colony-forming units (CFUs), and calculate the log reduction.
- Key Outcome: High-performance coatings can demonstrate a >4 log (99.99%) reduction in viable bacteria under standardized testing conditions [61].
Comparative Performance Data
Table 3: Summary of Key Performance Metrics from Experimental Studies
| Performance Metric | Nanoparticle-Enhanced CRISPR | Conventional Antimicrobial Coatings |
|---|---|---|
| Reported Efficacy (In Vitro) | >90% reduction in biofilm biomass [3] | Up to 99.99% (≥4 log) reduction in surface bacteria [61] |
| Editing/Efficiency Enhancement | 3.5-fold increase with gold NPs vs. non-carrier [3] | Not Applicable |
| Speed of Action | Hours to days (requires bacterial uptake and gene editing) | Minutes to hours (on contact) |
| Specificity | High (targets specific DNA sequences) | Low (broad-spectrum, can affect non-target microbes) |
| Durability of Effect | Potentially permanent (genetic change) but transient delivery | Long-lasting (coating lifetime), but can degrade [15] |
Visualizing Workflows and Mechanisms
The following diagrams illustrate the core mechanisms and experimental workflows for the two technologies, highlighting their fundamental differences.
Nanoparticle-Enhanced CRISPR Mechanism
Antimicrobial Coating Testing Workflow
The Scientist's Toolkit: Essential Research Reagents and Materials
This section details key reagents and materials essential for conducting research in the featured fields, based on the experimental protocols and technologies discussed.
Table 4: Essential Research Reagents and Materials
| Item | Function/Description | Relevance to Technology |
|---|---|---|
| Lipid Nanoparticles (LNPs) | Synthetic nanoparticles for encapsulating and delivering CRISPR components (RNP, mRNA) into bacterial cells. Ionizable lipids are key for endosomal escape [3] [41]. | Nanoparticle-Enhanced CRISPR |
| CRISPR Ribonucleoprotein (RNP) | Pre-assembled complex of Cas9 protein and guide RNA (gRNA). Offers immediate activity, high precision, and reduced off-target effects compared to DNA plasmid delivery [41]. | Nanoparticle-Enhanced CRISPR |
| Gold Nanoparticles (AuNPs) | Inorganic nanoparticles used as carriers for CRISPR components. Can enhance editing efficiency and be surface-functionalized for targeted delivery [3] [41]. | Nanoparticle-Enhanced CRISPR |
| Silver Nanoparticles (AgNPs) | Metallic nanoparticles with potent, broad-spectrum antimicrobial activity. Generate reactive oxygen species (ROS) and disrupt cell membranes. Common active agent in coatings [11]. | Antimicrobial Coatings |
| Cationic Polymers / Polyplexes | Polymers used to complex with nucleic acids, facilitating cellular entry. An alternative non-viral delivery vehicle for CRISPR cargo [41]. | Nanoparticle-Enhanced CRISPR |
| Dynamic Light Scattering (DLS) | Analytical technique used to measure the size distribution and stability of nanoparticles in suspension. Critical for quality control in NP synthesis [62]. | Both Technologies |
| Crystal Violet Stain | Dye used to stain and quantify total biofilm biomass in a standard microtiter plate assay [3]. | Nanoparticle-Enhanced CRISPR |
| Standardized Test Organisms | Reference bacterial strains (e.g., S. aureus ATCC 6538, P. aeruginosa ATCC 15442) required for validating antimicrobial efficacy claims according to JIS or ASTM protocols [15]. | Antimicrobial Coatings |
The path to clinical translation for nanoparticle-enhanced CRISPR antimicrobials and conventional antimicrobial coatings is distinct, reflecting their different technological maturities and intended uses. Antimicrobial coatings represent a mature, commercially viable technology with a clearer, well-defined regulatory pathway focused on material safety and environmental impact. Their primary challenge lies in enhancing durability and mitigating long-term ecological concerns. In contrast, nanoparticle-enhanced CRISPR represents the cutting edge of precision antimicrobials, still navigating the complex regulatory landscape of gene therapies. Its major hurdles are unequivocally demonstrating safety—particularly regarding off-target effects and delivery specificity—and establishing scalable, cost-effective manufacturing processes for its complex biological components.
The future of combating AMR will likely involve a synergistic approach. Conventional coatings will continue to play a vital role in infection prevention and control in healthcare and industrial settings. Meanwhile, the successful clinical translation of nanoparticle-enhanced CRISPR systems could revolutionize the treatment of drug-resistant infections, offering a programmable, targeted therapeutic platform. For researchers and developers, the choice between these technologies is not one of superiority, but of application: preventative surface protection versus therapeutic intervention against genetically encoded resistance.
Conclusion
The comparative analysis reveals that nanoparticle-enhanced CRISPR and conventional antimicrobial coatings represent complementary, rather than competing, paradigms in infection control. CRISPR-based strategies offer an unprecedented, precise tool for targeting the genetic roots of antibiotic resistance and biofilm formation, showing remarkable efficacy in preclinical models, such as over 90% biofilm biomass reduction. Conventional coatings provide a robust, broad-spectrum first line of defense on surfaces, capable of significant bacterial reduction post-cleaning. The future lies in interdisciplinary approaches that may integrate these technologies—for instance, using coatings for passive protection on medical devices while employing CRISPR-based therapies for active treatment of established, resistant infections. Overcoming delivery challenges for CRISPR and enhancing the durability and scope of coatings will be critical. Ultimately, the choice between these technologies will be guided by the specific clinical need: immediate surface protection or long-term, precision eradication of persistent pathogens.
References
- 1. Contact-killing antibacterial mechanisms of polycationic coatings: A review - ScienceDirect [https://www.sciencedirect.com/science/article/abs/pii/S0300944024000067]
- 2. Conventional methods and future trends in antimicrobial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9958398/]
- 3. integrating CRISPR/Cas9 and nanoparticles against biofilm ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12366227/]
- 4. Nano-scaled advanced materials for antimicrobial applications [https://pubs.rsc.org/en/content/articlehtml/2025/en/d4en00798k]
- 5. Revolutionizing Antimicrobial Solutions Nanotechnology ... [https://microbiologyjournal.org/revolutionizing-antimicrobial-solutions-nanotechnology-crispr-cas9-and-innovative-approaches-to-combat-drug-resistance-in-eskape-pathogens/]
- 6. Evaluation of different antimicrobial polymeric coatings for food contact surfaces | Discover Food [https://link.springer.com/article/10.1007/s44187-025-00487-3]
- 7. Bio-inspired biomaterial coating for enzyme responsive release of antimicrobial peptides - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12181015/]
- 8. CRISPR/Cas9 therapeutics: progress and prospects [https://www.nature.com/articles/s41392-023-01309-7]
- 9. Emerging technologies and innovative approaches to ... [https://www.sciencedirect.com/science/article/pii/S3050723525000033]
- 10. Antimicrobial Nanocoatings Market Trends, 2025 to 2035 [https://www.futuremarketinsights.com/reports/anti-microbial-nanocoatings-market]
- 11. Antimicrobial nanoparticles: a new horizon to combat ... [https://www.frontiersin.org/journals/nanotechnology/articles/10.3389/fnano.2025.1611126/full]
- 12. CRISPR's efficiency triples with DNA-wrapped nanoparticles [https://news.weinberg.northwestern.edu/2025/09/18/crisprs-efficiency-triples-with-dna-wrapped-nanoparticles/]
- 13. Advancing gene editing: the role of lipid nanoparticles in ... [https://www.drugtargetreview.com/article/188056/advancing-gene-editing-the-role-of-lipid-nanoparticles-in-crispr-delivery/]
- 14. Top 10 Antimicrobial Coating Companies to Watch in 2025 ... [https://www.researchandmarkets.com/articles/key-companies-in-antimicrobial-coating?srsltid=AfmBOorTGr48QvoRclAw7s0ye1A-tAPrpRq_0BQTX9zRu94a89kXec1l]
- 15. Advances in antimicrobial polymeric coatings for food ... [https://www.sciencedirect.com/science/article/pii/S2950194625003796]
- 16. Nanomaterial-enabled anti-biofilm strategies - RSC Publishing [https://pubs.rsc.org/en/content/articlehtml/2025/nr/d4nr04774e]
- 17. Advancing Nanotechnology: Targeting Biofilm-Forming ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11876546/]
- 18. Biofilms: structure, resistance mechanism, emerging control ... [https://pubs.rsc.org/en/content/articlehtml/2025/pm/d5pm00094g]
- 19. Nanotechnology-Based Delivery of CRISPR/Cas9 for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12346122/]
- 20. CRISPR/Cas9 gene editing strategy for cancer therapy [https://www.sciencedirect.com/science/article/abs/pii/S0141813025089469]
- 21. Combating Antimicrobial Resistance: Innovative Strategies ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12389514/]
- 22. Innovative approaches to combat antibiotic resistance: integrating... [https://pubmed.ncbi.nlm.nih.gov/40830872/]
- 23. amphiphilic copolymers as antimicrobial materials [https://pubs.rsc.org/en/content/articlehtml/2025/ma/d5ma00335k]
- 24. The Evaluation of Antibacterial Coatings Against ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11769392/]
- 25. integrating CRISPR/Cas9 and nanoparticles against biofilm ... [https://bmcmedicine.biomedcentral.com/articles/10.1186/s12916-025-04323-4]
- 26. Viral vs. Non-Viral Gene Delivery: A Critical Choice for ... [https://www.drugdiscoverynews.com/viral-vs-non-viral-gene-delivery-a-critical-choice-for-gene-therapy-developers-16819]
- 27. innovations and challenges in rAAV vector-based CRISPR ... [https://www.nature.com/articles/s41434-025-00573-2]
- 28. CRISPR Clinical Trials: A 2025 Update [https://innovativegenomics.org/news/crispr-clinical-trials-2025/]
- 29. CRISPR's Efficiency Triples With Spherical Nucleic Acid ... [https://news.feinberg.northwestern.edu/2025/09/05/crisprs-efficiency-triples-with-spherical-nucleic-acid-delivery-system/]
- 30. Effect of conventional dipping and twin-nozzle electrostatic ... [https://www.sciencedirect.com/science/article/pii/S2772502225001398]
- 31. Effective adsorptive removal of triclosan from water using ... [https://www.frontiersin.org/journals/chemistry/articles/10.3389/fchem.2025.1547169/full]
- 32. Dual-functional surface coatings integrating antimicrobial and ... [https://pubs.rsc.org/en/content/articlehtml/2025/mh/d5mh01132a?page=search]
- 33. Better models, better treatment? a systematic review of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12061920/]
- 34. A Semi-Solid In Vitro Biofilm Model for Evaluating ... [https://www.sciencedirect.com/science/article/pii/S2590207525000760]
- 35. Antimicrobial Coatings for Medical Device market [https://www.24chemicalresearch.com/reports/184149/global-antimicrobial-coatings-for-medical-device-market]
- 36. The Global Challenge of Antimicrobial Resistance [https://pmc.ncbi.nlm.nih.gov/articles/PMC12284435/]
- 37. Addressing antimicrobial resistance with Cas9, Cas12, ... [https://www.sciencedirect.com/science/article/pii/S2405844025003937]
- 38. CRISPR-Cas Systems: Transformative Precision Tools to ... [https://www.genesispub.org/crispr-cas-systems-transformative-precision-tools-to-combat-antimicrobial-resistance-in-multidrug-resistant-gram-negative-pathogens]
- 39. CRISPR challenges in clinical developments [https://www.sciencedirect.com/science/article/abs/pii/S1877117324001704]
- 40. Challenges and Opportunities in the Application of CRISPR ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12503735/]
- 41. CRISPR Delivery Methods: Cargo, Vehicles, and Challenges [https://www.synthego.com/blog/delivery-crispr-cas9/]
- 42. Advances in Nanomaterial‐Mediated CRISPR/Cas ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2025.1669104/abstract]
- 43. Lipid-Nanoparticle-Based Delivery of CRISPR/Cas9 Genome ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9176214/]
- 44. Design of experiments in the optimization of nanoparticle- ... [https://www.sciencedirect.com/science/article/abs/pii/S0168365923003073]
- 45. UW researchers test common disinfectants' abilities to fight ... [https://www.washington.edu/news/2025/08/11/test-common-disinfectants-fight-antibiotic-resistance-genetic-level-bacteria-hospitals-health-care/]
- 46. Reinvigorating AMR resilience: leveraging CRISPR–Cas ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11963374/]
- 47. CRISPR-Cas Systems in the Fight Against Antimicrobial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11608035/]
- 48. Unravelling the advances of CRISPR-Cas9 as a precise ... [https://www.sciencedirect.com/science/article/pii/S2213716525000360]
- 49. JIS Z 2801 - Test for Antibacterial Activity of Plastics [https://microbe-investigations.com/jis-z-2801-antibacterial-testing-of-plastics-and-non-porous-surfaces/]
- 50. JIS Z 2801 Test for Antimicrobial Activity of Plastics [https://microchemlab.com/test/jis-z-2801-test-antimicrobial-activity-plastics/]
- 51. Mechanisms of antimicrobial resistance in biofilms [https://www.nature.com/articles/s44259-024-00046-3]
- 52. Antimicrobial Coatings: Reviewing Options for Healthcare ... [https://www.mdpi.com/2673-8007/3/1/12]
- 53. Comparative performance of a panel of commercially ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4230231/]
- 54. Biofilms: Formation, drug resistance and alternatives to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9576500/]
- 55. Antimicrobial resistance (AMR) management using ... [https://www.sciencedirect.com/science/article/pii/S2666388024000017]
- 56. CRISPR's efficiency triples with DNA-wrapped nanoparticles [https://news.northwestern.edu/stories/2025/09/crisprs-efficiency-triples-with-dna-wrapped-nanoparticles]
- 57. CRISPR screening redefines therapeutic target ... [https://www.sciencedirect.com/science/article/pii/S2095177925001741]
- 58. CRISPR Off-Target Editing: Prediction, Analysis, and More [https://www.synthego.com/blog/crispr-off-target-editing/]
- 59. Structural parameters of nanoparticles affecting their toxicity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9970140/]
- 60. Toxicity and biodistribution comparison of functionalized ... [https://www.frontiersin.org/journals/nanotechnology/articles/10.3389/fnano.2025.1512622/full]
- 61. Discover Microban's Antimicrobial Coatings at the 2025 CTT ... [https://www.microban.com/news/microban-antimicrobial-coatings-ctt-summit-2025]
- 62. Nanoparticle Contract Manufacturing Strategic Business [https://www.globenewswire.com/news-release/2025/05/28/3089268/28124/en/Nanoparticle-Contract-Manufacturing-Strategic-Business-Report-2025-Outsourcing-Trend-in-Pharma-and-Biotech-Boosts-Demand-for-End-to-End-Nanoparticle-CDMO-Solutions.html]
- 63. Trends Shaping the Future of Cell and Gene Therapy ... [https://www.agcbio.com/biopharma-blog/trends-shaping-the-future-of-cell-and-gene-therapy-manufacturing]